New systems take researchers inside the workings of tissue, cells, organelles
The biomedical industry is constantly involved in biological research and development, not only examining the basic functions of cells and tissues to find targets for therapeutic intervention, but also testing new and existing drugs. Microscopy has been an essential tool for the pharmaceutical industry for many years, helping to visualize the action of drugs on tissue, cells and cellular organelles.
Most light microscopes see a very thin, two-dimensional slice of a specimen, leaving the foreground and background out of focus. A 3-D image is then built up from the 2-D slices using computer software.
This 3-D image of a mouse brain expressing yellow fluorescent protein in neurons down to 4 mm deep was captured using an Olympus FluoView FV1000-MPE multiphoton microscope system. Raw image data courtesy of Hiroshi Harna, Hiroshi Kurokawa and Atshushi Miyawaki. The image was processed in Imaris software (Bitplane). Courtesy of Olympus America Inc.
Some 3-D microscopy systems allow investigators to view much thicker tissue preparations without having to make hundreds of individual sections that must be viewed and analyzed to piece together a 3-D view of the preparation. These systems can save time in viewing and analyzing tissue.
For example, researchers at Riken Brain Science Institute in Japan have developed a liquid that effectively turns tissue clear and minimizes light scatter. This reagent can be used with either of two specifically designed objectives from Olympus America Inc., a provider of research microscopy and imaging systems for pharmaceutical, biotechnology and other applications.
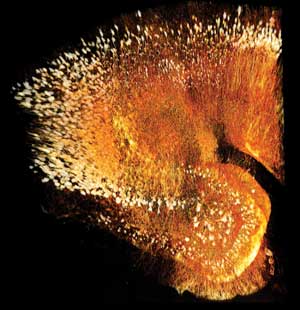
The resulting system, Scaleview, enables scientists to produce vivid 3-D images of structures deep inside mouse brains and other animal organs – up to 8 mm deep. Until now, even advanced optical methods did not allow researchers to see this deep beneath the surface because of light scatter within the tissue.

This 8-mm-deep image of mouse brain was captured using the Olympus FluoView FV1000-MPE microscope, the 8-mm Scaleview objective and the Scaleview-A2 reagent. Raw image data courtesy of Hiroshi Harna, Hiroshi Kurokawa and Atshushi Miyawaki, Riken Brain Science Institute, Laboratory for Cell Function Dynamics, Japan. Courtesy of Olympus America Inc.
The reagent, together with a 25x, 1.0-numerical aperture (NA), 4-mm working distance or a 0.9-NA, 8-mm-working-distance companion objective, is now available to researchers in North and South America.
“Particularly, 3-D microscopes can be useful in natural products’ drug discovery. There is much greater detail to be seen in fungi, plants and marine life, especially using fluorescent probes and chromagenic markers,” said Dr. Gary Greenberg, co-founder and president of Edge-3D LLC in Maui, Hawaii, a microscope technology company that specializes in 3-D.
“The microscopes enable deep-focus, real-time stereo and 3-D views,” he added. “In addition, in toxicity and efficacy screening for new molecular entities, the same applies. Thus, in thicker tissue specimens, dramatic changes can be observed, with such probes relating to efficacy or toxicity effects as well.
“Three-dimensional imaging also improves visualization by reducing misinterpretations and misdiagnoses. The popularity of confocal microscopes has highlighted the value of three-dimensional imaging.”
Confocal microscopy is one of the most important tools for examining the concentration and spatial distribution of fluorescently labeled target molecules and cells – and of their interactions in both fixed and living biological systems.
In traditional wide-field fluorescence microscopy, the entire specimen is lit evenly, exciting all parts of the specimen at the same time. The resulting fluorescence is detected by the microscope’s photodetector or camera and can include a large amount of unfocused signal that contributes to image haze.
In confocal imaging, only point illumination is used, and a pinhole placed in front of the detector serves to eliminate the out-of-focus signal. The only light produced by fluorescence to reach the detector is very close to the focal plane, which means that the images’ optical resolution can be much better than that produced by wide-field microscopes.
Because only one point in the sample is illuminated at a time, there is never a complete picture, and 2-D or 3-D scanning is required to build up a complete 2-D or 3-D picture.
These microscopes can be used in brain research to study the morphology of tissue with respect to neurodegenerative diseases such as Alzheimer’s and in developmental biology for cancer research.
Dr. Charles W. Hewitt, co-founder and CEO of Edge-3D, noted that drug development is highly dependent on the high-power compound light microscope (along with other microscopes such as transmission and scanning electron, atomic force and confocal – although the major use involves light microscopy).
“Many new drugs are derived from natural organisms, including bacteria, fungi, plants and marine life forms. All prospective drugs must pass extensive toxicity and pathology studies to prove safety,” he said. “This involves thorough examination of all organ and tissue systems, along with studying cellular metabolism and molecular markers of activation. Many molecular markers are designed to be visualized or measured by the microscope.”
Leica Microsystems of Wetzlar, Germany, a producer of microscopes and other scientific instruments, works with the pharmaceutical industry to ascertain how certain drugs influence organisms, tissues and so on.
Drug efficacy can be studied using 3-D microscopy to get inside the morphology of healthy versus diseased tissue, to look for changes of structure and expressions of proteins, and to see where they occur.
“Many pharmaceutical companies have been increasingly interested in multiphoton microscopes like the Olympus FV1000-MPE for intravital and animal imaging applications,” said Brendan C. Brinkman, a former core facility manager at the University of California, San Diego, and now product manager of laser scanning confocal microscopes in the Scientific Equipment Group at Olympus America. “Deep tissue can be penetrated by multiphoton instruments while creating high-resolution 3-D images.”

A photograph of the stigma of a Noni plant is reconstructed from 30 through-focus images, so that the whole stigma can be seen in 3-D. The plant is used in a variety of treatments by Hawaiians and is currently being studied by scientists for its bioactive compounds. This extended-depth-of-focus image was taken on the Edge-3D microscope. Courtesy of Edge-3D.
Bioengineering firms are increasingly using nonlinear optical techniques such as second-harmonic-generation imaging to view biological structures, including collagen matrices, without the photobleaching, phototoxicity or other negative effects of fluorescent labeling, Brinkman said. Bioengineers also are taking advantage of depth penetration for quality assurance of biomechanical structures.
“Other imaging modalities such as CARS [coherent anti-Stokes Raman scattering] are allowing for intrinsic imaging of lipids for applications as diverse as obesity drug testing, transdermal drug delivery, atherosclerosis and demyelinating disease interventions,” he added.
Other techniques
As the use of fluorescent labeling techniques has proliferated, both via specialized targeted fluorescent compounds and increasingly sophisticated transgenically modified model organisms, the ability to measure the interactions of molecules in real time by confocal techniques such as fluorescence lifetime imaging microscopy, fluorescence resonance energy transfer, fluorescence correlation spectroscopy and raster image correlation spectroscopy are becoming more common. The special ability of point-scanning confocal microscopes to direct light to specific areas has enabled optogenetic experimentation with important ramifications for neurobiology and testing of psychoactive agents.
“At the same time, innovative, advanced confocal instruments that are easier than ever to use and place in the lab have made use of confocal technology routine for clear, high-quality image documentation of cell proliferation and differentiation assays, both in fixed and live-cell applications,” Brinkman said.
Edge-3D also has ease of use in mind with its latest products. The company is developing real-time 3-D microscope technology for viewing both fluorescent and bright-field analytes, and developing unique 3-D software for viewing and analyzing such specimens.
The most promising instrument from Edge-3D is its high-magnification 3-D microscope, Greenberg said, which will be an alternative to low-power stereomicroscopy. Aside from the already available confocal technology, the company said its new microscopes will be affordable and available in mid-2012.
Superresolution microscopy
There is a growing trend toward high-content screening microscopy as well as superresolution microscopy, said Imtraud Steinmetz, applications specialist at Leica Microsystems.
Superresolution microscopy allows scientists to go beyond the diffraction limit, revealing subcellular structures in detail previously possible only with electron microscopy.

The Leica TCS STED CW system has made superresolution an indispensable method in many life sciences research institutes working with light microscopes. ©Leica Microsystems.
This trend is illustrated by a recent agreement signed by Leica Microsystems, the Max Planck Society in Germany and the German Cancer Research Center. For details on that agreement, see the BioScan article “Gated STED pushes boundaries of superresolution microscopy” on page 18.
Future opportunities
Previously, one main challenge with 3-D microscope technology was mass viewing: There was no convenient method to allow an audience or fellow colleagues to view stereo 3-D findings simultaneously. But this is changing rapidly. Many electronics companies have developed 3-D viewing platforms.
“Soon, it won’t even be necessary to use the 3-D glasses,” Greenberg said. “It will be immensely convenient to see the real microscopic imagery on such platforms by a group of colleagues, students or by an audience.”
For Brinkman, there is another important feature in 3-D microscopy technology that will be a help to pharmaceutical companies: increasing automation. This will provide greater reliability and throughput in testing pharmaceuticals on various biological systems using multiwell chambers and plates as well as lab-on-a-chip-type technologies.
“Optical advances that allow for imaging of whole organs of model organisms at high resolution will help expand our understanding of biological changes made by pharmaceutical interventions,” he said.