By Krista D. Zanolli, News Editor
Earlier this summer, Harvard University researchers reported the first successful biological laser based on a single living cell. Malte C. Gather and Seok Hyun Yun, whose backgrounds are in physics, not biology, said they understood that lasers play a crucial role in many applications in biomedical optics, but so far it had been inanimate lasers looking at biological matter.
“We were interested in fusing optics and biology in a nonconventional way,” Gather told Photonics.com. “Our first work in this arena was to show a laser made out of something that’s alive.”
To that end, Gather and Yun genetically engineered human embryonic kidney cells to produce green fluorescent proteins (GFP) – the substance that makes some marine animals, like jellyfish, bioluminescent. They then placed a single cell (measuring 15 µm in diameter) between two highly reflective distributed Bragg reflectors that were spaced 20 µm apart, which formed an optical resonator that was just big enough to fit a single cell inside. To lase, the GFP in the cell was pumped with pulses of blue (465 nm) light. The researchers found that within the tight confines of the optical resonator, the light bounced back and forth between mirrors, amplifying the emission from the GFP to a coherent green laser beam. Normally the cell would just fluoresce, but in this case, the cavity feedback caused it to lase. The living cell was unharmed. 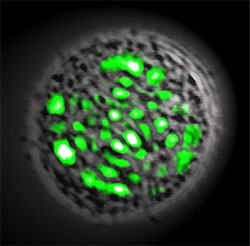
A single biological cell (shown in black and white) is genetically programmed to produce a light-emitting molecule, the so-called green fluorescent protein (GFP). When placed inside an optical resonator, this cell can generate green laser light. Due to the irregular internal structure of the cell, the laser spot has an apparently random pattern. (Images: Malte C. Gather, Seok Hyun Yun and Nature Photonics)
While the laser pulses lasted only a few nanoseconds, they were bright enough to be detected, and the researchers said they appeared to carry useful information for analyzing the properties of a large number of cells instantaneously. They said the ability to generate laser light from a biocompatible source placed inside the body could be useful for photodynamic therapies or novel forms of imaging.
“We are currently investigating whether the specific spectral and spatial output patterns of lasing cells can allow us to detect subtle differences between healthy and abnormal cells,” Yun told Photonics.com. “If successful, this can lead to a new modality for cytometry.”
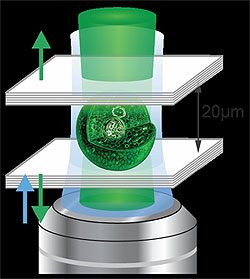
Schematic of a living laser. When a single biological cell genetically programmed to produce green fluorescent protein is placed inside an optical resonator consisting of two parallel mirrors separated by 20 µm (0.02 mm), the cell can generate green laser light.
“One of our long-term goals will be finding ways to bring optical communications and computing, currently done with inanimate electronic devices, into the realm of biotechnology. That could be particularly useful in projects requiring the interfacing of electronics with biological organisms,” Gather said in a press release announcing the work.
“GFP has revolutionized our understanding of cellular processes and contributed a major improvement to image resolution. The possibilities of photochromic proteins as biological optical data storage media have also been investigated,” said Steve Meech of the School of Chemistry at the University of East Anglia in Norwich, England in a ‘News + Views’ article in Nature Photonics in July. Meech was not involved in the work. “The work of Gather and Yun suggests that GFP may soon have a significant impact in the field of photonics. It is too early to say what will become of a biological self-generating and self-healing laser medium, but it certainly offers an exciting new range of possiblities for fluorescent proteins.”
Bacteria hops onboard
More recently the duo demonstrated that Escherichia coli (E.coli) bacteria cells are also capable of generating laser light.
E.coli are typically not harmful and, when genetically manipulated, have been used to produce human insulin, vaccinations and even biofuels.
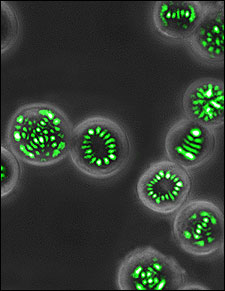
Microscope image of living lasers in action.
In a recent Optics Letters paper, Gather and Yun said that genetically engineering bacteria tends to be more robust and more efficient than for mammalian cells, adding that unlike in their previous work, the genetic transformation of the bacteria was stable – meaning that the capability to synthesize fluorescent protein is maintained during cell replication. They said that stable transformation allows for long-term use of biological lasers, for instance, by fully harnessing their self-healing nature – the ability to replenish the optical gain material in response to photobleaching of GFP.
“It is somewhat easier to trigger bacteria to produce GFP in large quantities and also to culture large quantities of bacteria. So if we were ever to scale up these biolasers to generate higher powers, bacteria would probably be the species to use,” Gather told Photonics.com.
The future of the living laser in biology
In an interview in the July issue of Nature Photonics, the researchers suggested possible medical applications where the laser could be amplified inside the body to reach deep tissue, but Gather told Photonics.com that, for a cell to “self-lase,” more work still needs to be done.
For this to work, “Two things need to happen: There needs to be an internal energy source,” Gather said. “For now the lasers receive their energy from an external light source. Bioluminescence would be the obvious way to get rid of the external light source and to use biological power to drive the laser. Secondly, the laser requires a cavity. At the moment, this is an artificial structure with macroscopic dimensions. In order for the cell laser to stand alone, this cavity would have to be made dramatically smaller. Nanolaser cavities could solve this problem. Imagine implanting tiny nano-objects into the cell to provide sufficient optical feedback.”
For now, external light is still needed to stimulate the biological laser, which currently limits the depth of tissue penetration.
When asked how this breakthrough will influence the future of biophotonics, Gather said, “Our hope is that our work inspires researchers in our field and related fields to think of novel ways of interfacing living matter and light. We have shown that a cell can be used to drive a laser. Who knows what else living matter can be used for in the realm of optics?”
Krista D. Zanolli