Frequency-domain technique aids correct placement of implants.
Alex Cable and Lori Howe, Thorlabs Inc.
Cochlear implants are used to stimulate directly the nerve structures in patients with nonfunctional receptor cells. Such electrodes traditionally have been used only in deaf patients, but recently these devices have been implanted in patients with only partial hearing loss, making the exact placement of the device more critical for auditory function.
The implant must be located to the right side of the membrane (scala tympani) for optimal function, and the location of the hole and electrode implant must be within 0.5 mm. However, placement of the implant is complicated because the cochlea is deep in the head, and during standard surgical procedures only a small opening is created, making visualization and localization challenging. Further complicating the procedure is the large working distance required, which limits the usefulness of standard surgical microscope techniques.
Recently, researchers from the University of Rostock in Germany obtained real-time images of a human cochlea by combining a frequency-domain optical coherence tomography (OCT) imaging system with a surgical microscope, which enabled more precise device location during cochlear implant surgery.1,2 This technique allows surgeons to localize exactly the scala tympani before opening the fluid-filled inner ear when inserting the electrode array of a cochlear implant (Figure 1).
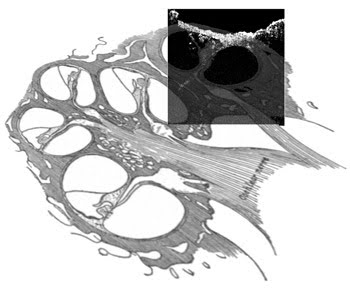
Figure 1. A schematic of the inner ear anatomy is shown with an overlay of the imaged area.
Most cochlear implant surgery involves drilling a hole through the promontory bone and exposing the scala tympani membrane from below. If possible, the covered membrane should be left intact until there is an appropriate-size opening for the proper insertion of the electrode implant. Implantation surgery in patients with partial hearing loss makes the proper identification of location and the minimized exposure of the delicate portions of the inner ear essential to retaining audiological functioning. This is particularly significant when the surgery is performed for combined electric and acoustic stimulation in patients with significant residual hearing at low frequencies. Membranes should be opened in a controlled and careful manner with special surgical instruments that create the smallest possible opening to avoid complications.
How it works
OCT can help with placement of the implant because it provides real-time noninvasive 2- and 3-D imaging with depths of a few millimeters and a resolution of a few microns, depending on the application. Frequency-domain OCT achieves greater sensitivity and higher imaging speed than time-domain techniques used previously, making it a powerful tool in biomedical applications, including real-time surgical guidance.
The technique uses light with a short coherence length to create an image from coherent backscattered light. A broadband light source is used because a broader bandwidth results in higher axial resolution, though the lateral resolution depends on the imaging optics. The light is split into two paths and directed either to the sample or to the reference mirror.
The light returning from each arm of the interferometer is recombined and directed into a spectrometer, which spatially separates the light to allow analysis of the interference pattern. The modulation of the cross-correlation spectrum contains distance information of the scattering structures within the tissue along a vertical (axial) axis. A Fourier transform of the measured spectra provides the OCT A-scan data. Multiple scans provide cross-sectional images (B-scans) similar to those from a sonogram, but at higher resolution.
The researchers used a spectral radar OCT system from Thorlabs HL AG of Lübeck, Germany, coupled to the camera port of a surgical microscope from Möller-Wedel GmbH, also of Germany (Figure 2). The OCT system uses a superluminescent diode with a central wavelength of 840 nm. It can image with an axial resolution of 14 μm (in air) at a depth of 3.6 mm and with a maximal sensitivity of >110 dB, while providing real-time information (1220 A scans per second).
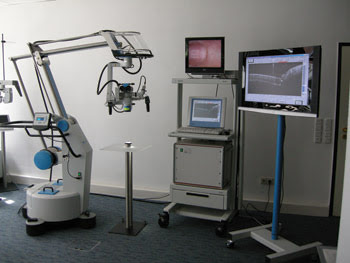
Figure 2. An optical coherence tomography (OCT) system was combined with a surgical microscope to aid in cochlear implantation surgery.
The surgical microscope system was modified for OCT to include unique antireflection coatings and compensation for the dispersion in the multielement zoom optics. The built-in zoom function of this microscope allows image magnification to vary by a factor of six, with a corresponding change in the scan depth (e.g., from 10 to 60 mm). The OCT imaging system adapts for changes of the working distance from 232 to 290 mm by including a motor control on the reference mirror. The researchers used this OCT system to visualize the membrane system that separates the scala vestibuli and scala tympani without actually opening the fluid-filled channels (Figure 3).
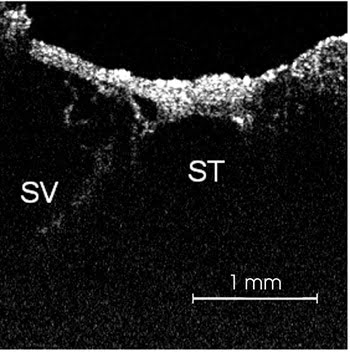
Figure 3. OCT was used to visualize the membrane system (Reissner’s and basilar membranes) that separates the scala vestibuli (SV) and scala tympani (ST) without actually opening the fluid-filled channels.
Implications for cochlear surgery
Until the advent of OCT, there was little information to guide the surgeon on the anatomical site of the scalae or on the lateral projection of the basilar membrane. In some cases, this led to inadvertent opening of both scalae and to local damage of the basilar membrane at the site. The frequency-domain OCT surgical microscope described here provides real-time feedback to the surgeon for precise anatomical location of the scalae and of the projection of the basilar membrane on each unique individual before opening the membrane of the cochlear wall. This allowed the surgeon to create an opening just large enough for the electrode.
This new surgical OCT system is undergoing clinical trials for cochlear implantation surgery and may prove beneficial for providing real-time surgical guidance during other delicate surgical procedures such as brain or ophthalmic surgery.
Meet the author
Alex Cable is president and CEO of Thorlabs Inc. in Newton, N.J.; e-mail: [email protected].
Lori Howe is in the marketing department at Thorlabs.
References
1. E. Lankenau et al (2007). Combining optical coherence tomography (OCT) with an operating microscope. Advances in Medical Engineering. T.M. Buzug et al, eds. Springer, Berlin, Heidelberg, pp. 343-348.
2. H.W. Pau et al (2007). Optical coherence tomography as an orientation guide in cochlear implant surgery. ACTA OTOLARYNGOL, Vol. 127, pp. 907-913.