Dr. Yolanda Fintschenko, Labsmith Inc., and Dr. Blanca H. Lapizco-Encinas, Tennessee Technological University
High-quality video and images from an inverted fluorescence synchronized video microscope show promise for imaging micro- and nanofluidic bioanalyzer prototypes.
Researchers working with lab-on-a-chip and cell experiments to date have relied on general-use microscopes to view the flow of materials through microchannels and to image microparticles. These instruments typically are large and expensive and lack the features for viewing live cells and other particles. Their look-down design inhibits access for high-voltage electrodes, syringe pumps and other hardware necessary for micro- and nanofluidic research and development experimentation.
A modular, inverted fluorescence synchronized video microscope (Figure 1) has been developed for imaging micro- and nanofluidic bioanalyzer prototypes. The system combines bottom-up viewing and illumination with a motionless sample stage, enabling visualization of microsystems without perturbing the fluid flow. The motionless stage also acts as an optically accessible “lab bench,” with unhindered access for external electrical and fluid connections.

Figure 1. The inverted fluorescence synchronized video microscope. Inset image shows epifluorescence module imaging a microfluidic channel from below. Images courtesy of LabSmith Inc.
Microanalytical methods hold great promise for rapid microbial detection, separation and concentration. Researchers working with microanalytical methods require an imaging platform that allows them to verify – in real time – the presence of particles and cells passing through microchannels or capillaries. Traditional microscopes, in which the optics are housed in a stationary head above a moving stage, have several drawbacks for these experiments:
• The moving stage can easily perturb the microfluidic system.
• The microscope’s objective impedes access to the microfluidic chips, fluid circuits or cell plates and other
hardware.
• Standard illumination can quench the fluorescent dyes used to view the particles or microorganisms.
The inverted fluorescence synchronized video microscope was designed to provide high-quality images and video of micro- and nanosystems. The instrument’s small size allows it to be used on a desk or benchtop. The stage is immobile, leaving the experimental setup unperturbed by the imaging process. Images are displayed on a computer, so there is no need for overhead viewing optics.
Figure 2 shows an exploded view of the microscope’s major components. The optics are contained within the camera module. An LED ring illuminator module can provide external illumination at a variety of wavelengths for fluorescence and dark-field microscopy. Stroboscopic illumination reduces quenching of fluorescent dyes. The camera and illumination modules both attach to an X-Y stage that moves beneath a stationary stage top. This design lets researchers construct microsystems directly on the microscope and allows unimpeded connection to other equipment.

Figure 2. Exploded view of the microscope’s major components.
An epifluorescence camera module can provide greater wavelength selectivity for improved signal-to-noise. This module contains illumination and filters as well as the camera. Light returned from the sample follows a common optical path.
The microscope’s operating software also can record pictures or movies and can insert velocity probes for particle image velocimetry experiments.
Bioanalytical efficiency
An example of the efficacy of the microscope is in the research of Dr. Blanca H. Lapizco-Encinas, associate professor of chemical engineering and director of the Microscale Bioseparations Laboratory at Tennessee Technological University in Cookeville. Her team is studying an improved method for single-step separation and concentration of macromolecules, microorganisms and nonorganic particles.
The method, insulator-based dielectrophoresis (iDEP),1,2 is a variant of dielectrophoresis, a process in which nonuniform electric fields concentrate and separate particles.3 In the iDEP technique, two remote electrodes apply a direct current across a microchannel filled with a conductive electrolyte and an array of insulating structures. The structures do not conduct current and thus create the nonuniform electric fields necessary for DEP.
By varying experimental parameters and the shape, size, spacing and orientation of the insulator posts, particles and cells of specific sizes can be trapped, separated and concentrated in seconds or minutes, rather than the hours or days required by traditional concentration techniques. Because iDEP microdevices are inexpensive and relatively simple to fabricate, the method holds significant promise for miniaturized high-throughput sampling systems. The electric field gradient is easily controlled across the entire volume, enabling scalability.
For the iDEP method to be widely implemented, it is necessary to understand the conditions that result in the best trapping of various-size particles and organisms. To that end, Lapizco-Encinas and her team developed extensive, predictive models encompassing a range of experimental conditions. To corroborate the models, a set of iDEP experiments was conducted, in which a microchannel was fabricated containing an array of cylindrical, insulating posts, 200 µm in diameter and spaced on 250-µm centers (Figure 3). A DC field is applied both to trap the particles and to drive the fluid flow through the channel by means of electro-osmosis.4
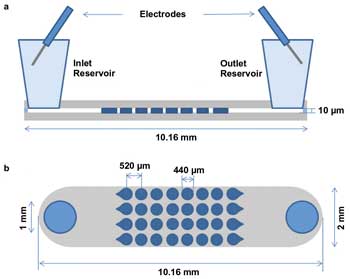
Figure 3. Schematic representation of the microfluidic channel setup for studying the parameters of the iDEP method, (a) side view and (b) top view.
Real-time examination of events in the microchannel was required over the course of the experiments to verify the predictive models. An inverted fluorescence video microscope was selected as the imaging platform for the iDEP experiments.
“The instrument was perfectly suited to our work,” said Héctor Moncada Hernández, a senior PhD student in Lapizco-Encinas’ laboratory. “We were able to construct our experimental setups directly on the microscope, as if it were a table. Since we were working with voltages of 1000 V or more, it was also important that we did not have a microscope objective or other metal parts in the field that could ground our electrodes.”
Figure 4 shows the setup for the iDEP experiments, in which a vacuum manifold and a circular glass microfluidic chip are placed atop the stationary stage of the microscope.

Figure 4. Experimental setup shows vacuum manifold and a circular glass microfluidic chip on the stage of the microscope.
In a first set of studies, a sample of fluorescent 1-µm microspheres was introduced at the inlet of the microchannel and a voltage applied to drive the fluid flow through the channel (Figure 5). Images and video of the fluid flow allowed the researchers to fine-tune the voltage, as well as the pH and conductivity of the suspending buffer, to maximize particle trapping (Figure 5a).
In a second set of experiments, a sample containing E. coli was introduced into the microchannel. The cells were labeled with SYTO 9 fluorescent dye for visibility. Whereas the illuminator of a standard microscope can quench the dye, leaving the cells indiscernible, the stroboscopic illuminator of the inverted fluorescence video microscope minimizes quenching, enabling high particle visibility over longer experiments.

Figure 5. A DC potential of 800 V is applied across a microchannel with suspended 1-mm microspheres. The particles appear in bright white between the insulator posts. Researchers used images such as these to fine-tune parameters to maximize particle trapping. (a) Particles are being successfully immobilized by employing a suspending medium with conductivity of 100 µs/cm and pH of 6. (b) Immobilization decreases when pH is increased to 9. (c) Immobilization decreases when conductivity is decreased to 25 µs/cm.4
In addition, the microscope’s epifluorescence camera module was used to improve visibility. “The epifluorescence capability helped us to see low concentrations of particles that we may have missed using standard illumination,” Lapizco-Encinas said. Figure 6 shows E. coli cells trapped at a potential of 1000 V, as imaged with standard illumination (left) and with improved visibility resulting from the use of epifluorescence (right).
A third set of studies shows the great potential of the iDEP method for rapid microorganism concentration and separation.5 Water from a pond was filtered to remove larger particles, then spiked with labeled E. coli and Saccharomyces cerevisiae yeast cells. An electric field was applied to immobilize and concentrate both types of cells. The field was then lowered to release the bacteria, and 30 seconds later, the field was lowered again to release the yeast cells.
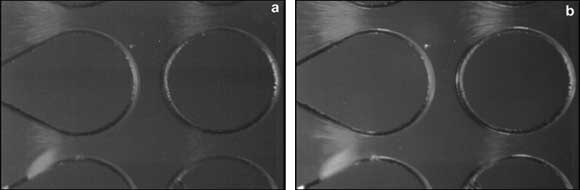
Figure 6. E. coli cells trapped at a potential of 1000 V, viewed with a 4x objective. (a) Image taken using standard LED illumination. (b) Same image, taken with the epifluorescence module, which improves imaging of low concentrations.
Fluorescence measurements made with the microscope allowed researchers to plot the passage of high concentrations of each type of cell as clearly separated events. Figure 7 shows a dielectropherogram of the events, in which an electric field gradient eluted the concentrated cells from the microchannel.
A further advantage of the inverted fluorescence microscope is its integrated software for capturing images and video of fluid flow and analyzing the results. The software includes facilities for automatically triggering video capture based on the presence of microparticles or other events. This capability ensures that critical events are not missed, even over the course of hours- or days-long experiment runs. Particle imaging velocimetry tools are integrated with the microscope software, which allows researchers to easily map the velocity of the flow through microfluidic channels.4
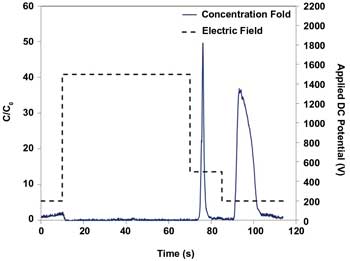
Figure 7. Dielectropherogram shows “peaks” of concentrated E. coli and S. cerevisiae cells as a function of time and applied DC voltage. Simultaneous concentration and separation in <2 min was visualized with the microscope by fluorescence measurements.5
The inverted fluorescence synchronized video microscope addresses the requirements of researchers working with micro- or nanofluidic experiments. It brings together the features required for microfluidics work, including a stationary stage, high-quality imaging, and software to capture publication-quality images and video.
Meet the authors
Dr. Yolanda Fintschenko is the director of new technologies, marketing and sales at LabSmith Inc. in Livermore Calif.; e-mail: [email protected]. Dr. Blanca H. Lapizco-Encinas is associate professor of chemical engineering and director of the Microscale Bioseparations Laboratory at Tennessee Technological University.
References
1. E.B. Cummings et al (2003). Characterization of electrokinetic mobility of microparticles. Anal Chem, Vol. 75, pp. 4724-4731.
2. B.H. Lapizco-Encinas et al (2004). Dielectrophoretic concentration and separation of live and dead bacteria in an array of insulators. Anal Chem, Vol. 76, pp. 1571-1579.
3. H.A. Pohl (1951). The motion and precipitation of suspensoids in divergent electric fields. J Appl Phys, Vol. 22, pp. 869-871.
4. J.I. Martínez-López et al (2009). Characterization of electrokinetic mobility of microparticles in order to improve dielectrophoretic concentration. Anal Bioanal Chem, Vol. 394, pp. 293-302.
5. H. Moncada Hernández et al (2010). Simultaneous concentration and separation of microorganisms: insulator-based dielectrophoretic approach. Anal Bioanal Chem, Vol. 396, Issue 5, pp. 1805-1816.