RICHLAND, Wash., Sept. 25, 2008 – While a group of fluorescent molecules may appear to work synchronously, a closer look reveals maverick molecules flash and work at varying rates, according to scientists at the Department of Energy’s Pacific Northwest National Laboratory (PNNL).
As a single fluorescent molecule flashes as it gains or loses an electron. The PNNL team is using a combination of two techniques that allow them to load or empty fluorescent dye with electrons, then watch for the individual flashes of light as molecules gained and lost their parcels.
The researchers hope this method will let them better understand so-called electron transfer reactions and also hope it will set the stage for a better understanding of the underlying principles of certain reactions common to biofuel production.
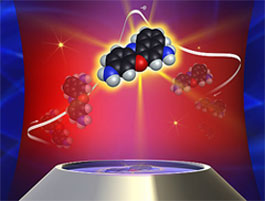
A combination of well-known techniques allows researchers to control and visualize fluorescent molecules one at a time. In this illustration, fluorescent microscopy (at bottom) picks up a flash of light from a single cresyl violet molecule as it loses its expendable electron (top). The method will allow researchers to pick apart deeper underlying principles of so-called electron transfer reactions. (Photo: PNNL)
Electron transfer reactions are central to processes that capture the sun's energy via photosynthesis or that allow cells to generate the energy needed to sustain life. Engineers and chemists studying renewable energy want to reverse-engineer photosynthesis and similar processes to create hydrogen and other biofuels. These reactions rely on electrons jumping from one molecule to another: passing the electrons along releases their chemical energy so it can be applied to something practical, such as making sugar in plants.
"If we could understand these electron transfer reactions better, then we could better design the proteins that generate energy," said PNNL project lead Eric Ackerman.
When studying these reactions, chemists generally look at a whole group of molecules reacting and measure the average energy they give off, much like looking at a whole organization of workers and averaging their productivity.
The team combined two well-established techniques to zoom in on electron transfer reactions. In one, called cyclic voltammetry, a molecule fluoresces when zapped with electricity. The PNNL team chose a dye called cresyl violet: at a particular voltage, the dye molecule loses an electron and in the process sends out a flash of white light.
But traditional cyclic voltammetry allows studying such molecules only in big batches. Being able to watch individual molecular reactions would be like being able to see the details in the frames of a film.To view single molecules, PNNL chemists Chenghong Lei and Dehong Hu combined forces. Lei built an electrochemical cylinder about the size of a brazil nut that held a drop of dye. The team fitted this onto Hu’s single-molecule fluorescent microscope at EMSL, DOE's Environmental Molecular Sciences Laboratory on the PNNL campus. This set-up resulted in a new instrument capable of performing both techniques simultaneously.
By rapidly cycling the voltage up and down, the team found that some individual molecules were a bit erratic. Although most of the molecules behaved as expected, some that should have been lit, weren't. And turning off the electricity, all should have blinked out but some kept burning in the absence of a voltage.
"What we want to know is what causes these variations. And can we control these variations in a favorable way with external experimental and environmental conditions?" said Lei.
Studying molecules one at a time can provide insights into chemical reactions that can't be observed by studying them en masse. Such insights could help chemists improve upon the design that nature gave them. Their new instrument provides a way to study and control the molecules.
"If we could make a desired molecule consistently work at its maximum rate, that would be a big improvement," said Ackerman.
In addition, the researchers said they plan to use cresyl violet or another fluorescent molecule side-by-side, or even incorporated into proteins of interest, such as those involved in electron transfer reactions that generate biofuels. As the proteins gain or lose electrons, the fluorescent components would lose or gain theirs to reveal what is going on.
This work was supported by the Department of Energy's Basic Energy Sciences, part of the Office of Science.
For more information, visit: www.pnl.gov