Hank Hogan
For University of Pittsburgh scientists investigating the molecular signaling between compartments of a neuron’s dendrites and the accompanying cellular modifications that play a part in long-term memory, traditional microscopes were inadequate. To test their hypotheses, the researchers had to simultaneously image events occurring in the neuron’s secretory pathway, including the endoplasmic reticulum organelle and the plasma membrane.
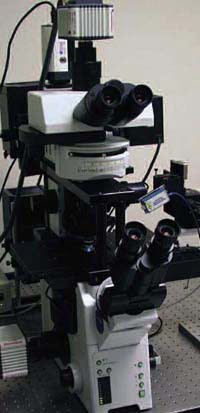
The microscope consists of upright and inverted research instruments mounted atop each other. Two optical paths enable the simultaneous use of two imaging techniques. Courtesy of Kenneth N. Fish, University of Pittsburgh.
“I would need to build the Bugatti of microscopes,” quipped Kenneth N. Fish, an assistant professor of psychiatry at the university.
Fortunately, Olympus America Inc. of Melville, N.Y., a subsidiary of Olympus Corp. of Tokyo, was in the process of developing what Fish required for his work. Late in 2005, the company announced a package that combines an upright and an inverted research microscope into one instrument, the Up/Down Microscope.
Nicolas B. George, a group manager for research microscope systems at Olympus America, said the company constantly receives requests for more microscope flexibility. Mechanically merging the two instruments allows researchers to choose the best imaging method for a given situation. George noted that plans call for extending the microscope’s capabilities — for example, offering a spinning disk confocal module for the upper microscope that will complement the total internal reflection fluorescence module available for the lower one.
Fish worked with the instrument in its development stage. Today he uses a more polished setup to follow proteins from endoplasmic reticulum export to plasma membrane targets. Employing two fluorescent labels, he images one from the top while using Förster resonance energy transfer to image the other from the bottom. When the two are collocalized in the cell, he switches to total internal reflection fluorescence imaging, using a third label bound to a peptide in the surrounding media. In this way, he can follow a particular signaling subunit as it moves from the endoplasmic reticulum to the plasma membrane, determining when it reaches the plasma membrane and ensuring that it is part of a mature receptor complex.
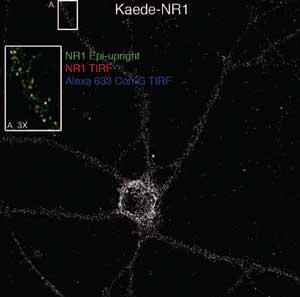
The microscope images neural NR1 subreceptors tagged with the green-emitting fluorophore Kaede. The inset shows a magnified view of the region labeled A, with images taken by total internal reflection fluorescence using the bottom microscope overlaid those taken using the upper instrument. Blue, yellow and white indicate receptors in various stages.
The current thinking is that transmembrane proteins leaving the endoplasmic reticulum in dendrites return to within 100 µm or so of the nucleus-containing soma for processing. However, recent studies have shown that proteins can leave the endoplasmic reticulum at least 350 µm from the soma and that signals arriving at the plasma membrane there can result in increases in local endoplasmic reticulum export. Because the idea of proteins traveling back and forth for processing seems to make little sense, the researchers hypothesize that the requisite modifying enzymes exist in these distant locations.
Although there is not yet a definitive answer, Fish said that the results from the initial experiments are intriguing. They have shown that some proteins leaving the endoplasmic reticulum in tertiary branches actually travel to even more distant regions on their way to the local plasma membrane.
Getting to this point has required some unexpected technological development. For instance, there was no software capable of running two microscope Z-motors at the same time. Another issue was that the software drivers used by the cameras from Hamamatsu Corp. of Bridgewater, N.J., for image capture did not allow for two devices to be used in the same way simultaneously. Alignment between the microscopes also was a challenge. These problems were solved through back-and-forth efforts among Fish, Olympus, Hamamatsu and software maker Intelligent Imaging Innovations Inc. of Denver.
The effort has been worth it, the scientist said. He doubts that a conventional system could have produced the amount of data the Up/Down Microscope has and believes that it also would have suffered other drawbacks.
“True simultaneous imaging of two different planes is something it would never be capable of doing,” he said.
Contact: Kenneth N. Fish, University of Pittsburgh, Pittsburgh, Pa.; +1 (412) 648-9366; e-mail: [email protected]. Nicolas B. George, Olympus America Inc., Melville, N.Y.; +1 (631) 844-5107; e-mail: [email protected].