
Two photons monitor neurons
Technique allows optical activation of neural activity.
Hank Hogan
Going longer in wavelength could help reveal the hidden functioning of neuronal circuits. This is possible courtesy of a completely noninvasive optical method that a group of researchers at the University of California, Irvine, developed to identify, activate and detect the activation of specific groups of cells. A combination of ultrafast lasers, confocal microscopy and genetics allows manipulation of cells or even subcellular systems, enabling high-resolution study of neuronal activity.
The researchers used a near-infrared microbeam generated by a Coherent femtosecond laser. Researcher Samarendra K. Mohanty noted that this spectral region was chosen for good reason, saying that, if you want to go deep, you have to use near-infrared excitation wavelengths.
The researchers used a multiphoton-activation approach to take advantage of recent advances. Various researchers have used genetic manipulation techniques to introduce the light-activated molecule channelrhodopsin-2 (ChR2) into specific groups of electrically excitable cells such as neurons. The modified neurons express the channel, and it responds to light, providing researchers a way to target specific cells. This approach eliminates the need to place electrodes inside every single neuron being studied — a challenging task.
The light intensity required to trigger ChR2 is low, so that a filtered lamp or a small laser diode is sufficient. However, the peak absorption for the channel is in the blue, around 460 nm. Light around this wavelength is strongly absorbed and scattered by tissue, making the channel less useful when working with living tissue or animals, or when interrogating cells deep within a specimen.
Going deeper
Mohanty noted that, to circumvent the problem, there is a definite need for new approaches. For reasons having to do with the nature of ChR2 and the desire to selectively activate cells or subcellular regions, the researchers elected to use a multiphoton microbeam method. Such an approach would allow them to work with an unmodified version of ChR2 while enabling imaging at greater depths. At the same time, the response to the excitation light would come from a much more restricted cellular volume.
The researchers transfected HEK 293 cells with a construct of ChR2 fused with enhanced yellow fluorescent protein. Then they incubated the strongest fluorescing cells with calcium orange, a dye that indicates the release of calcium ions and associated neuronal activity. They followed an analogous procedure in preparing a hippocampal tissue slice so that cells within it would have a similar behavior.
They chose calcium orange and the enhanced yellow fluorescent protein in part because the response of the two is spectrally separated, with the dye’s excitation peak around 540 nm and its emission detected above 600 nm. In contrast, the protein’s excitation peak is around 510 nm and its emission peak about 40 nm higher. In addition, the excitation spectrum of ChR2 doesn’t overlap with that of the marker, the yellow fluorescent protein. This combination of spectral characteristics is what allowed optical detection of the channel activation process.
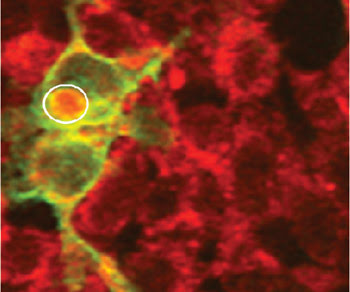
The encircled region in a ChR2-expressing HEK 293 cell is identified by yellow fluorescent protein expression detected in the green channel. The region shows an increase in calcium fluorescence (calcium orange in red channel) subsequent to two-photon activation using a 916-nm near-IR microbeam. Image courtesy of Samarendra K. Mohanty, University of California, Irvine.
In a demonstration, the researchers identified the transfected cells by confocal imaging, using a Zeiss laser scanning microscope. They then used the near-infrared microbeam, tuning it from 860 to 1028 nm to compare the relative activation efficacy of various wavelengths. As part of their study, they determined the threshold for two-photon intensity below which there was no damage to the cells. That provided them with an upper intensity threshold, Mohanty noted. “Within this limit, the scanning near-IR microbeam could activate the ChR2-expressing cells.”
Calcium imaging
Because they detected this activation using calcium imaging, the process was completely optical and noninvasive. In confirming their activation method, they performed some patch clamp recording, attaching an electrode to cells and monitoring the optoelectrophysiology of the cells. They did this for only a limited number of cells and single-photon activation, though.
They compared the single-photon and two-photon activation spectrums. They looked at the effects of different activation wavelengths spanning from 458 to 514 nm for the single-photon source and the corresponding two-photon range from 916 to 1028 nm. In both cases, they found that the shorter wavelengths produced the strongest response. The two-photon excitation required a higher average intensity and produced a nonlinear response, a result that Mohanty noted was not surprising.
Finally, the researchers also examined two-photon activation versus depth. They found that, again as expected, the two-photon method yielded greater activation than the single-photon approach, with the difference growing larger at greater depths. The work was published in the July 11, 2008 online version of Biophysical Journal.
The next step for the group, having demonstrated the technique, will be to use it to help understand the signaling and role of different layers in the retina. Ultimately, the goal is to intervene in the progress of diseases such as retinitis pigmentosa, an inherited eye disorder.
Mohanty noted that the group would like to see an affordable and compact ultrafast two-photon source coupled to fiber optics. Ideally, the fiber bundle would also collect the resulting fluorescence.
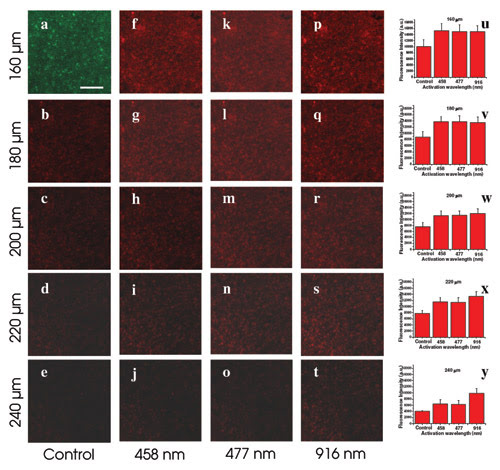
Longer wavelengths lead to activation at greater depth, as shown in this comparison of the maximum depth from which calcium orange fluorescence is detected. Researchers transfected neuronal cells in a mouse hippocampus to express the light-activated molecule ChR2 and a marker yellow fluorescent protein. They detected activation using calcium orange fluorescence. They observed ChR2 expression before activation at depths of 160, 180, 200, 220 and 240 μm, respectively (a-e). They looked at single-photon activation with a laser microbeam at 458 nm and at 477 nm at the same depths (f-j and k-o), as well as at two-photon irradiation with a near-IR laser microbeam at 916 nm (p-t). The histograms show fluorescence intensity as a measure of activation efficiency vs. wavelength of activation at depths of 160, 180, 200, 220 and 240 μm (u-y). The scale bar in (a) represents 200 μm. Courtesy of Samarendra K. Mohanty, University of California, Irvine. Reprinted with permission from Biophysical Journal.
One of the issues limiting the new method, the researchers reported, is the 543-nm excitation wavelength of calcium orange. It doesn’t penetrate tissue as well as near-IR does, and that limits imaging at depth.
A two-photon source at the right wavelength would solve that problem. However, that would require two separate two-photon sources, with one about 900 nm for activation and the other 1100 nm for imaging. Getting that into a compact arrangement would require new technology but could provide some benefits, Mohanty said. “Two-photon activation and calcium imaging using two near-IR wavelengths that can be switched fast and that are programmable would enable imaging at larger depths.”
Published: September 2008