
OCT Branches Out
For optical coherence tomography, it’s time to branch out. Improvements in imaging,
the use of a wider array of wavelengths, and advances in contrast agents promise
to make OCT a tool that can be used to diagnose and treat disease – as well
as to make better consumer goods. However, issues related to cost, speed and new
capabilities present challenges.
OCT is an interferometric technique, with the interaction between
a sample and reference beam yielding images at wavelength- and tissue-dependent
depths. It is used, among other things, for imaging structures in the eye.
It also makes sense to apply it to structures on top of the eye,
specifically contact lenses, said University of Arizona biomedical engineering professor
Jennifer K. Barton. After all, OCT is a good tool for measuring transparent and
semiturbid material, such as a contact lens. What’s more, there is a need
for nondestructive quality assurance, something difficult to do.
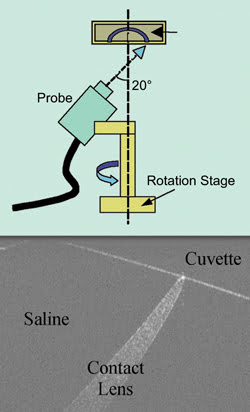
With an OCT setup (top), contact lens thickness in a saline solution (bottom) can be
measured, a quality assurance task currently done manually. Courtesy of Bryan R.
Davidson and Jennifer K. Barton, University of Arizona.
“It is actually a big problem, particularly at the edges,
and particularly with soft lenses, which need to be measured in saline,” Barton
said. “OCT is the only imaging technique we know of that can measure the three-dimensional
structure of a transparent lens in saline in near-real time.”
Current quality assurance involves manually slicing contact lenses
and measuring that slice at two points using a microscope. In trained hands, the
technique works, but it is slow, destructive and limited to specific locations.
In research described in a January/February 2010 Journal of Biomedical
Optics paper, graduate student Bryan R. Davidson and Barton used OCT to measure
contact lenses’ prism, the difference between the thinnest and thickest part
of the lens at a specified distance from the edge. For a spherical lens, this parameter
should be zero. In an astigmatism-correcting toric lens, the prism should be a specific
number. Any deviation from the expected indicates a potential manufacturing problem.
The two researchers from the Tucson, Ariz.-based institution showed
that repeatability of the thickness measurement was 2 μm, even though the axial
resolution of the OCT system was more than three times larger. These results were
made possible by taking advantage of the transparent nature of contact lenses and
the speckle in the image, Barton said.
This proof-of-concept demonstration took nearly 7 min to produce
a 36-point measurement report. However, with optimized processing and a faster acquisition
time, that might be cut by two orders of magnitude. This would be necessary to actually
implement the technique on an assembly line. Barton said that a manufacturer reportedly
has used something similar to the prototype setup done in a quality assurance lab.
As for the future, she said, the next step will be to take advantage
of OCT’s 3-D capabilities rather than collecting a series of 2-D images, as
was done in this demonstration. That will require much faster imaging and processing.
System cost also must come down, something that light source advances
might make possible. The fact that there is no need for FDA regulatory approval
also may help cut costs, which Barton believes is necessary.
“An OCT system needs to be competitively priced with other
machine vision systems to be widely adopted,” she said.
When it comes to medical applications, the OCT market is expanding
in other ways; e.g., the Toronto-based Millennium Research Group, a medical market
technology analyst firm, issued a report in September 2010 predicting a 60 percent
annual growth rate through 2015 in cardiology-related OCT in the US.
The same report noted that St. Jude Medical of St. Paul, Minn.,
is currently the only player in the US OCT cardiovascular catheter market. However,
others are expected to launch rival systems soon.
In a related development, there are research programs developing
OCT for blood flow measurements. One such effort – under way at the joint
MIT, Harvard Medical School and Massachusetts General Hospital Athinoula A. Martinos
Center for Biomedical Imaging in Charlestown, Mass. – uses a version of OCT
that determines blood flow by tracking signal changes generated by the velocity-dependent
Doppler effect.
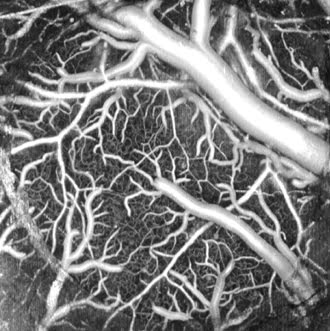
OCT can image blood flow, providing scientists with a new research
tool. Courtesy of Vivek J. Srinivasan, Massachusetts General Hospital.
The researchers’ goal is to create new tools for the study
of experimental stroke, Alzheimer’s disease, brain injury and related diseases.
Today, the gold standard for cerebral blood flow measurement is auto-radiography,
which requires animal sacrifice and histology. It is hoped that Doppler OCT can
take the place of such a procedure, which necessarily limits what research can be
done.
“Our new techniques of cortical cerebral blood flow measurement
using Doppler OCT will enable real-time, noninvasive measurements that can be repeated
in the same animal over time. This will enable more accurate tracking of disease
progression,” said Vivek J. Srinivasan, a radiology instructor and assistant
in biomedical engineering at Harvard Medical School and Massachusetts General Hospital,
respectively.
Srinivasan noted that the investigators reported on their progress
in January 2010 in Optics Letters and Optics Express. Their results showed good
agreement with previously published autoradiography values. The scan time generally
ran between 10 s and 1 min.
One key to pulling this off was the development of sophisticated
algorithms that compensate for movement of the bulk tissue. Uncorrected, such movement
leads to artifacts that obscure the signal from flowing blood. Another important
advance was the development of methods that accurately quantified flow independent
of blood vessel orientation.
The researchers used a prototype system, developed in collaboration
with Thorlabs of Newton, N.J., that had a superluminescent diode source with a 1310-nm
center wavelength and an InGaAs detector. A commercial version could be available
soon, Srinivasan said.
What such a system and the accompanying algorithms could offer
is quantitative measurements of critical vascular parameters without the use of
a dye. Because imaging is rapid, it could be performed under dynamic conditions.
Additionally, because of the high penetration depth of OCT, imaging
could be performed through thinned skulls in rats and mice. This would avoid invasive
procedures that may cause brain inflammation and alter results.
Ultimately, what the researchers are looking for is what happens
after a stroke, Srinivasan said. “We are particularly interested in imaging
of the stroke penumbra and how the processes of neurogenesis and angiogenesis contribute
to recovery and repair days to weeks after stroke.”
Besides what is going on at universities, companies also are working
to expand OCT capabilities, often through technology innovations. One example comes
from Sensors Unlimited – Goodrich ISR Systems Div. of Princeton, N.J., and
another from Xenics of Louvain, Belgium. Both recently announced cameras that operate
in the short-wave IR, which runs from 1 µm up to approximately 1700 nm. These longer
wavelengths allow greater tissue penetration than is the case at 840 nm, an OCT
standard.
In the case of Sensors Unlimited, its 1024-pixel line-scan InGaAs
camera is being used for OCT imaging at 1050 nm. Of critical importance to the performance
of such systems is the almost-92-kHz speed of the camera, said Douglas S. Malchow,
the company’s business development manager for industrial applications.
Faster image acquisition helps reduce the impact of patient movement,
he said. There also is an added advantage, however. Because OCT works through interference
between a reference and sample beam, the clarity of the image is degraded by air
turbulence or mechanical changes in the optical path. “Another benefit of
the fast speed is to prevent those drifts from blurring the image detail,”
Malchow said.
Raf Vandersmissen, CEO of sInfraRed, Xenics’ Singapore-based
subsidiary, noted that the company’s new line-scan camera runs at speeds of
up to 40 kHz. It offers the ability to switch between a high-sensitivity and high-dynamic-range
mode. This can be important in OCT because various tissues have various scattering
characteristics, placing demands on the detector.
“You need to have a flexible camera because you have to
adapt to different situations,” Vandersmissen said.
Researchers also are engaged in expanding OCT capabilities, with
technology innovations on the macro- and microscopic levels. A case in point comes
from University of Illinois at Urbana-Champaign bioengineering professor Stephen
A. Boppart, who is part of a group developing targeted magnetic nanoprobes that
could be similar to iron oxide ones in the regulatory approval pipeline for use
as MRI contrast agents.

Diagnostics and therapy with one platform, using OCT and magnetic
nanoparticles (lower left) driven into motion by a magnetic field (two images, bottom
right). The accompanying OCT signal (top left) can image diseased tissue and the
response to magnetomotive nanoparticle therapy. Courtesy of Stephen Boppart and
the Beckman Institute Visualization Laboratory, University of Illinois at Urbana-Champaign.
The nanoprobes under development by Boppart and his co-workers
move minutely in response to oscillating magnetic fields and can be imaged using
OCT. As a result, they can probe the elasticity, viscosity and other mechanical
parameters of tissue, providing a way to image these for such applications as cancer
detection, tissue biomechanics research and, possibly, therapy.
The Illinois group published some results in the May 4, 2010,
issue of Proceedings of the National Academy of Sciences, showing that it is possible
to pick up tumor signals with OCT using nanoprobes targeting cancer cells. Future
innovations could include better ways to deliver the magnetic field where needed,
either on coils external to the body or perhaps in miniaturized versions that fit
on a catheter. The hope is for a platform that can be used to spot disease, deliver
treatment and determine response to therapy, all through a combination of imaging
and magnetic field-induced action.
As Boppart said, “We’ve got targeted agents. We can
detect these with magnetomotive OCT and MRI. The next phase is theranostic agents,
to look at these magnetic particles as both diagnostic and therapeutic.”
/Buyers_Guide/Xenics/c16191