Novel microscopy techniques help biologists dig deep.
Hank Hogan, Contributing Editor
A look at some of the latest work in multiphoton microscopy shows that advances in the technique are enabling new images and research, including uncovering the workings of muscle and the growth of bone. Other advances in technology show that, although you can’t be in two places at once, you almost can pull off the feat through multifocal multiphoton microscopy.
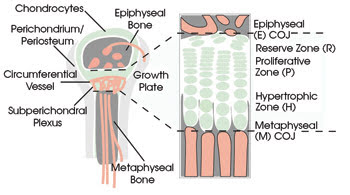
This diagram of a growing bone shows the three major vascular sources to the avascular growth cartilage. Chondrocytes align in axial rows and undergo a proliferation phase followed by a hypertrophy (swelling) phase. Their secreted matrix acts as the scaffolding for bone formation at the metaphyseal end. Note that the proliferative-to-hypertrophic transition occurs roughly at the axial center of the growth plate. Courtesy of Rebecca M. Williams, Cornell University.
Muscle microscopy
Imaging brawn while it is working takes brains. It also helps to have access to the latest multiphoton microscope technology, as evidenced in research conducted by scientists from the University of Heidelberg in Germany. They used multifocal multiphoton microscopy to image very fast and highly localized doubly charged calcium ion bursts in muscle, showing the tool’s usefulness for investigating the calcium release underlying muscle contraction.
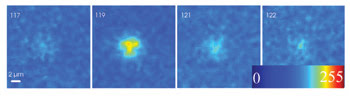
A time series of an elementary Ca2+ release event is imaged in two dimensions with a multifocal multiphoton imaging system operating at 60 Hz. Consecutive images are shown. In conventional confocal XYT imaging, the event would be present in one image but gone in successive images because conventional confocal imaging has an acquisition rate almost two orders of magnitude slower. Courtesy of Oliver Friedrich, University of Heidelberg.
“The elementary calcium release events we study in our lab reflect the biological ‘quantum’ of calcium release originating from discrete release sites,” said team member Oliver Friedrich, an assistant professor of medical biophysics. During recent years, study of these release events has brought insight into the excitation-contraction coupling in muscle.
Usually such research involves the introduction of a fast calcium dye into a tissue sample. When calcium is released, the resulting fluorescence from the dye is recorded, typically with a confocal microscope. Spontaneous elementary calcium-release events are random and rare, and they occur quickly, lasting between tens and hundreds of milliseconds.
The fast timescale makes it impossible to follow what is happening across a swath of muscle with a confocal scanning microscope. Some researchers have turned to line scans that repetitively survey a row of pixels, which provides good time resolution but only for one spatial dimension.
The Heidelberg researchers used a setup from LaVision BioTec of Göttingen, Germany, connected to an Olympus microscope. They used a Spectra-Physics Ti:sapphire femtosecond laser emitting at 830 nm as a light source. The incoming beam was split into 64 equally bright beamlets that were imaged as a linear array with 250-nm spacing between foci on the sample. The resulting two-photon images were achieved using optics and a CCD camera from PCO AG of Kelheim, Germany. Splitting the laser into beamlets allows a sixfold increase in the cell area that can be scanned, as compared with confocal point source microscopy.
With this setup, the researchers studied mouse muscles using the calcium dye Fluo-4. They collected data in 320 × 240-pixel images, acquiring stacks of such images taken at different times. “Image rates from several hundred hertz up to 4000 Hz are possible, depending on the image pixel resolution and foci number,” Friedrich said in describing the system’s specifications.
He added, “However, camera speed still limits acquisition rates to the lower-hundred-hertz range. There is still room for improvement.”
The scientists used wavelet processing to remove noise from the data and identified calcium events with an automated detection algorithm. With this information, they developed an event model for the calcium release.
Friedrich explained that these initial studies validated the technique, which offers several advantages over a confocal scanner. In addition to increasing the cell area imaged, it achieved a time resolution of 15 ms for X-Y images with the camera speed used, two orders of magnitude faster than with a confocal system. The possibility exists for the time resolution to be even greater if the CCD camera can be switched out for a faster CMOS version. In addition, the researchers found no sign of photodamage using the multifocal method, even though as many as 500 images were collected, an outcome arising from the use of multiphoton microscopy. Altogether, these attributes should allow detection of spontaneous events that previously would have been undiscovered. The work is detailed in the July issue of IEEE Transactions on Medical Imaging.
Thus, the technique could provide useful calcium imaging in a variety of cell-signaling settings. It also could be used for imaging other research subjects, such as red blood cells, Friedrich said. “We also applied this technique with the same system to visualize malaria parasites within plasmodium-infected erythrocytes.”
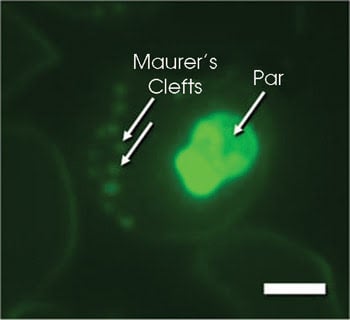
This multifocal multiphoton image shows a plasmodium-infected erythrocyte stained with LysoSensor Green. This dye stains parasitic membranes within the parasite (Par) as well as membrane folds (Maurer’s clefts) that link the parasitic protein export and nutrition pathways to the host membrane. The dye is excited with the 840-nm line of a femtosecond laser. Scale bar: 1.5 μm. Courtesy of Petra Rohrbach, Hygiene Institute, University of Heidelberg.
Watching bone grow
Growing bones, it seems, know a trick or two, according to studies conducted at Cornell University in Ithaca, N.Y. Researchers there used multiphoton microscopy to image the tissue within the bone growth plate of living animals.
They used multiphoton microscopy, research scientist Rebecca M. Williams said, because it can image relatively deeply within tissue. In a living animal, bone growth arises from cartilaginous discs at both ends of the bone. Because cartilage is generally free of blood vessels, all nutrients, oxygen, signaling molecules and waste must be transported through the tissue over relatively long distances for the tissue to survive and function.
Although cartilage is avascular, the surrounding connective tissue, the perichondrium, is not. Both it and the neighboring bone are highly vascularized, and for in vivo studies, both must remain intact so that transport patterns can be analyzed. Because the perichondrium is a 50- to 100-μm-thick collagenous capsule, imaging into the growth plate requires multiphoton microscopy.
For in vivo studies, the researchers injected anesthetized mice with fluorescent tracers and monitored them as they moved through the vasculature, into the perichondrium and then into the cartilage of the mice.
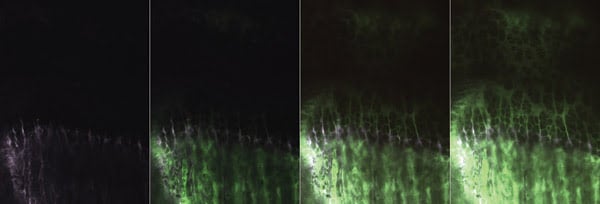
This series of multiphoton images shows the tracer (green, fluorescein) entry to the growth cartilage at 3-min intervals. Initially, the bone front is labeled with tetracycline (violet). Fluorescein enters primarily from the bone vasculature at the two ends and remains within the extracellular space. The entry pattern is used to determine spatially heterogeneous transport coefficients for the region. Images are 440 × 600 μm in scale. Courtesy of Rebecca M. Williams, Cornell University.
Multiphoton techniques also offered advantages when it came to ex vivo imaging, particularly with regard to photobleaching studies. The scientists looked at bone growth ex vivo by performing photobleaching recovery studies. They extinguished fluorophores in a region and tracked how long it took for fluorescence to recover. Multiphoton methods allowed them to carry out photobleaching in a three-dimensionally defined region. They thereby could measure diffusion coefficients in thicker tissue to determine how these varied by location within that tissue.
For the studies, the researchers used a custom-built system based on a BioRad laser scanner. Williams noted that the system had some imaging limitations but that it also offered some advantages. For one thing, it had space around the microscope stage for animal-monitoring equipment. The system also was flexible and allowed the researchers to implement beam modulation synchronized with the scanner so that they could blank the beam during laser flyback or during user-controlled photobleaching studies.
They used a Spectra-Physics Ti:sapphire laser tuned to 880 nm because tissue and water are relatively transparent at this wavelength. At the same time, their chosen fluorescent traces, fluorescein alone and attached to molecules of various weights, absorbed well at this wavelength. They used emission filters designed to reject the excitation light and standard Hamamatsu photomultiplier tubes as detectors.
As detailed in the Aug. 1 issue of Biophysical Journal, the researchers found a relatively permissive region at the midplane of the growth plate, a place where chondrocytes transition to early hypertrophic stages. It is also a region where communication occurs between chondrocytes and cells in the surrounding perichondrium. They found that transport in the living mouse was affected significantly by fluid flow resulting from a pressure difference between the bone vasculature and the cartilage.
“We were surprised at how great a role convection plays in solute transport within this region. The dogma is that cartilage, being avascular, is relatively sluggish. We initially tried to analyze our results using only diffusion and found that the analysis just didn’t work,” Williams said.
Studies continue in this area, and the group may have new results as a consequence of new technology. “We are currently developing a custom scanning system with the ability for synchronizing the image acquisition and beam modulation to a variety of mouse physiological parameters.”
Researchers from the Colorado School of Mines in Golden have extended two-photon microscopy into a new plane. They demonstrated a technique that uses time to multiplex multiphoton signals, enabling the imaging of two or more focal planes at once.
Multiple focal planes
Research leader Jeff A. Squier, a physics professor, noted that two-photon microscopy produces a sharp cross-sectional image. Biology, though, is inherently three-dimensional, and the new technique can extend multiphoton microscopy into that third dimension. What’s more, Squier said, the technique isn’t restricted to just two-photon microscopy. “It would also work with other nonlinear imaging methods such as second-harmonic-generation imaging, third-harmonic-generation imaging and so on.”

This second-harmonic-generation image shows a single dystrophic muscle cell. The arrangement of myosin, or motor protein, forms repetitive signal patterns in the sarcomeres of the cell. In Duchenne muscular dystrophy, abnormal cell morphologies such as fiber branching can be studied on the subcellular level using multiphoton microscopy. Courtesy of Oliver Friedrich, University of Heidelberg.
In a demonstration, the researchers used a homebuilt two-photon absorption fluorescence scanning microscope. This consisted of a homebuilt Ti:sapphire laser generating an 800-nm wavelength and 70-fs pulses with a repetition rate of 23 MHz. They split this beam into two pulse trains, one of which traveled through the unaltered optics while the second went through a path with a deformable mirror and focused at a depth different from that of the first. The maximum displacement between the two planes depended on the optics used. For example, the investigators achieved a maximum displacement of 110 μm in one case and 30 μm in another.
The fluorescence signal appeared within a few nanoseconds of the arrival of the exciting photons and then died away. It was this rapid response and subsequent behavior that made the scheme work because the fluorescence generated from one pulse appeared and disappeared before the next pulse arrived.
The researchers detected the two-photon signal with a Hamamatsu photomultiplier tube. The incoming laser pulse train acted as a master clock, enabling them to alternately connect one of two photon counters to the detector. In this way, they collected data from one focal plane and then the other, switching the electronic connection with every pulse.
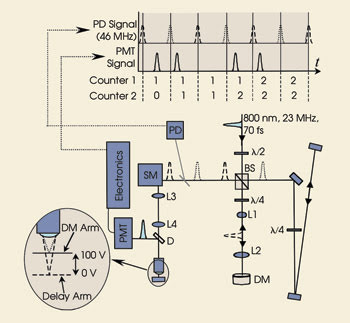
Incoming laser pulses (800 nm, 70 fs, 21.5 MHz) are split into two pulse trains. One strikes a deformable mirror and so focuses at a different depth in the sample than the other. Using fast electronics to switch the output of a photomultiplier tube from one counter to another enables multiphoton imaging at two focal planes. Courtesy of Jeff A. Squier, Colorado School of Mines and the Center for Microintegrated Optics for Advanced Bioimaging and Control. The top inset shows the relative time of the master clock and PMT signal. (BS = polarizing cube beamsplitter, L1–L4 = lenses (L1 = 100 mm, L2 = 200 mm, L3-L4 = 100 mm), DM = deformable mirror, PD = photodiode, SM = scanning mirrors, D = dichroic mirror). Reprinted with permission of Optics Letters.
The nonlinear microscope was relatively conventional, Squier noted. As for the electronics, he said that they weren’t costly. “The heart of the counting circuit is a field-programmable gate array — a really inexpensive flexible electronic system.”
In a test of the technique, the scientists set the deformable mirror to create a second focal point 9 μm below the first and imaged 15-μm carboxyl microspheres mixed with fluorescence Lucifer yellow dye, suspending the spheres in agarose. By focusing one arm at the top of the microspheres, the other was focused at their midpoint.
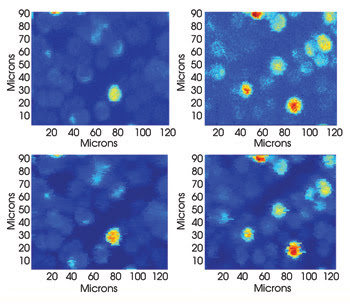
In the top row is a traditional two-photon fluorescence absorption image of labeled carboxyl beads. One arm of the system at a time is blocked, and the bead image is recorded. The top images demonstrate that the arms are focused to separate levels. Those in the lower row were taken with both arms unblocked. By using fast-switching electronics, the images from the two depths are recorded simultaneously. The images from each row appear very similar. Courtesy of Jeff A. Squier, Colorado School of Mines.
For evaluation, they blocked one arm and imaged with the other. Then they switched the arms so that they had one set of images from the top of the microspheres and the other from near their equator. Finally, they used their setup to image with both arms simultaneously. When compared, the sets of images agreed very well, with the researchers noting that the use of two arms at the same time produced images with less noise than those taken with the arms individually. The work is detailed in the June 15 issue of Optics Letters.
Squier noted that, with the technique proven with controlled specimens, the next step will be to image living systems. His group is collaborating with another group on this research. The ultimate goal, he added, is to push the limits of high-speed high-resolution three-dimensional multiphoton imaging in neuronal and other scattering tissue. Although that is some time in the future, other improvements are already under way.
“While we are presently at two focal planes, we are working to extend this to five. That will be the next improvement in the system.”