Muscles can move quickly – and individual proteins move even faster. So how do you capture such blazingly fast events?
In the study of biological systems – cell biology being the premier example – nonlinear microscopy techniques are highly regarded for their ability to focus tightly on minute parts of a structure. Two-photon excited fluorescence (TPEF) and second-harmonic generation (SHG) microscopy, in particular, have overtaken even such stalwart techniques as confocal laser scanning microscopy because they reduce the amount of photobleaching endured by the key fluorophores used to illuminate targets such as cell components.
“Damage induced by a laser remains as one of the most important challenges for a microscopist,” said Pablo Loza-Alvarez, leader of the superresolution light microscopy and nanoscopy facility at the Institute of Photonic Sciences in Barcelona, Spain. “SHG, THG [third-harmonic generation] or any other label-free technique will not avoid laser-induced damage, but they will certainly reduce the sample-manipulation and related problems of having to chemically or genetically modify the sample.”
SHG microscopy and other nonlinear techniques are fluorescent-label-free but may not provide all of the information needed to understand the imaged target. Also, these methods require the use of ultrafast lasers – typically, Ti:sapphire instruments – but their cost, complexity and general fussiness have led many microscopists to seek other solutions. More compact devices have been auditioned as potential light sources, but most tend to have neither the peak power nor the correct wavelength output for the task.
“All imaging techniques provide different but complementary information from [a] sample,” Loza said. “In particular, both SHG and THG can produce contrast in a label-free fashion. However, these two techniques have limitations, and there are a number of situations in which you cannot use SHG or THG.”
To balance the need for high (but not too high) output power and repetition rate while maintaining small size and low cost, Loza and his colleagues turned to a combination of a vertical external cavity surface-emitting laser (VECSEL) and a quantum-dot-based semiconductor saturable absorber mirror (SESAM). They chose VECSEL technology because it offers high beam quality and stability as well as short pulses and high power, and because it can be tuned to operate at several wavelengths key to two-photon fluorescence imaging.
The system built by Loza’s group is centered on a Nikon inverted microscope and a mode-locked VECSEL from M-Squared Lasers (Figure 1). The laser features a central wavelength of 965 nm, a pulse rate of 1.5 ps and a repetition rate of 500 MHz. The researchers collected the SHG two-photon fluorescence signals with a pair of Hamamatsu photomultiplier tubes.
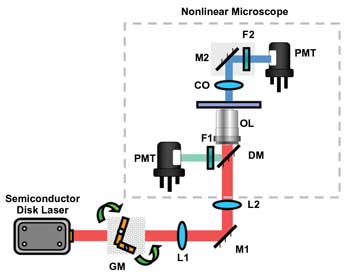
Figure 1. A compact semiconductor disk laser, or VECSEL, mode-locked by a quantum-dot-based semiconductor saturable absorber mirror permits microscopic imaging with fluorescent dyes while also providing second-harmonic information. The red path shows the excitation beam, the blue path, the SHG emission and the green path, the two-photon excitation emission. GM = galvanometric mirror; L = lens; M = mirror; F = bandpass filter; OL = objective lens; DM = detection mount; CO = condenser optics; PMT = photomultiplier tube. Courtesy of Biomedical Optics Express.
They used quantum dots in the system’s mode-locking SESAM design instead of more typical quantum-well-based materials for several reasons, Loza said. The quantum dots have fewer losses as well as less fluency and significantly broader spectral bandwidths. Lastly, quantum-dot structures make available several recombination paths, enabling ultrafast recovery under both absorption and gain conditions.
When used on Caenorhabditis elegans samples, the researchers found that the VECSEL/SESAM design permitted efficient imaging using green fluorescent proteins (GFPs). Importantly, the system also generated an SHG signal that provided additional information. The emission wavelength of the VECSEL, also called a semiconductor disk laser (SDL), is at the peak of the two-photon-action cross section, thus relaxing the amount of peak power needed to excite the GFP, Loza explained.
“Considering that many applications in many labs are based on GFP imaging, this SDL could be an interesting alternative nonlinear laser source in TPEF microscopy,” he said. “Importantly, the SDL can also be used, in a simultaneous way, to produce an SHG signal. This adds versatility to the use of this laser and to the microscopy system itself.”
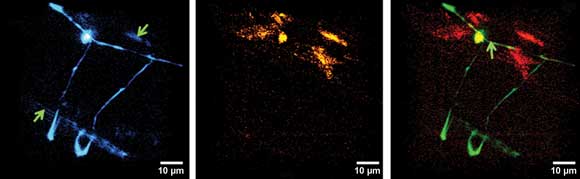
Figure 2. a) Two-photon excited fluorescence (TPEF, shown in blue) in a set of motor neurons expressing GFP inside C. elegans. b) SHG signal (orange) from the vulval muscles of the same sample. c) A merged image showing both TPEF and SHG images. Courtesy of Biomedical Optics Express.
The researchers are working on a new generation of VECSEL-based systems, which they believe will produce shorter pulse durations at larger repetition rates. This, they say, will increase the available peak power and therefore have a more efficient nonlinear interaction.
Measuring muscle movement
Most people move at a languid pace, but athletes are nearly always in motion. Some, like runners, move at a steady pace for determined periods, even if they’re sprinting. Others, such as basketball players, mainly move at a brisk pace for several seconds at a time, then make quick jumps, sudden spins and swift runs toward an opponent. The faster the muscles go from zero to incredible is the province of trainers and physicians in the field of sports medicine.
Anytime a muscle moves, it becomes hyperintense; this state gradually decreases as the muscle comes to rest. Measuring muscle activity during moments of hyperintensity is a complicated task constrained by the need to balance noninvasive methodology with the need to get specific information during muscle motion.
Techniques such as electromyography, in which the electrical activity of the muscles is analyzed, are useful, but only to a point. They cannot evaluate deep-muscle activity, which is especially important to the study of the musculature of the body’s trunk.
“Many orthopedic sports doctors and athletic trainers take a deep interest in trunk muscle now,” said Noriyuki Tawara of the Japan Institute of Sports Sciences in Tokyo. “However, traditional noninvasive functional assessment technique of trunk muscle has been surface electromyogram. The limit of the surface electromyogram is that it is impossible to evaluate the deep-muscle activity.”
Magnetic resonance imaging (MRI), however, can evaluate muscle activity via the transverse relaxation time (T2) of exercised muscle, which increases compared with that of the muscle at rest, he said. An early technique, called muscle functional MRI (mfMRI), could visualize macro-muscle activity but requires long minutes to acquire data. Furthermore, Tawara said, mfMRI could be used to image only the musculature of the limbs – and then only if the muscles were exercised until they were fatigued.
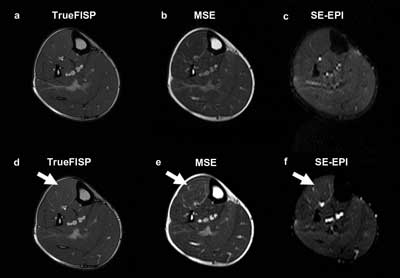
Figure 3. Magnetic resonance provides images of the lower leg at rest (a-c) and after exercise (d-f). Various MR techniques provide detail of muscle movements. TrueFISP = true fast imaging with steady-state precession; MSE = multiple spin-echo; SE-EPI = spin-echo echo-planar imaging. Courtesy of Magnetic Resonance in Medical Sciences.
“We wanted to get the detectability of the slightest impact on [deeper] trunk muscle activity induced by acute exercises,” he said.
To accomplish this, Tawara and his colleagues at Tokyo Metropolitan Hospital and at Nihon University, also in Tokyo, added some speed to mfMRI.
Their method combines signals from two pulse sequences: spin-echo echo-planar imaging (SE-EPI) and true fast imaging with steady-state precession (trueFISP). Standard mfMRI uses only SE-EPI to calculate T2. Fusing the T2 images with the trueFISP ones should provide the ultrafast muscle data required, they hypothesized.
Using a 1.5-tesla whole-body scanner made by Siemens AG of Erlangen, Germany, the group tested its fusion technique on the subjects who were instructed to move a particular muscle in their right leg while under tension.
“The results of our research demonstrated the tracking of changes of muscle’s MR signal intensity every 30 seconds,” Tawara said. “[Because] changes in MR signal intensity induced by exercise empirically return to rest quickly, traditional muscle functional MRI was applied only to evaluate the muscle activity induced by exercise until [exhaustion]. Using our method, the slight impact of muscle induced by exercise was captured by MRI.”
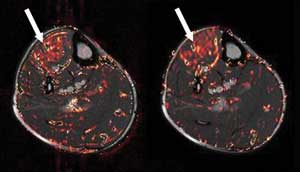
Figure 4. Fast-mfMRI fuses images from multiple spin-echo
(MSE) and spin-echo echo-planar imaging (SE-EPI) to increase the speed
of data acquisition and, thus, refine studies of muscle movement. Areas
of deep red indicate where muscle activation is greatest, whereas red in
nonmuscle regions is primarily blood vessels. Courtesy of Magnetic
Resonance in Medical Sciences.
One limit of the group’s fast-mfMRI technique is that image distortion sometimes comes into play, caused by configuration of the SE-EPI pulse sequence. Tawara said that his team is considering this problem now and also wants to develop the technique for whole-body imaging, including the neck, shoulders and pelvis.
Watching proteins fold
Driving every biological process are the molecules that make up every fraction of the corpus: Muscles, blood and neurons, and every bone, segment of flesh and organ can be considered a result of molecules in motion. Certainly, individual molecules might make an enzyme or other protein that, in turn, affects a biological process, but how each protein forms and, more importantly, the shapes the protein can take, also have a bearing on events.
X-ray crystallography and nuclear magnetic resonance (NMR) can be used to study the structures of most proteins, but they cannot reveal the changes in a protein’s or enzyme’s molecular conformations unless they are moving very slowly. Unfortunately, most protein conformational changes in particular move very fast – in a few femtoseconds or so.
For example, x-ray crystallography is wonderful for determining the structure of proteins as static molecules, said Michael D. Fayer, a professor of chemistry at Stanford University in California. Likewise, NMR frequently is used to obtain structural information for proteins that cannot be crystallized, and NMR and techniques such as single-molecule spectroscopy can obtain information of molecular motions on slow timescales, he added. More rapid changes, which can determine specific biomolecular actions, cannot be visualized with these methods.
In a paper published online by Accounts of Chemical Research on March 20, Fayer and his colleague Megan C. Thielges describe a novel technique for exploring these ultrafast structural changes. Dubbed two-dimensional infrared vibrational echo spectroscopy, the method shares kinship with 2-D NMR, but reveals processes at much shorter timescales.
“Two-dimensional IR vibrational echo methods allow the very fast structural dynamics of proteins to be examined on the 100-fs to 100-ps timescale,” Fayer said. “Such fast structural fluctuations allow a protein to sample its energy landscape and find configurations appropriate for a particular function.”
For example, a protein such as hemoglobin binds oxygen at its active sites, but there is no direct channel from outside the molecule to these sites. Instead, oxygen and other small ligands move through the hemoglobin structure – like characters from “The Matrix” dodging bullets – because the protein structure itself is making very fast changes.
The researchers used a Ti:sapphire oscillator and a regenerative amplifier to pump an optical parametric amplifier and difference frequency stage. The regenerative amplifier boosted the low-power 800-nm pulses to high-energy ones. The resulting 5-µm beam comprised pulses about 90 fs long. The beam was split into three excitation pulses (Figure 5). The delays between the pulses created variations in the phase relationships between them and, ultimately, a vibrational echo signal.
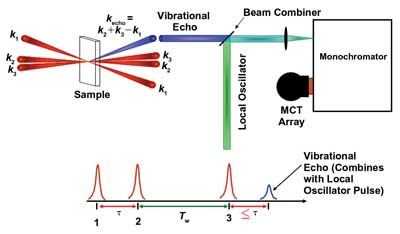
Figure 5. The novel pulse sequence and geometry of the 2-D infrared vibrational echo spectroscopy technique permits a look at ultrafast protein folding. Courtesy of Accounts of Chemical Research.
This echo signal was overlapped with a fourth beam called a local oscillator. Spectra from these signals garnered information on frequency changes in the C-O stretch within the protein (or other molecule being studied) as it underwent its conformational twists and turns.
The researchers’ 2-D IR vibrational echo experiments allow the measurement of timing and magnitude of structural fluctuations. The technique can not only visualize changes in a protein’s or enzyme’s structure when it binds to a substrate, but also how various substrates influence structural fluctuations and their correlation with chemical reactions.
Fayer and Thielges used their technique on two proteins, myoglobin and cytochrome P450cam. CO binds the active sites of both of these heme proteins. The researchers bound cytochrome P450cam to five different substrates and measured the dynamics of the protein with each. They found a strong correlation between the structural fluctuations of the enzyme on the 100-ps timescale and the binding affinity and the specificity of hydroxylation of the substrates, Fayer said.
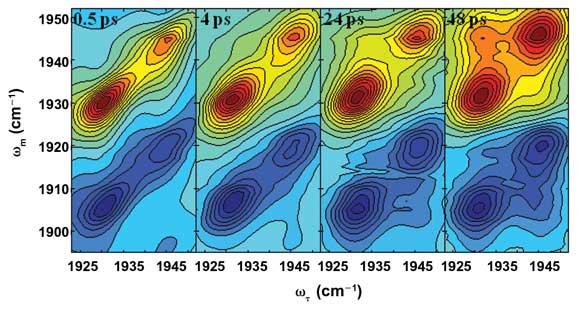 Figure 6. Two-dimensional IR spectra show CO during conformational changes in myoglobin over time. Courtesy of Accounts of Chemical Research.
Figure 6. Two-dimensional IR spectra show CO during conformational changes in myoglobin over time. Courtesy of Accounts of Chemical Research.
“For a protein … undergoing equilibrium structural fluctuations, there is no initial or final form, but rather a continuous sampling of a range of structures,” he said. “This sampling can be around a relatively deep minimum on the energy landscape. Then there can also be transitions from one substate to another. These substates reflect different relatively deep minima on the landscape.”
Currently, however, 2-D IR vibrational echo spectroscopy cannot provide detailed structural information, Fayer said.
“Advances are being made, but, so far, structural information is limited to small peptides or qualitative details of protein structure,” he noted.
The next step for Fayer and Thielges is to improve overall sensitivity and data collection speed. The next generation of IR detectors should boost sensitivity, Fayer said. Speed will come from tweaking the existing system.
“We have recently constructed a 2-D IR system that uses pulse shaping to change the timing between pulses rather than mechanical delay lines,” he said. “The pulse-shaping approach greatly speeds data acquisition and gives control of the phases of the pulses, which permits data to be taken in new ways.”
The pulse-shaping approach is fast, he added, but not as sensitive as the earlier version, which uses mechanical delay lines. Both systems have their place, he said.
“Two-dimensional IR vibrational echo spectroscopy is in its early days,” he added. “In 1960, an NMR spectroscopist would never have imagined many of the current applications of NMR. For 2-D IR, advances in lasers and detectors – as well as in methodology – will increase the applications.”