Biophotonics experienced and enabled a huge number of fascinating advances in 2012, and we at Photonics Media did our best to share them with industry, academia and the world at large. Here’s a look at some of the top-read biophotonics stories on Photonics.com from January to October.
FIRST PLACE: THE TOP STORY OF 2012
Quantum dots show great promise for biological imaging, but concerns have been raised about their potential for toxicity. So when an international team released this report deeming them safe for primates, the photonics community as a whole took notice.
Primate Study Deems QDs Nontoxic – at Least for the Short Term
BUFFALO, N.Y., May 22 – Cadmium-selenide quantum dots are nontoxic to primates over a one-year period, a study has found. The quantum dots could hold promise for treating or detecting diseases such as cancer through nanomedicine.

Transmission electron microscopy shows clusters of quantum dots. In
application, each cluster is encased in a single capsule with an average
size near 50 nm.
Cadmium-selenide quantum dots, among the most studied, are suitable not only for medical applications, but also as components for LEDs, quantum computers and solar cells.
The study, undertaken by the University at Buffalo (UB), the Chinese PLA General Hospital, China’s ChangChun University of Science and Technology and Singapore’s Nanyang Technological University, aimed to address concerns of health professionals who worry that quantum dots might be dangerous to humans.
Four rhesus monkeys were injected with cadmium-selenide quantum dots in the study. Their blood and biochemical markers were tested for more than 90 days, and their organs developed no abnormalities. They also lost no weight. Two monkeys were then observed for an additional year and also showed no signs of illness.

A solution of cadmium-selenide quantum dots glows orange under
ultraviolet light. This luminescence forms the basis for their use in
bioimaging. Images courtesy of University at Buffalo.
“This is the first study that uses primates as animal models for in vivo studies with quantum dots,” said Paras Prasad, a UB professor of chemistry and medicine. “So far, such toxicity studies have focused only on mice and rats, but humans are very different from mice. More studies using animal models that are closer to humans are necessary.”
Further research on the nanocrystals’ long-term effects in primates is needed; most of the potentially toxic cadmium from the quantum dots stayed in the liver, spleen and kidneys of the animals studied over the 90-day period. This is a serious concern that warrants further investigation, said Ken Tye Yong, a Nanyang Technological University assistant professor.
SECOND PLACE
Thousands of Cells Tracked via Lensless Imaging
LOS ANGELES, Sept. 18 – A novel lensless computational imaging platform is being touted as a new way to observe and track large numbers of rapidly moving objects under a microscope, capturing precise motion paths in three dimensions.
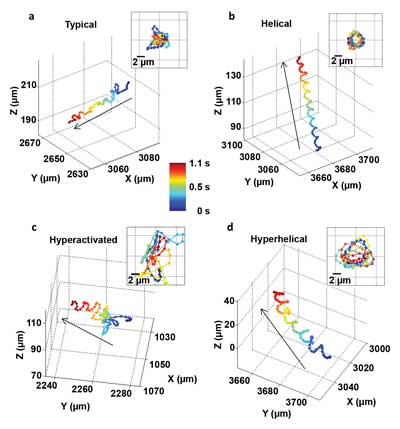
These images show, for the first time, that human male gamete cells swim primarily in four types of patterns. The insets show the front view of the trajectories.
For the first time, the 3-D helical swimming patterns of more than 24,000 individual human sperm cells were directly recorded for as long as 20 seconds over wide fields of view and through large sample volumes. The work was done by a team at the UCLA Henry Samueli School of Engineering and Applied Science, led by associate professor of electrical engineering and bioengineering Aydogan Ozcan.
“We can very precisely track the motion of small things, more than a thousand of them at the same time, in parallel,” Ozcan said. “We were able to achieve submicron accuracy over a large volume, allowing us to understand, statistically, how thousands of objects move in different ways.”
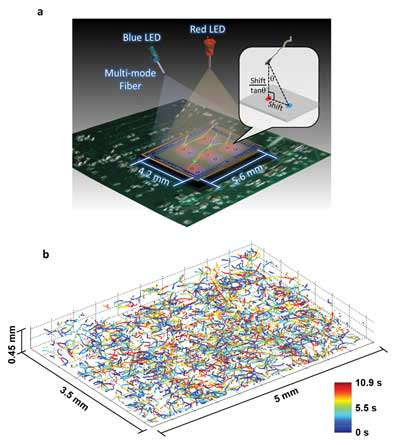
This illustration and data set depict the new microscopy technique developed by Ozcan and his colleagues at UCLA. The top image shows a schematic of the system, which involves two partially coherent light sources (a red 625-nm-wavelength LED and a blue 470-nm-wavelength LED) that simultaneously illuminate the microscope field of view from two angles. A CMOS sensor records the resulting holograms, and software uses that information to encode the exact positions of the target cells. The bottom image shows the reconstructed 3-D trajectories of 1575 human male gamete cells in a volume of 7.9 µl. Images courtesy of the Ozcan Research Group at UCLA.
Observing human sperm cells has generally been limited to conventional lens-based optical microscopes. The small size of a sperm head (about 3 to 4 µm) requires a high-magnification lens to observe its motion, and sperm cells’ relatively fast speed (about 20 to 100 µm/s) makes it difficult to track them over time as they move in three dimensions, into and out of the microscope’s small observation field.
To address these challenges, the UCLA team developed a computational lens-free, on-chip imaging platform that uses the holographic shadows of sperm cells. Lens-free images of the cells were acquired simultaneously using two different wavelengths of light, one red and one blue. The offset light beams created holographic information that, when processed using sophisticated software, accurately revealed the paths of objects moving under a microscope.
THIRD PLACE
Cancer Genes Detected with Liquid Lasers
ANN ARBOR, Mich., Feb. 1 – Genes linked to cancer and other diseases may now be easier to detect with a new method that uses liquid lasers to distinguish mutated DNA from healthy DNA by a single base.
Researchers at the University of Michigan say their technique works better than the current approach, which uses fluorescent dye and other biological molecules to find and bind to mutated DNA strands. When a patrol molecule catches one of the mutated genes, it emits a fluorescent beacon. Although it sounds effective, the patrol molecules tend to bind to healthy DNA as well, giving off a background glow that is only slightly dimmer than a positive signal.

Using this liquid laser, researchers developed a highly sensitive technique to identify mutated DNA that differs from healthy DNA by a single base. The fine white horizontal line is the capillary cavity that enables the laser to amplify the intrinsic difference in the light signals from healthy and mutated DNA. Courtesy of Nicole Casal Moore.
“Sometimes, we can fail to see the difference,” said Xudong Fan, an associate professor in the Department of Biomedical Engineering and principal investigator on the project. “If you cannot see the difference in signals, you could misdiagnose. The patient may have the mutated gene, but you wouldn’t detect it.”
In the conventional fluorescence method, the mutated DNA signal may be only a few tenths of a percent higher than the background noise. With Fan’s approach, the signal is hundreds of times brighter.
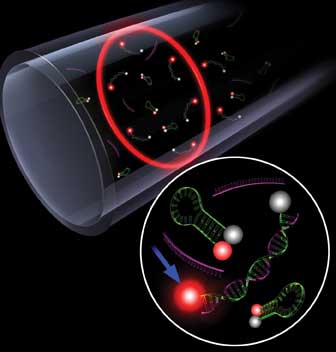
Researchers developed a highly sensitive technique based on laser emission for differentiating a target DNA strand from strands that contain single base mismatches. Laser emission amplifies the small difference in signals that are generated by the different strands after they bind with a molecular beacon. The conversion is similar to analog-to-digital. Courtesy of Christopher Burke.
Last year, Fan and his team discovered that they could employ DNA to turn a liquid laser on and off. His group is one of just a few in the world to accomplish this, Fan said. At the time, they had no practical applications in mind. That is, until now. They began to investigate what was causing the different laser outputs, using their findings to detect differences in the DNA.
FOURTH PLACE
Quantum Dots Switch Neurons On, Off
SEATTLE, Feb. 13 – Light from electrons confined by quantum dots has been used to activate and control targeted brain neurons, demonstrating a noninvasive method for studying cell communication and learning how specific cells may contribute to brain disorders.
The method offers an alternative to electrodes on the scalp or implanted within the brain; electrodes activate large areas of neural territory, making it impossible to target particular cells or cell types to understand cellular communication.
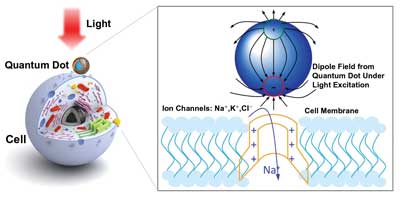
Optically excited quantum dots in proximity to a cell control the opening of ion channels. Image adapted from Jiang et al (2006), Chem. Mater., 18 (20), pp. 4845-4854.
For years, researchers sought methods that could activate nerve cells in a noninvasive way while also being highly targeted. A team of Stanford University researchers altered mammalian nerve cells to carry light-sensitive proteins from single-celled algae, allowing them to rapidly flip the cells on and off, just with flashes of light. But this photostimulation process required that the light-controlled cells first be genetically altered to flip the switch.
Now a team of scientists at the University of Washington, led by electrical engineer Lih Y. Lin and biophysicist Fred Rieke, has developed an alternative method that uses quantum dots to confine electrons within three spatial dimensions. When these otherwise trapped electrons are excited by electricity, they emit light, but at very precise wavelengths, determined both by the size of the quantum dot and the material from which it is made.
The team cultured cells on quantum dot films so that the cell membranes were in close to the quantum-dot coated surfaces. The electrical behavior of individual cells was measured as the cells were exposed to flashes of light of various wavelengths; the light excited electrons within the quantum dots, generating electrical fields that triggered spiking in the cells.
FIFTH PLACE
New Atomic X-ray Laser Created
LIVERMORE, Calif., Feb. 3 – The shortest, purest x-ray laser pulses ever achieved fulfill a 45-year-old prediction, opening the door to new medicines, materials and devices.
Stanford Linear Accelerator Center (SLAC) scientists created the pulses by aiming their Linac Coherent Light Source (LCLS) at a capsule of neon gas, setting off an avalanche of x-ray emissions to create the first “atomic x-ray laser.”

A powerful x-ray laser pulse from SLAC National Accelerator Laboratory’s Linac Coherent Light Source comes up from the lower-left corner (green) and hits a neon atom (center). Courtesy of Gregory M. Stewart/SLAC.
“X-rays give us a penetrating view into the world of atoms and molecules,” said physicist Nina Rohringer, who led the collaboration with scientists from SLAC, Lawrence Livermore National Laboratory and Colorado State University.
The new laser fulfills a 1967 prediction that x-ray lasers could be made by first removing inner electrons from atoms and then inducing electrons to fall from higher to lower energy levels, releasing a single color of light in the process. But until 2009, when LCLS turned on, no x-ray source was powerful enough to create this type of laser.
To make the atom laser, LCLS powerful x-ray pulses – each a billion times brighter than any available before – knocked electrons out of the inner shells of many of the neon atoms in the capsule. When other electrons fell in to fill the holes, about one in 50 atoms responded by emitting a photon in the hard x-ray range, which has a very short wavelength. Those x-rays then stimulated neighboring neon atoms to emit more x-rays, creating a domino effect that amplified the laser light 200 million times.
Rohringer said she now will try to create even shorter-pulse, higher-energy atomic x-ray lasers using oxygen, nitrogen or sulfur gases.