By Gary Boas, News Editor
Researchers have made tremendous progress in recent years in developing optical imaging for the study and management of disease. By taking advantage of the near-infrared window — the wavelength range where scattering and absorption of light are optimal for such purposes — they have shown that they can track disease progression, monitor the behavior of drugs, identify biomarkers specific to disease and treatment outcomes, and more — all in vivo, noninvasively. Optical imaging is especially of interest, with respect to other imaging modalities, because it is relatively inexpensive, easy to use and does not require ionizing radiation.
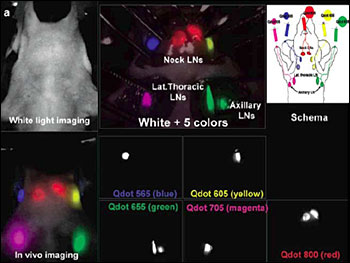
Researchers are probing the possibilities of using quantum dots (QDs) in the management of cancer. One group has reported multiplexed in vivo imaging with five different quantum dots, showing five distinct lymphatic drainages. This could enable researchers to predict the route of cancer metastasis into the lymph nodes (LNs). (Reprinted with permission of Nano Letters)
Concurrent with these advances, the labels employed with optical imaging have grown ever more sophisticated. Quantum dots offer a range of advantages over the organic dyes traditionally used for optical contrast. Because of a high quantum yield, a large absorption cross section and significant saturation intensity, they are up to 20 times brighter than organic fluorophores, enabling greater penetration into tissue in the near-infrared range. They also feature a high extinction coefficient and a marked resistance to photobleaching.
At the same time, quantum dots can incorporate a number of components in a single nanoprobe, facilitating multimodal imaging with optical imaging and computed tomography or MRI. This could enable simultaneous acquisition of functional and anatomical information, for example, allowing localization of the imaging target. For these reasons, researchers are eyeing quantum dots for an array of biomedical applications, including deep tissue imaging — at millimeter depths — which could have important implications in the diagnosis and management of cancer.
"Deep tissue imaging can provide more information regarding the pathological status of the disease, which makes the treatment more effective and efficient," said D. Sakthi Kumar, an investigator in the Bio Nano Electronics Research Center at Toyo University in Kawagoe, Saitama, Japan.
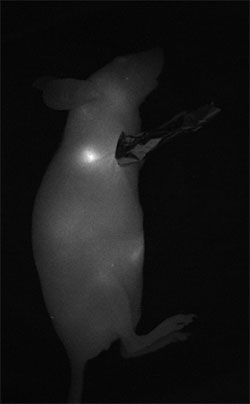
Sentinel lymph node mapping is one of the more promising clinical applications of quantum dots. In this image, the regional lymph node of a mouse is visualized a few minutes after subcutaneous injection of quantum dots in the right anterior paw. (Courtesy of Thomas Pons, ESPCI)
In a recent review of deep tissue imaging with near-infrared quantum dots, Kumar and colleagues noted specifically that quantum dots could contribute to visualization of molecular aspects of tumors such as vascular endothelial growth receptors, which serve as molecular markers for angiogenesis, epithelial growth factor receptors in breast tumors and sentinel lymph node mapping prior to surgery.
Thomas Pons, a researcher at ESPCI (the City of Paris Industrial Physics and Chemistry Higher Educational Institution), points to sentinel lymph node imaging as one of the more readily achievable clinical applications of quantum dots, "because QDs injected subcutaneously accumulate very easily in the regional lymph node."
Several groups are working toward this goal and have reported noninvasive, real-time sentinel lymph nodes in animal models. One team has described multiplexed in vivo imaging with five different quantum dots, visualizing five different lymphatic basins. In theory, clinicians could use this approach to predict the route of cancer metastasis into the lymph nodes.
Researchers also are exploring tumor imaging with targeted near-infrared quantum dots. Achieving high detection sensitivity could prove more challenging, Pons said, because it will require realizing a robust quantum dot surface chemistry that can evade the immunological defense system — for prolonged circulation in the bloodstream — and that features a sufficiently high specificity for cancer cells versus normal tissue.
"Visualizing micrometastases with QDs will require a better understanding of the interactions between the inorganic QD and its surrounding organic ligands, and also the development of highly specific targeting moieties," he said.
Minimizing toxicity Several additional challenges must be addressed in developing quantum dots generally, including those related to size and an intermittent blinking. Although quantum dots are only nanometers in size, Kumar said, conjugation with biomolecules can increase the diameter considerably, making delivery into the cells more difficult. Researchers are working to solve this problem, he added, by synthesizing ultrasmall quantum dots. Recently, for instance, investigators have described strain-tuned quantum dots that can be made to emit light in the near-infrared even while maintaining the necessary size.
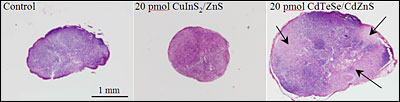
Researchers are developing quantum dots using less toxic materials, a necessary step if quantum dots are to be used for clinical purposes. Shown are the right axillary lymph nodes of mice seven days after injection of the same quantity of the more toxic CdTeSe/CdZnS quantum dots (right) and recently described CuInS2/ZnS quantum dots (left). The black arrows indicate inflammation sites resulting from toxicity. (Reprinted with permission of ACS Nano)
The reasons for the intermittent blinking are not yet fully understood, but some believe it is a result of Auger ionization. One way researchers have sought to overcome this, Kumar said, is by growing a thick shell around the core of the quantum dot, thereby suppressing the nanocrystal ionization. They have not yet fully overcome the blinking problem, however. Further development is needed if quantum dots are to be used for real-time imaging within the cell.
The major challenge, however, lies in addressing the cytotoxicity of quantum dots — and specifically of the compounds from which they are made, including CdTe, HgTe and PbS. For example, accumulation of CdTe-based quantum dots in the sentinel lymph node can lead to a strong local inflammation resulting from the release of toxic heavy metals. Quantum dots also tend to accumulate in the liver and spleen with prolonged retention before elimination. If they are to be used for clinical applications, therefore, researchers must develop new ones made of less toxic materials.
In a recent issue of ACS Nano (Vol. 4, No. 5), Pons and colleagues reported the synthesis of highly luminescent near-infrared CuInS2/ZnS quantum dots and their application for in vivo imaging in mice.
"We have demonstrated that the inflammatory response elicited by the presence of these materials was reduced by a factor of about 50 compared to CdTe-based QDs," he said. "This reflects the much lower toxicity of these materials and allows imaging using QD doses far below the toxicity threshold."
The researchers now are focusing on aspects of quantum dot synthesis and functionalization. For instance, they are developing brighter quantum dots based on CuInSe2 quantum dots that provide emission in the near-infrared range up to 1 µm (instead of 800 to 850 nm for CuInS2 quantum dots), and are researching new surface chemistries that are more stable and show less nonspecific adhesion to improve their targeting specificity. In addition, they are developing multimodal probes for simultaneous fluorescence/PET or fluorescence/MRI imaging.
[email protected]