
Property Management: A Review of Optical Materials
Optical materials are as old as the natural quartz and obsidian crystals early civilizations used
to change the way people saw the world. Artificial materials came later, with mirrors
and other optics produced following the growth of art glass in Egypt, India and
China. Today, the prospects for choosing the perfect material to make lenses for
endoscopes and cameras, beam shapers for laser systems, or segments of a giant telescope
mirror are wide-ranging and often daunting, even for seasoned pros.
In general, optical materials can be broken down into categories,
such as transmissivity vs. reflective or glass vs. ceramic vs. metal. Nonetheless,
they are typically discussed only as the sum of their properties, or in terms of
whether they exhibit the qualities necessary for the task at hand.
Reflective optics need less specialized materials because, by
nature, light should not be passing through them. Instead, attributes such as ease
of polishing, the ability to accept a coating and durability under heat count the
most. Refractive index, homogeneity and the number of bubbles or similar features
in the glass are most important to transmission optics.
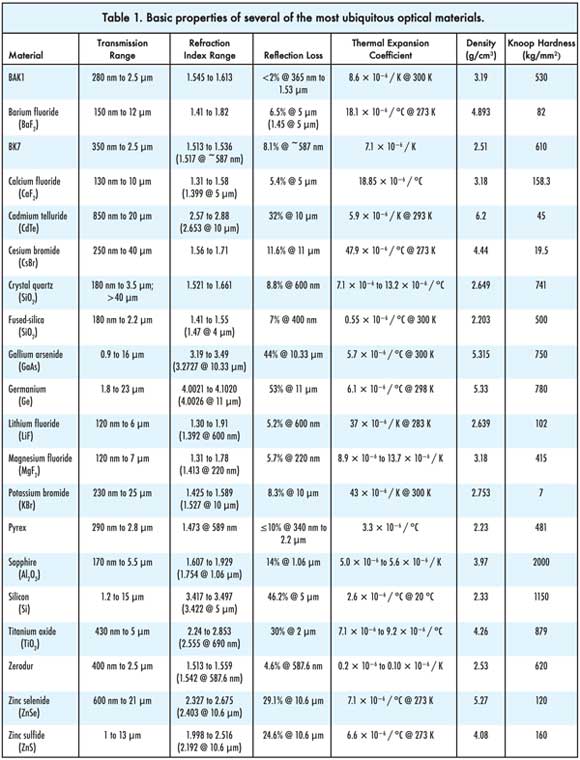
(For a larger view of this table, please click on this link)
Optical material properties
The innate (or engineered) properties of an optical material directly
affect the selection process. Several factors must be considered, but the most important
properties are:
• Index of refraction
• Dispersion
• Transmission value at any given wavelength
• Thermal expansion coefficient
• Density
• Hardness
• Elasticity
• Cost
The first three indicate how well a material will transmit, reflect
or otherwise manipulate a light beam. A material’s thermal resistance, density,
hardness and elasticity all relate to how well an optic will withstand the rigors
of the environment in which it is used. And of all of these, the cost will tell
you where else on your checklist you must make compromises in quality. Nearly everywhere,
demanding better specs for your optic will equal a higher price.
“Client education is very much an issue,” said Brandon
Light of Optimax Systems Inc. in Ontario, N.Y. “You want to move beyond the
refraction index” when discussing the specifications with your optics supplier.
To Light and other engineers, the material selection is the fundamental
consideration. The most important qualities of a material – beyond the refraction
index – are not easy to pin down.
“We work with whatever meets the design and application
needs, with particular attention given to relative cost and melt frequency,”
said Gregg Fales of Edmund Optics in Barrington, N.J. He and Light also cite macroeconomic
factors – such as the availability of a material – as having at least
a modicum of importance.
Various glasses are getting more expensive because of the shrinking
availability of rare-earth materials imported from China, Light said. Overall, the
variety of glasses is dwindling. For example, New York-based Corning Inc. stopped
producing calcium fluoride glass, leaving only two factories making it – one
in Germany and one in Japan – to meet global demand.

Calcium fluoride is used for microlithography
applications because of its compatibility with high-energy lasers. Courtesy of Schott
AG.
Optical engineers consider specific requirements for each new
situation. Making the right match is only a matter of understanding those needs
well, knowing what is available and prioritizing one’s desired results.
The following is a rundown of the characteristics of some of the
most often used materials, from glass to metals and beyond:
BAK1: A common borosilicate glass used for lenses and other transmission
optics, BAK1 has moderate abilities and comes at a moderate cost. It is similar
to other borosilicates, such as BK7, but has a somewhat better transmission in the
near-ultraviolet range.
Barium fluoride (BaF2): Used chiefly for windows in spectroscopy
applications, BaF2 is not found in a native state, and so all of it must be synthesized,
making it relatively expensive to produce. It cleaves easily, is etchable and polishes
well, all features that aid processing. It is highly susceptible to thermal shock,
however.
BK7: Another common borosilicate, BK7 is a relatively hard material
with good scratch resistance. It can be formed into optics with high homogeneity;
thus, a low bubble or inclusion content. It also is easy to work with but not recommended
for temperature-sensitive applications, such as precision mirrors.

Synthetic quartz glass is highly scratch-resistant and birefringent in nature. Courtesy of Schott AG.
Cadmium telluride (CdTe): CdTe is used in IR transmission applications,
such as spectroscopy, up to 20 μm, as well as for IR filter substrates, low-power
CO2 lasers and some solar photovoltaic applications. It is highly toxic, however,
and thus expensive to manufacture and problematic for dismantling and recycling
systems that use it.
Calcium fluoride (CaF2): Featuring low absorption, CaF2 has a
high damage threshold that lends itself to use as excimer laser optics. With a high
thermal expansion coefficient and low index of refraction, it can be trusted to
remain stable even without antireflection coatings when used to make windows, lenses,
prisms and other optics.
Cesium bromide (CsBr): Mainly used in radiation-detection settings,
CsBr can be found in some optical applications, including beamsplitters and prisms
in broadband spectrophotometers, as well as for x-ray fluorescence screens and
absorption-cell windows.
Crystal quartz (SiO2): Featuring high transmittance in the infrared
to ultraviolet range, SiO2 is used primarily to make wave plates and polarizers
because of its birefringent nature. It also is hard and scratch-resistant.
Fused silica: Generally free of inclusions and bubbles, fused
silica offers a very low thermal expansion coefficient, high homogeneity and a high
energy damage threshold, making it very suitable for both transmission and reflection
optics. It is used in energetic laser applications, photolithography and light
pipes as well as standard optics such as prisms, mirrors and lenses. UV-grade fused
silica offers these benefits for deep-UV applications.

Suitable for both reflection and transmission optics, fused silica
has a very low thermal expansion coefficient and high damage threshold. Courtesy
of Schott AG.
Gallium arsenide (GaAs): In its basic form, GaAs is a semiconductor
useful for making LEDs and solar panels. Optical-grade GaAs is used in infrared
transmission. It is more expensive than germanium or zinc selenide, but it is hard
and durable with a low absorption coefficient.
Germanium (Ge): Used primarily in infrared applications, Ge is
a high-refractive-index material commonly used for attenuated total reflection prisms
for spectroscopy, as a beamsplitter that doesn’t require coatings, as a substrate
for optical filters and for lenses. It is best suited for low-power applications,
including thermal imaging.
Lithium fluoride (LiF): Used mainly for applications in the ultraviolet
range, LiF has the best transmittance in the UV of all available materials. It also
is used for x-ray monochromator plates, but it is very fragile and hard to process.
Magnesium fluoride (MgF2): As is LiF, magnesium fluoride is used
mostly for UV optics. It also is found in infrared applications and excimer laser
systems because of its resistance to thermal and mechanical shock and its high energy
damage threshold. The material sometimes is used in biological and military imaging
applications as well. Grown in vacuum and very durable, MgF2 is slightly birefringent
and easy to work to high tolerances.
Potassium bromide (KBr): It is most commonly used for infrared
applications, such as lenses, beamsplitters and windows for gas and liquid sample
cells in Fourier transform infrared spectrophotometers. A very soft material, KBr
also is water-soluble and susceptible to degradation from atmospheric moisture.
Pyrex: Another borosilicate, Pyrex has too low homogeneity and
too high bubble content and other inclusions to be considered an optical glass,
so it is more often used for lab equipment than for optics. Nevertheless, its low
thermal expansion coefficient and resistance to heat shock and high temperatures
in general means that some use the material for common nontransmission optical applications,
such as windows and mirrors, where toughness matters.
Rutile (TiO2): Typically used as a whitener for pigments, TiO2
also can be employed as a high-numerical-index (in the visible range) material that
can be used between metallic layers in an optical system, such as between two coupling
prisms or waveguides. Because of its high numerical aperture, it also is extensively
used as a durable thin-film coating for beamsplitters, mirrors and other optics.
Sapphire (Al2O3): Tough and durable, artificially grown sapphire
crystals are used extensively for optical windows and substrates in a wide variety
of wavelengths, primarily infrared and ultraviolet. Sapphire often is found in lightguides,
prisms, windows and other optics required in extreme environments.
Silicon (Si): Silicon is used for optical windows and filter substrates,
mostly in infrared applications. Its applications run a very wide gamut, however,
with optically relevant devices as small as nanowires to as large as 18-in. wafers.

Schott's glass ceramic Zerodur is used as the primary mirror in space-bound telescopes. Courtesy of Schott AG.
Zerodur: A glass ceramic made by Schott, Zerodur offers a thermal
expansion coefficient near zero (fused silica is next closest at 0.52). With a high
number of inclusions, however, it is best suited for mirror substrates rather than
transmission optics.
Zinc selenide (ZnSe): Ubiquitous in infrared imaging and biomedical
applications, as well as for prisms in attenuated total reflection spectroscopy
accessories, ZnSe has a high index of refraction and high thermal shock resistance.
It is fairly soft but offers low absorption, so it is best for high-power CO2 laser
applications at 10.6 μm.
Zinc sulfide (ZnS): Used for infrared imaging where rugged optics
are necessary, such as IR viewers with military applications and as windows in IR
spectrometers. The material can be treated in such a way as to be clear rather than
sulfur yellow. This permits use in visible wavelengths as well, but at the expense
of durability.
/Buyers_Guide/Optimax_Systems_Inc/c11021
Published: September 2011
Glossary
- fused silica
- Glass consisting of almost pure silicon dioxide (SiO2). Also called vitreous silica. Frequently used in optical fibers and windows.
- glass
- A noncrystalline, inorganic mixture of various metallic oxides fused by heating with glassifiers such as silica, or boric or phosphoric oxides. Common window or bottle glass is a mixture of soda, lime and sand, melted and cast, rolled or blown to shape. Most glasses are transparent in the visible spectrum and up to about 2.5 µm in the infrared, but some are opaque such as natural obsidian; these are, nevertheless, useful as mirror blanks. Traces of some elements such as cobalt, copper and...
- hardness
- In the most general sense, the resistance of a solid surface to damage.
- pyrex
- Trade name for a type of borosilicate glass manufactured by Corning Glass Works, noted for its low coefficient of thermal expansion. Used most often for mirror blanks; not suitable for light transmission.
- reflective
- The term reflective is an adjective that describes the ability of a surface or material to reflect light or other forms of radiation. It implies the capability of bouncing back or redirecting incident light waves. The reflective property is often quantified by the reflectivity or reflectance, which is the ratio of reflected light intensity to the incident light intensity.
Key points about the term reflective:
Surface property: When a surface is described as reflective, it means that the...
- si
- Systeme Internationale d'Unites, the international metric system of units.
- telescope
- An afocal optical device made up of lenses or mirrors, usually with a magnification greater than unity, that renders distant objects more distinct, by enlarging their images on the retina.
- transmissivity
- The internal transmittance per unit thickness of a nondiffusing material.
- zerodur
- Schott Glass Technologies' trade name for a glass-ceramic material with a very low coefficient of thermal expansion.
BaF2BAK1beam shapersBK7CaF2CdTeceramicCoatingsCsBrdefenseenergyFeaturesFiltersfused silicaGaAsGeglasshardnessindustrialKBrlensesLiFMaterialsmetalmirrorsOpticsPrismsPyrexreflectiveSispectroscopytelescopetransmissivityWafersZerodurZnSereflective optics