Stephan Briggs, Edmund Optics
Biocompatible microparticles allow noninvasive, optically driven on-demand controlled release of methotrexate to treat psoriasis.
Drugs are conventionally delivered orally or intravenously – but “conventional” does not always mean “ideal”: Oral drugs yield reduced bioavailabilities, and intravenous delivery is not practical for home administration.
The primary treatment for psoriasis, a chronic autoimmune disease that affects the skin and joints, is a drug called methotrexate (MTX). When given orally or intravenously, overdoses are common and may result in liver toxicity or failure – even, ultimately, death. In addition, intravenous delivery is painful and requires administration by a trained physician. Therefore, topical application has been suggested as a preferred treatment method for this condition.
Researchers have studied using transdermal delivery of microspheres loaded with MTX and gold nanorods.1 The goal is to reduce the need for frequent applications of MTX, and the research focuses on the method of delivery and the vehicle, a biocompatible polymer-based (alginate-chitosan) microparticle with embedded gold nanorods, used to incorporate MTX. Upon exposure to near-infrared (NIR) light, the thermal energy of the gold nanorods will increase and convert to resonant energy, disrupting the bonds within the polymer microparticles and allowing for a small release of MTX.
Necessary to the design is that the user must be able to control the drug’s release from the microparticle through exposure to NIR light; also, the microparticle must degrade in a time-dependent way upon NIR exposure. “Any microparticle or drug remaining should be minimal, quantifiable and nonharmful to the user, and a given number of microparticles must be adequate to encapsulate a therapeutic amount of the given drug,” researchers wrote in 2010.1
Market analysis
Drug-delivery technology became its own market and focus, separate of pharmaceuticals, during the 1950s and 1960s. The first sustained-release oral drugs appeared at that time through developments by Smith Kline & French Laboratories, known today as GlaxoSmithKline.2 The earlier medications and drugs were crude, to say the least, and solely intended to provide short relief and therapeutic care. In the late 1970s, advanced drug delivery continued to evolve and began to tackle fundamental problems associated with pharmacotherapy. The mid-1990s identified six commercial reasons for development and research into this field: convenience and compliance, efficiency, franchise protection, generics, market expansion and new markets.
The overall analysis can be segmented by market size, region, therapeutic analysis and route of administration. The concentration of this study was on transdermal delivery, so much of the focus will be targeted to the skin and topical treatments.
Overall, the market assessment is a bit convoluted: Various regions of the world regulate treatments more than others do, and other drugs or deliveries are available only in certain parts as well. The annual value of the global advanced drug delivery market was $16 billion in 1997. With an average 20 percent annual growth rate, the market in 2000 was approximately $27 billion; 2010 was approximately $171 billion; and today, it is approximately $250 billion. Values may have shifted slightly, as reports and regions are calculated differently. The regional division of the advanced drug delivery market in 2001 was heavily shared in North America (33 percent), Europe (28 percent), Japan (18 percent) and the rest of the world (21 percent).2
The analysis by therapeutic area is heavily dominated by a few large players. These include cardiovascular drugs, anti-inflammatory drugs, anti-cancer drugs, anti-asthma therapies, diabetes treatments and gene-therapy drugs. Many drugs overlap in their areas of treatment, so it is very difficult to say which therapeutic area has the largest presence.
The analysis by mode of administration is a bit easier to judge. Oral, inhalation, transdermal, mucosal and parenteral drug delivery make up the primary areas of administration. Of these, in the year 2000, oral delivery made up 50 percent of the total share, with parenteral and inhalation making up 19 percent, respectively, and transdermal and mucosal filling in the final 12 percent at an even split.2

An infinity-corrected objective with 2x magnification is used for maximum focus and transmission of 780-nm light from an LED array.
The transdermal market, still relatively young, had its inception in the 1990s with the advent of the nicotine patch for aid in quitting smoking. Another heavily used application was that of hormone replacement treatment and therapy for the elderly and those deficient in proper hormone levels. In 2001, the transdermal market was approximately $3 billion and in 2006, approximately $7 billion. There was a major jump in 2010 to $21 billion, and transdermal delivery today services about 12 percent of the global drug delivery market, which is estimated to reach a staggering $32 billion in 2015.3
Study criteria, constraints
The primary goal of the study was to encapsulate and release methotrexate from the microparticle, noninvasively, to treat psoriasis. The total treatment amount to be administered had to encapsulate five doses, or 6.25 mg, of MTX over a one-week period. This reduces the need to administer multiple applications of the drug that can potentially lead to irritation, inflammation and infection. It is recommended that MTX dosages do not exceed 1.25 mg per day. The application of NIR light, centered at 780 nm, should trigger a linear release profile of 1 mg of MTX, which is quantifiably less than the maximum dosage. Leakage of the drug from the capsules is possible but will be minimal, and this buffer alleviates any threat of overdosage.1
NIR light is emitted from a diode and focused onto the alginate-chitosan micro-particle with embedded gold nanorods by an infinity-corrected objective. This will cause the conduction electrons in the gold to absorb the light energy and convert it to heat energy. The subsequent temperature increase will cause the bonds between the alginate and chitosan to disrupt, leading to the release of encapsulated MTX into the skin, and will absorb into the epithelial cell junctions.
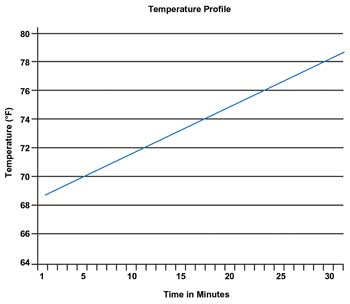
Temperature profile with a 9.1 °F (5.1 °C) increase over a 30-min period. T0 = 69.2 °F; Tfinal = 78.3 °F. Images courtesy of Edmund Optics.
Research has shown that a 4 °C change is needed to sufficiently disrupt the polymer bonds, so this is the target change in temperature required. The drug delivery vehicle must degrade in the body after the drug release is completed. The materials in the microparticles are alginate, chitosan and gold – all of which are natural materials. The level of gold in the body must be quantifiable, yet safe, and the gold nanorods should not exceed 1.5 µm.5 The gold nanorods chosen for this study have an axial diameter of 10 nm and a longitudinal length of 38 nm, well below the requirement of 1.5-µm maximum size.1
Methods, materials
The main component of the gold-NIR, light-to-heat conversion is the gold nanorod solution, which has peak absorption centered at 780 nm. Nanospheres typically are used for experimental procedures such as tumor ablation and drug delivery, but it has been proved that gold nanorods exhibit six times greater photothermal heat generation compared with nanopsheres. The gold nanorods were acquired from Nanopartz Inc. of Loveland, Colo., and the improved photothermal efficiency allowed for shorter NIR exposure and greater temperature increase while using a smaller concentration of nanorods. A 100-ml vial of gold nanorods can cost more than $500. To mitigate cost, lower concentrations of the gold nanorods should be used whenever possible.1
The optical apparatus assembled for testing the gold nanoparticle temperature profile used an infinity-corrected objective optimized for maximum focus and transmission of 780-nm light, an array of LEDs with peak illumination at 780 nm, along with a variety of standard spherical optics to collimate the output of the LEDs for maximum focusing efficiency. Because the LED array was a single band, standard planoconvex lenses were used because chromatic aberration was not an issue.
Achromatic and aspheric lenses were explored and yielded similar results, but to reduce cost, the design implemented two planoconvex lenses. The infinity-corrected objective was required to achieve the tightest spot of focus for maximum light-to-thermal energy efficiency. The LED array, planoconvex lenses and infinity-corrected objective were mounted, aligned and centered using an optical cage system.
MTX is an expensive, toxic and harmful substance to handle, so methylene blue (MB) was used in the studies. It has a similar molecular weight to MTX, and the release of the dye can be easily observed because it is bright blue in color. Low-viscosity sodium alginate was amalgamated with 5 millimoles of MB, then added to 750 millimoles of calcium chloride to create 2 percent calcium-alginate particles using a syringe and pump for consistency in particle size. There was a major disparity in the size of the microparticles because human error was a factor when pressure was applied by hand.
The alginate particles with encapsulated MB were allowed to cross-link in the calcium chloride solution for 30 min, then washed in deionized water and placed in a 1 percent chitosan solution for one hour. The interaction between calcium chloride and sodium alginate is very rapid, and particles formed almost instantly once placed in solution. The chitosan shell was used around the alginate core because the ionic bonds of the calcium-alginate particles were too weak and loose, resulting in leakage of encapsulated MB. The alginate-chitosan microparticles are formed as a result of the opposite ionic charges, creating a strong ionic bond. The resulting interaction between the two polymers is a very strong chemical bond that strengthens the integrity of the microparticle. The method for creating the microparticles can be seen in the Figure to the left.1
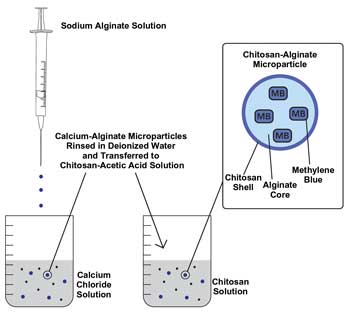
The process for creating the microparticles: The chitosan-alginate microparticles are formed by opposite ionic charges; chitosan’s charge is positive and alginate’s, negative. The resulting interaction between the two polymers is a powerful chemical bond that strengthens the integrity of the microparticle.1
Recommendations
The collected data yielded positive results and met the initial criteria and constraints. Initial testing and proof of concepts were performed in well plates and petri dishes. Methylene blue release profiles in polymer shells with gold nanorods induced by NIR light were then performed in porcine skin. The tests must be transferred over to in vitro studies using epidermal and dermal layers from a human. The properties should transfer from the porcine to human, but to fully understand the release profile, this study will have to be conducted.
Improvements in the polymer bond of alginate and chitosan must be addressed to ensure that no leakage occurs and that the structural integrity of the polymer micro-sphere remains intact. Lastly, the distribution of gold nanorods in the polymer microsphere must be further investigated. The ratio of gold in one sphere must be equal to the ratio of spheres in all of the other spheres.
Meet the author
Stephan Briggs is a product line engineer at Edmund Optics in Barrington, N.J.; email: [email protected].
References
1. K. Adlon et al (August 2009-June 2010). Biocompatible microparticles for optically driven on-demand controlled drug release of methotrexate. Drexel University, Biomedical Engineering Design Team 11, master’s in science design engineering project.
2. A.M. Hillery et al, eds. (Sept. 27, 2001). Drug delivery and targeting: For pharmacists and Pharmaceutical Scientists. First Edition, CRC Press, Taylor & Francis Group, New York.
3. S. Baker (Sept. 9, 2011). Transdermal delivery market predicted to reach $31.5 billion by 2015: PharmaLive special report. Retrieved March 2012 from Pharmaceutical & Medical Packaging News: http://www.pmpnews.com/news/transdermal-delivery-market-predicted-reach-315-billion-2015-pharmalive-special-report.
4. P. Lakshmi et al (2007). Niosomal methotrexate gel in the treatment of localized psoriasis: Phase I and phase II studies. Indian J Dermatol Venereol Leprol, pp. 157-161.
5. U.I. Walther et al (2002). Cytotoxicity of ingredients of various dental materials and related compounds in L2- and A549 cells. J Biomed Mater Res, Vol. 63, Issue 5, pp. 643-649.
Important terms and concepts:
• Bioavailability: The rate and extent to which an active agent is absorbed and becomes available at the site of action and therefore gives a therapeutic response.
• Zero-order release: Drug release does not vary with time, so the delivery system maintains a constant effective drug level in the body for extended periods.
• Variable release: Delivery system provides drug input at a variable rate to match biorhythms and hormone levels.
• Targeted-drug delivery: Delivery system achieves site-specific drug delivery.
• Temporal-drug delivery: Control of delivery to produce time-specific delivery for prolonged or fast-acting release.