The eyes have it, at least when it comes to new, noninvasive diagnostics. For example, there’s optical coherence tomography, or OCT. Improved OCT technology promises better monitoring and treatment of glaucoma and age-related macular degeneration, two leading causes of blindness. Other innovations involve wavefront sensing, where technological innovations could result in better lenses, more accurate lasik for correcting vision and better imaging of the eye. A look shows what’s possible now and where things may be headed.
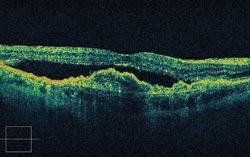
An OCT image shows, in cross section, distortion of the retina resulting from wet age-related macular degeneration. Courtesy of Carl Zeiss Meditec.
Mapping mountains
For those interested in eyes, OCT offers many advantages. An interferometric technique, OCT allows imaging of structures millimeters deep within the eye with micron resolution. One problem has been speed: OCT is slow enough that movements in the eye have distorted data, unless sparse scan patterns and limited sampling density were employed. That situation has changed, however, with the advent of spectral-domain OCT, which is fast enough that much denser scans can be used and 3-D data cubes thereby collected. The artifacts that remain typically don’t make a difference.
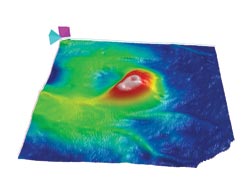
Scanning the retina with OCT details the state of its health. Here, a topographical map shows thickening due to new blood vessels, a condition known as wet age-related macular degeneration. Courtesy of Carl Zeiss Meditec.
An example of this transformation can be seen in a fourth-generation retinal OCT product from Carl Zeiss Meditec of Dublin, Calif. According to Scott Meyer, the company’s director of strategic business development, the system acquires data 65 times faster than previous products could. Other companies that offer similar spectral-domain OCT systems include Heidelberg Engineering of Germany, Topcon Medical Systems of Paramus, N.J., and Bioptigen of Research Triangle Park, N.C.

Mapping the effect on the retinal pigment epithelium of drusen – deposits that accumulate behind the photoreceptors during dry age-related macular degeneration. With OCT imaging, the progress of the disease and the response to treatment can be monitored. Courtesy of Carl Zeiss Meditec.
The increase in speed allows the collection of more diagnostically useful data. That capability has some significant implications, said Philip J. Rosenfeld, a professor of ophthalmology and a retina specialist at the University of Miami in Florida. “We have much more confidence that the numbers and the maps that we get out of spectral-domain OCT are reproducible and more reliable.”
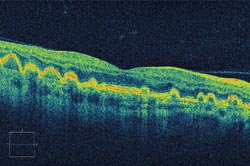
A cross section of the retina, via OCT, reveals that the drusen associated with dry age-related macular degeneration creates undulations in the retina, thus distorting vision. Courtesy of Carl Zeiss Meditec.
Rosenfeld uses OCT primarily to look at the structure of the retina and the interface between the retina and the tissues above and below it. He’s particularly interested in imaging dry macular degeneration, a disease in which the photoreceptor layer in the back of the eye develops deposits of hardened extracellular material called drusen. He likens these accumulations to mountain ranges, with the photoreceptor layer thinnest where the drusen peaks are highest.
Drusen can evolve into what is known as geographic atrophy, a more advanced form of the disease with significant vision loss. For that reason, being able to track the growth of these deposits and their response to treatment is important. Thanks to OCT advances, Rosenfeld’s group has made progress in this area. “One of the algorithms that has been developed here is able to provide quantitative analysis of drusen volume,” he said.
The algorithms subtract an assumed normal thickness of the layer from the measured thickness, resulting in an expression of thickness over area. This way of evaluating the health of the eye’s macula has its roots in a grid-based system originally developed years ago.
As for the future, Rosenfeld would like to have more reliable algorithms and to be able to image deeper into the eye. He’d also like to have still faster systems with higher resolution. However, the amount of data generated by the latest systems already strains storage and data analysis. Upping the data rate would make that problem more acute.
OCT also can play a role in treating glaucoma, a disease characterized by progressive damage to the optic nerve and associated with increased pressure inside the eye. Untreated, glaucoma leads to vision loss and eventual blindness. All therapies currently approved by the FDA are directed at lowering the intraocular pressure, which in turn slows or halts the progression of the disease.
The problem is that one person may suffer damage at a low pressure while someone else may be able to tolerate high pressure for years without apparent effect. Thus, a pressure reading is an inexact indicator. With OCT, it is possible to make a direct reading on the parts of the eye damaged by glaucoma.
“We do measurements on the tissues relevant to the disease,” said Joel S. Schuman, a professor of ophthalmology at the University of Pittsburgh and director of its School of Medicine Eye Center. He is one of the inventors of the OCT ophthalmology technique, with intellectual property licensed by the University of Pittsburgh to Bioptigen. He also receives royalties for intellectual property licensed by MIT to Carl Zeiss Meditec.
Getting ahead with OCT
Schuman explained that the data from the glaucoma-relevant measurements from a patient is compared with the results from a healthy population. It also can be compared with the patient’s own previously recorded profile. These comparisons enable doctors to know with more certainty who has early-stage glaucoma and who is getting worse. They also allow physicians to know how aggressively the disease should be treated.
The current crop of OCT systems gathers three-dimensional data. Such volumetric representations of the eye are important when planning a surgical intervention, which is sometimes done when treating glaucoma. For diseases where retinal surgery is involved, Schuman would like to have a system that performs OCT through a microscope or via a fiber probe. That, he said, would allow OCT of the eye through an endoscope. “I see that as having a utility in intraoperative OCT.”
He added that other advances of likely benefit would be even faster scanning and better axial and transverse resolution. The use of different wavelengths appropriate to the structure also would help, with longer wavelengths allowing imaging deeper into the eye.
Opening up a new wavefront
Of course, OCT isn’t the only ophthalmology-related innovation. There also are advances taking place in wavefront sensing, which could bring benefits to everything from laser-based vision correction to high-resolution retinal imaging. One example of this comes from PhaseView, which is based in Palaiseau, France, with offices in Seattle.
Wavefront sensing measures the aberrations in light waves passing through a lens or reflecting off the retina, for example. Correcting such deviations improves optical quality. In theory, this can allow an optical system, such as the eye or an instrument imaging the eye, to achieve perfect performance.
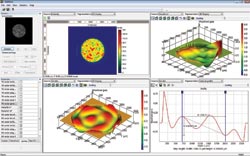
Shown is the capture of the ophthalmology wavefront, courtesy of a digital wavefront camera and associated software. In the upper left is a live image of the retina, followed in the upper right by an image in false colors and the wavefront with aberrations visible. Shown at bottom are an image of high-order aberrations and related data. Correcting such aberrations with lenses or lasers results in better eyesight. Courtesy of PhaseView.
The most common wavefront sensing methods involve Shack-Hartmann lenslet arrays, and these are the basis for the majority of ophthalmology applications. PhaseView, however, uses a lensless procedure that measures differences in intensity on several focal planes. From that data, its systems derive the wavefront.
The advantages of this approach, according to Gael Launay, marketing and sales manager for PhaseView, are that every pixel of the sensor is used, and the resolution is not limited by the size of a lenslet array. The technique also simultaneously provides true high-resolution intensity and phase data with high-dynamic-range wavefront measurements, something Launay said is not possible with the Shack-Hartmann approach. Those attributes allow reconstruction of the image of the wavefront with no difference between a video image and phase data information. “What you see is what you get,” Launay said.
The disadvantage is that the approach is computationally challenging, but PhaseView claims to have solved this problem. Its current systems can go up to a 25-Hz measurement rate for an aperture diameter up to 8 mm. Future products may go even faster, since the company is currently testing the use of more powerful computational engines involving field-programmable gate arrays or digital signal processors.