
Watching muscles work
Hank Hogan
When muscles contract, they do so thanks to thousands of tiny biological motors. The motors pull on filaments, which slide past one another, causing the muscles to become shorter, even though the filaments do not. Although this theory has been around for decades, researchers still have questions about the workings of sarcomeres — basic contractile units of striated muscle composed of myosin motors and associated actin filaments.
Because of an inability to visualize individual sarcomeres in place, researchers previously studied muscle tissue by taking invasive measurements of cadavers and animals. Those methods, however, yielded only averages. Now researchers from Stanford University in California have created a minimally invasive microendoscopy system that images sarcomeres at work, and they demonstrated its effectiveness on live mice and humans.
The tool could help in understanding neuromuscular diseases that involve mutations in sarcomeric proteins. A better understanding of how sarcomeres work also could help the biomechanical analysis used in preoperative diagnostics, rehabilitation and athletics.
The researchers constructed the optical microendoscope using gradient refractive index microlenses from Grintech GmbH of Jena, Germany. They clad the lenses — which were 350 to 1000 μm in diameter — in stainless steel tubes for insertion and minimally invasive imaging.
In building their device, they adapted a laser-scanning microscope from Prairie Technologies Inc. of Madison, Wis. The microscope objective focuses ultrashort pulsed laser illumination from a wavelength-tunable Ti:sapphire laser from Spectra-Physics onto the face of the gradient refractive index microendoscope. The excitation and resulting emission traveled part of the way along the same path, with a dichroic mirror sending the emission off to a photomultiplier tube for detection.
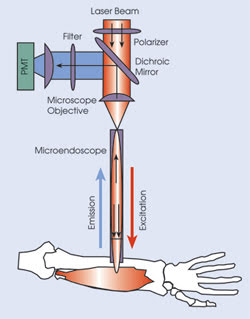
Researchers used a laser-scanning imaging system to visualize sarcomeres in live subjects. A microscope objective focuses the laser onto the face of a gradient refractive index microendoscope. The microendoscope demagnifies and refocuses the beam within the muscle and returns emitted light signals that reflect off a dichroic mirror before detection by a photomultiplier tube (PMT).
Because they wanted to avoid the use of external labels, the researchers looked at two naturally occurring signals known to allow sarcomere imaging and known to arise from different parts of the contractile unit. The first was autofluorescence from nicotinamide adenine dinucleotide (NADH) and flavoproteins, which are concentrated in mitochondria. The second was a second-harmonic-generated signal from the myosin rod domains.
The researchers found that second-harmonic generation with 920-nm illumination provided better visibility. With this technique, they looked at average sarcomere length and variability within individual muscle fibers and adjacent neighbors in anesthetized mice. They found up to a 20 percent variation within a 25-μm diameter, a result, they noted, that could not have been discovered without being able to examine individual sarcomeres.
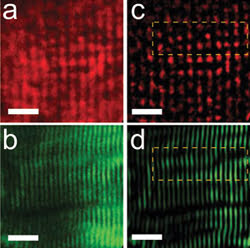
The researchers also used the system to image single mouse muscle fiber in culture. They acquired images of the same fiber using epi-detection of two-photon-excited autofluorescence (a) as well as transdetection of second-harmonic generation (b). Bandpass-filtered versions of (a) and (b) are shown in (c) and (d), with the yellow boxes indicating the regions of interest used to make (a) and (b).
They also looked at changes in humans, asking volunteers to contract and extend their fingers while the microendoscope captured sarcomere lengths via high-speed video. They saw systematic variations in length of about 0.15 μm as a result of the movement. The volunteers reportedly experienced little discomfort.
The researchers believe that such imaging could provide clinically important information about neuromuscular disorders in the future. Being able to visually measure the basic unit of muscle contraction could help diagnose the severity of the problem and the effectiveness of treatment.
Nature, July 06, 2008, pp. 784-788.
Published: September 2008