
Improving diagnosis of autoimmune diseases
Beads encoded with nanocrystals improve sensitivity of flow cytometry
Gary Boas
The indirect immunofluorescence assay is the gold standard for detecting the circulating antibodies that are implicated in most autoimmune diseases. The assays can be time-consuming, however, and because interpretation of the results is subjective, standardization of the method has proved difficult. Researchers have therefore sought to develop alternative techniques that are rapid, objective and automatable.
Microbead-based flow cytometry methods that use organic dyes as fluorescent labels meet these criteria. However, performing bead-based assays on large numbers of molecules requires encoding of each of the microbeads, and only a limited number of unique codes are available on polymeric beads encoded with organic dyes.
Using semiconductor nanocrystals rather than organic dyes as labels offers several advantages, including greater multiplexing capabilities as well as higher-photostability and higher-sensitivity antigen detection. Studies have demonstrated the potential of nanocrystal-encoded microbeads for genomic applications, specifically for multiplexed detection of oligonucleotide probes hybridization.
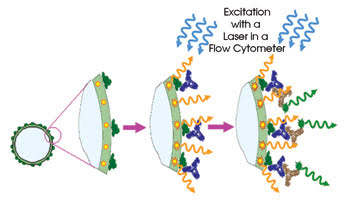
Investigators have reported a flow cytometry technique that uses nanocrystal-encoded microbeads for diagnosis of autoimmune diseases. The microbeads are optically encoded with orange-emitting nanocrystals and coated with human DNA fragments, and then with incubated serum samples from patients with the diseases. Excitation with a laserinduces FRET from the nanocrystals, which, in turn, causes a fluorescence emission. This emission enables detection of the antigen interaction with the surface of the microbead. Images reprinted with permission of Nano Letters.
In an August Nano Letters paper, investigators from Université de Reims Champagne-Ardenne in France and from Ludwig Maximilians Universität in Munich, Germany, reported a study in which they applied nanocrystal-encoded microbeads to proteomics and, in particular, to antibody profiling and clinical diagnosis of autoimmune disease.
“We believe our strategy represents a significant conceptual advance,” said Igor Nabiev, principal investigator of the study, “and presents a compelling case for how the nanoaspects are important in the success of new diagnostics.” He added that the study may be the first in which detection of an antigen interaction with the surface of a nanocrystal-encoded bead was achieved based on the emission of a labeled secondary antibody excited through Förster resonance energy transfer (FRET) from the nanocrystals. “In this case, the antibody-bead interaction turns the fluorescence signal off and on, avoiding background signal from noninteracting labels, thus increasing the antibody detection sensitivity of nanocrystal-encoded beads in immunodiagnostics and proteomics.”
To demonstrate the technique, the researchers optically encoded latex microbeads with nanocrystals of different colors and coated them with 68-kDa recombinant fragments of human DNA topoisomerase I, nRNP or Sm-antigen. DNA topoisomerase I has been shown to aid in clinical immunodiagnostics of anti-Scl-70 antibodies, which serve as markers of systemic sclerosis.
They observed unwanted aggregation of the nanocrystals while encoding the microbeads, but they overcame this problem by developing a layer-by-layer deposition technique that produces fluorescent microbeads homogeneous enough in size to be used with flow cytometry immunoassays.
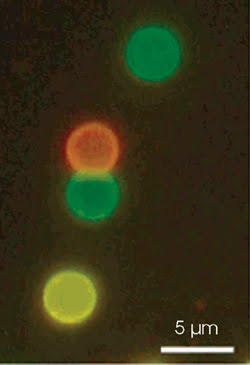
Nanocrystal-encoded microbeads enable greater multiplexing capabilities with respect to flow cytometry methods that use organic dyes as labels as well as higher-photostability and higher-sensitivity antigen detection. The fluorescence micrograph above shows microbeads encoded with nanocrystals of varying colors.
They incubated the microbeads with anti-topoisomerase I-specific monoclonal antibodies or with serum samples from patients with autoimmune disease. Then they performed flow cytometry assays using a BD Biosciences FACS STAR system, with the 488-nm line of an argon-ion laser providing excitation. The appearance of a fluorescence shift from the orange to the green enabled them to identify serum-positive patients. And the results correlated well with those from indirect immunofluorescence and ELISA techniques, confirming the efficacy of the method.
They further explored the sensitivity of the technique and sought to determine its detection limit by performing experiments with single microbeads. They used a confocal laser scanning microscope based on an inverted microscope made by Zeiss with a 1.45-NA 100× oil-immersion objective. A laser operating at 532 nm provided excitation; alternatively, they produced a broad excitation band from 450 to 500 nm using a fluorescence excitation lamp and a band filter set.
Fluorescence was collected by the same objective and sent to an avalanche photodiode or to a spectrograph for detection. The experiments confirmed that nanocrystals on a presurface layer of the microbeads serve as FRET donors and excite specifically bound labeled antibodies.
The study opens the door to nanocrystal-based clinical assays with previously unavailable sensitivity, Nabiev said. The extremely narrow and symmetric signals from the beads enabled measurements without any fluorescence compensation — a parameter that determines the limit of sensitivity of the assays when using organic dyes. Also, the scientists showed that they could detect fewer than 30 dye-labeled antibodies interacting with the autoantibodies at the surface of the bead, based on FRET.
Planar surface arrays are the medium of choice for diagnosis of autoimmune diseases, but these are limited by the means by which they bind autoantigens, and they suffer from drying, which can distort or otherwise interfere with those areas of the antigen surface that can elicit an immune response. Nabiev noted that liquid phase microbead-based assays using minimally disruptive techniques for labeling antigens are not hampered by those limitations.
Published: September 2007