
Optical trap pulls apart cell division mystery
David L. Shenkenberg
Cell division must happen during the life of every organism, but if unregulated, it causes cancer. However, the protein mechanisms behind mitosis are not fully understood. To further explore cell division, researchers at the University of Washington in Seattle have employed optical tweezers and a piezo microscope stage.
In particular, they studied whether tension directly or indirectly affects microtubules, protein strands that organize and subsequently pull apart chromosomes during mitosis through growth and shortening processes. It is believed that tension controls microtubule growth and shortening, but the mechanism of microtubule control remains unknown. “This is one of the great mysteries of cell biology,” said principal investigator Charles L. Asbury.
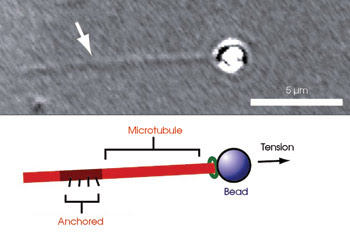
Researchers investigated microtubule growth and shortening in response to tension by attaching microtubules to a bead and pulling the bead with an optical trap.
The scientists viewed the microtubules with a Nikon inverted microscope. On a microscope slide, they placed microtubules and beads attached to a protein complex from yeast, which binds to microtubules. They pulled the beads with forces ≤2 pN using the optical trap, a custom-built device that employed a Spectra-Physics infrared laser. Because the beads were joined to the complex connected to the microtubules, pulling the beads pulled the microtubules. The opposite ends of the microtubules were anchored to the microscope slide. To keep the tensile force constant, the Physik Instrumente microscope stage on which the slide rested moved to accommodate changes in microtubule length.
To orchestrate stage movement, the investigators employed a custom position-detection system and custom software written in National Instruments’ LabView. The system continuously and automatically monitored the position of the bead relative to the trap center with nanometer precision. The researchers had to interfere only if the bead detached from the microtubule. They measured changes in microtubule length using the stage position as a reference.
They discovered that microtubules quickly respond to rapid changes in tension. Increasing tension decreased the rate and likelihood of microtubule shortening and increased the likelihood of its regrowth, as reported in the July issue of Nature Cell Biology. The investigators compared these results with previous in vivo and microtubule compression studies and said that they believe an analogous mechanism helps govern microtubule behavior in vivo and that the mechanism promoting microtubule elongation under tension is likely to be different from that which slows growth under compression.
Asbury said that the group plans to develop and apply tools to further study microtubule interactions with the yeast protein complex and other components of kinetochores, specialized structures that link chromosomes to microtubules during cell division.
Published: September 2007