Dr. Kevin K. Tsia, The University of Hong Kong
A new platform delivers the best of both worlds – throughput and accuracy – for biomedical and life sciences applications.
Optical technology arguably has been playing a decisive role in advancing modern biomedicine. Enabling high detection sensitivity and/or high imaging resolution, optical microscopy and spectroscopy have been the gold standard adopted in all life sciences and clinical laboratories for shedding light on undeciphered biological processes – e.g., diseases – of tissues/organisms on the cellular and molecular levels.
However, an unspoken rule is applied to optical microscopy or spectroscopy: Higher measurement sensitivity comes at the expense of reduced measurement speed. This limitation stems from the fundamental trade-off between sensitivity and speed seen in all conventional digital imaging sensors; the key photodetection element in optical microscopes and spectrometers is the CCD or CMOS camera. Another speed obstacle is the additional time to read out the data from the sensor array. Higher frame rates can be obtained by a partial readout process that sacrifices the number of pixels and, therefore, the image or spectral resolution.
Currently, image sensors are mostly capable of monitoring real-time dynamics of the biological specimens with a timescale of no less than milliseconds (for imaging purposes) or tens of microseconds (for purely spectroscopy purposes). For a shorter timescale of nanoseconds or less, one would resort to pump-probe measurements or other similar approaches to capture the time-resolved dynamics in a repetitive manner, but not in real time. High-speed imaging/spectroscopy with real-time capability in such a short timescale, if realized, could bring new insight into the ultrafast and stochastic biological processes previously inaccessible.
More importantly, the aforesaid limitations of image sensors would become even more prominent in massively high throughput measurement and screening applications, such as drug discovery, aberrant-cell screening in stem cell research, rare-cancer cell detection, emulsion droplets/particle synthesis and histopathology. These often require enumeration and characterization of a large population of specimens (e.g., >100,000 particles) with stringent statistical accuracy, while keeping the measurement duration as short as possible.
However, it is challenging to enjoy high throughput and accuracy simultaneously. The detection sensitivity of the image sensors directly influences the measurement accuracy, whereas the detection speed links to the measurement throughput. This explains why the current state-of-the-art imaging flow cytometers that employ the conventional image sensors can achieve an imaging throughput of only ~200 to 1000 cells per second, compared with the throughput of ~100,000 cells per second of the classical non-imaging flow cytometers.1
An entirely new platform that can deliver the best combination of throughput and accuracy is pivotal not only in enhancing the efficacy and efficiency of the biomedical diagnostics, but also in fostering basic life sciences discovery. One example is the recent blooming interest in developing new approaches for single-cell analysis, which is intended to assess cell-to-cell differences and thus to elucidate the variation in disease processes within a large population of cells. To achieve this goal, we need a fundamentally new optical diagnostic (imaging or spectroscopic) tool. Optical time-stretch, a trick newly introduced to the realm of biophotonics, could benefit biomedical applications where there is a strong need for speed and accuracy.
Speeding up, stretching out
The central idea of using optical time-stretch (also known as dispersive Fourier transform) for imaging and spectroscopy is ultrafast retrieval of any information encoded in the spectrum of a broadband optical pulse by converting it into a
serial temporal data format in real time (Figure 1). Such mapping is done by stretching the pulse via group velocity dispersion in a dispersive medium – e.g., a dispersive optical fiber. The encoded information can be the spectral signature of a biological specimen and its associated dynamical processes, or a spatial image.
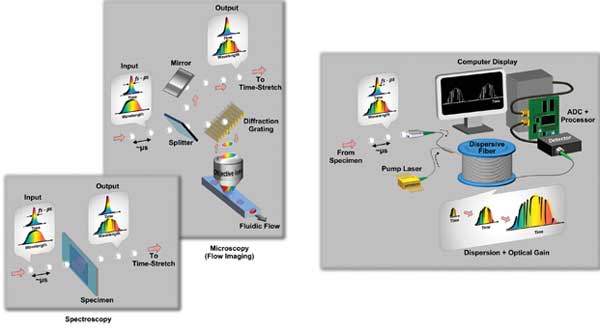
Figure 1. Schematics of optical time-stretch imaging and spectroscopy: (left) information encoding in the case of spectroscopy and imaging; (right) the pulse with the encoded spectrum then undergoes the optical time-stretch process for subsequent photodetection and signal post processing. The pump laser provides optical amplification of the encoded pulses within the dispersive fiber. Images courtesy of Dr. Kevin Tsia, The University of Hong Kong.
In the case of imaging, an additional mapping step is necessary before the time-stretch process – i.e., mapping the spatial information to the spectrum of the pulse by the use of diffractive optics. A single diffraction grating is used in the 1-D line-scan mode (particularly useful for unidirectional flow imaging), whereas an orthogonally oriented pair of a diffraction grating and a virtually imaged phased array is employed in the 2-D mode. The time-stretched pulse, now stamped with image or spectral information, is captured by a high-speed single-pixel photodetector and analog-to-digital converter, instead of the CCD/CMOS imager.
The key strengths of optical time-stretch are threefold: The first is ultrafast shutter speed – the temporal pulse width essentially defines the shutter speed in time-stretch spectroscopy and imaging. Hence, the use of ultrashort pulse (femtoseconds-picoseconds) provides extremely rapid spectral or image acquisition in a single shot.
The second strength is an ultrafast frame rate: Its time-domain operation uses a single photodetector and a high-speed electronic digitizer to acquire extremely high resolution spectra and images in real time. The spectral or image frame rate is governed by the repetition rate of the laser pulse train (typically on the order of 10 MHz for most of the femtosecond-picosecond lasers).
And the third strength is enhanced sensitivity: During the time-stretch process, optical amplification also can be incorporated to overcome the fundamental trade-off between optical loss and group velocity dispersion, circumventing the trade-off between sensitivity and speed. This is particularly useful if the captured spectral or image signal is buried below the noise floor of the photodetector.
So far, one of the most successful amplification schemes is stimulated Raman amplification in the dispersive fiber. Fiber Raman amplification has the advantages of widely tunable gain spectra and flexible gain spectral shaping by multiple-pumping schemes. Another viable approach could be fiber optical parametric amplification, which also offers wide gain bandwidth with dispersion engineering of the highly nonlinear fiber. From a practical point of view, even if the application does not require such ultrafast operation, the method enables more averaging, which on most occasions yields better spectral/image quality without severely sacrificing the required speed.
Unprecedented speed
Optical time-stretch has been successfully used for ultrafast real-time absorption spectroscopy of gas molecules with record high (sub-gigahertz) spectral resolution (Figure 2a).2 This could be useful for high-throughput exhaled breath analysis in which different molecular biomarkers – e.g., carbon monoxide, ethane, nitric oxide, ammonia – can be detected and differentiated in terms of their unique absorption spectra.
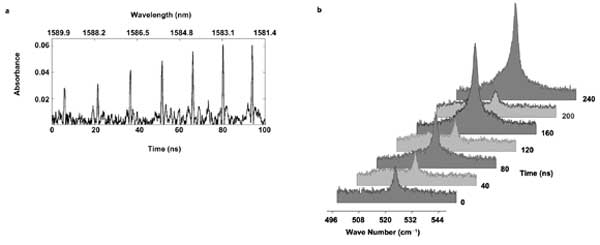
Figure 2. (a) Measured absorption spectral lines of carbon monoxide by optical time-stretch (high-resolution 8 pm or 950 MHz). (b) Temporal fluctuation (due to the unstable source) of the stimulated Raman spectra of crystalline silicon captured in real time by optical time-stretch.
These biomarkers have been found to be clinically relevant to disease diagnostics. In addition to absorption spectroscopy, optical time-stretch has been employed for high-speed stimulated Raman spectroscopy of crystalline silicon (Figure 2).3 This opens the door to realizing ultrafast and high-throughput molecular spectroscopy based on coherent Raman effects (e.g., stimulated Raman scattering or coherent anti-Stokes Raman scattering).
This is of great interest in the field of high-throughput drug screening and discovery, where Raman-based spectroscopy has been regarded as an ideal tool for providing the spectral fingerprint catalogs of individual unique drug molecules.
Tapping into different modalities
Introducing an additional space-to-wavelength mapping step, one could turn a time-stretch spectrometer into a time-stretch imaging system. Ultrafast bar-code reading, fluidic flow imaging and laser ablation dynamic monitoring have been made possible using time-stretch imaging, also coined in 2008-09 as serial time-encoded amplified microscopy (STEAM) – achieving a record high imaging speed of millions of frames per second.4
More recently, STEAM has successfully been employed for imaging flow analysis and screening of budding yeast and rare-cancer cells in blood with an unprecedented throughput of 100,000 particles per second and a record measurement accuracy – i.e., a false positive rate of 10−6 (Figure 3).5 This imaging throughput is not achievable with any existing optical imaging platform, including flow cytometry.

Figure 3. High-speed microparticle imaging in flow by STEAM (flow speed at 4 m/s – corresponding to a throughput of ~100,000 particles per second). The STEAM images are also compared with images captured by a high-speed CMOS camera that clearly show the motion blurs.
The ultrahigh-speed operation of optical time-stretch can be tapped into different optical imaging modalities – expanding the scope of its utility in biomedical diagnostics, such as differential interference contrast microscopy, quantitative phase-contrast microscopy and optical coherence tomography. Time-stretch differential interference contrast microscopy has been demonstrated for high-speed label-free imaging of white blood cells in microfluidic flow (~millimeters per second) with enhanced image contrast.6
To access more cell information, another label-free technique – quantitative phase-contrast imaging – can be incorporated with optical time-stretch. Recently, a laser vibrometer based on quantitative phase-contrast time-stretch imaging was developed to perform real-time ultrafast 1-D cross-sectional imaging of acoustic vibrations with nanometer resolution (Figure 4a).7 Further development could make time-stretch imaging more appealing for noninvasively (label-free) revealing the quantitative information of the cellular/subcellular structures (e.g., cell volume, mass, refractive index, stiffness) with nanometer precision and at ultrahigh speed – potentially a high-content, high-throughput and high-accuracy imaging platform.
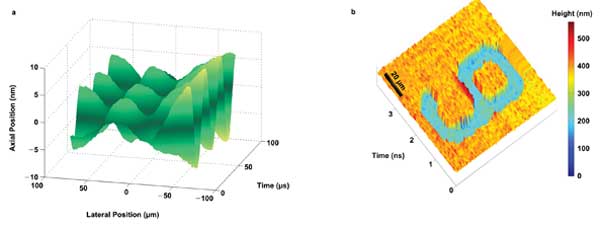
Figure 4. (a) Acoustic diaphragm vibration (at 30 kHz) detected by the STEAM vibrometer with 1-nm axial resolution and 30-ps dwell time. (b) Microscopic image of digit “9” captured quantitative phase-contrast time-stretch microscopy at 1 µm.
In many clinical diagnoses based on OCT – e.g., ophthalmology, dermatology and gastroenterology – it is always desirable to perform a complete volumetric (3-D) scan in real time or even at video rates, similar to the case of medical ultrasound imaging. In this scenario, the OCT line-scan rates have to reach beyond 1 MHz – a speed not easily achievable with the conventional OCT modalities. Although careful designs of the swept-source OCT based on a Fourier domain mode-lock laser have been implemented to accomplish multimegahertz OCT,8 optical time-stretch also naturally fits in well as an attractive alternative to achieve OCT at greater than megahertz. In time-stretch OCT, the time-stretch process can be implemented in the front end as an all-optical swept source at a sweep rate of >10 MHz9 without the mechanical wavelength-tuning mechanism employed in the typical tunable laser sources. More importantly, the sensitivity of time-stretch OCT can be greatly enhanced by the simultaneous optical amplification. Improved intensity noise of the broadband source – e.g., supercontinuum and optimized optical gain (>30 dB) – could result in an OCT sensitivity comparable to the existing Fourier domain mode-lock laser-based OCT.
The road ahead
Most previous work on time-stretch imaging and spectroscopy operated in the telecommunication band (1550 nm) because of the wide availability of the low-loss dispersive fibers in this wavelength range. In contrast, such fiber is not readily available in the near-IR regime (~800 nm–1 µm), which is well known to be the optimal diagnostic window in biophotonics. Thus, extending optical time-stretch to this favorable near-IR range is not straightforward. The first and foremost task is to identify the appropriate dispersive elements at these wavelengths.
To this end, 1-µm time-stretch microscopy has been demonstrated by using specialty single-mode fibers10 and telecommunication single-mode fibers as few-mode fibers11 (Figures 4b and 5a). They are found to be effective for achieving cellular resolution in bioimaging applications. An intriguing property of few-mode fibers is that the higher-order few-mode fiber modes could also be used for time-stretch applications. This additional degree of freedom could pave a new way for optimizing the time-stretch performance, such as by tailored dispersion engineering of the individual few-mode fiber modes. Notably, a chirped fiber Bragg grating has also been employed as a highly dispersive, low-loss medium, despite its limited bandwidth, for ultrafast time-stretch operation at 800 nm.12
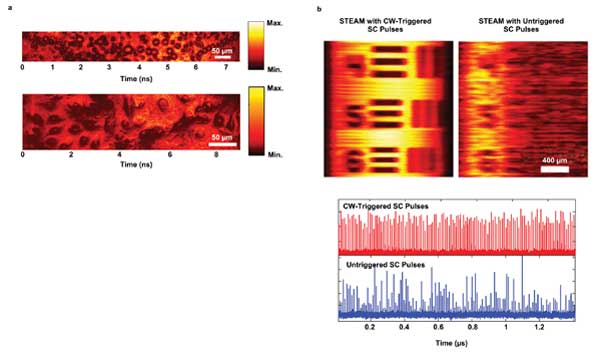
Figure 5. (a) 1-µm time-stretch images of (top) oil emulsion droplets and (bottom) nasopharyngeal epithelial cells. (b) STEAM images of a resolution target (USAF-1951), using a CW-triggered supercontinuum (left) and using an untriggered supercontinuum source (right). Also shown are the real-time pulse traces of the untriggered supercontinuum and the CW-triggered supercontinuum.
Temporal stability of the broadband source – e.g., supercontinuum – is another key enabling factor for high-quality time-stretch operation. Because the long-pulse supercontinuum generation (picosecond-nanosecond) is well known to result in an ultrabroadband spectrum with considerable pulse-to-pulse fluctuation, all the earlier works on time-stretch employed ultrashort-pulse femtosecond supercontinuum sources, which offer better temporal stability. A simple and handy approach has recently developed to actively stabilize the picosecond supercontinuum by adding a minute continuous-wave trigger – drastically altering the initial nonlinear dynamics in supercontinuum generation (Figure 5b).13 This demonstration could help extend the applicability of the supercontinuum sources in time-stretch from using the ultrashort-pulse (femtosecond) to the longer-pulse (picosecond-nanosecond) regimes.
Optical time-stretch has already opened a wide range of opportunities to accelerate the development of biomedical diagnostics where there are strong needs for scaling the throughput and enhancing the accuracy, e.g., high-throughput single-cell analysis. In its early adolescent stage, optical time-stretch could still find a lot of room for further tailoring to any specific imaging or spectroscopic arena, in conjunction with the continuing advances in laser sources, photodetectors and dispersive fibers.
It is vital to remember that realizing ultrafast real-time operation of time-stretch inevitably requires a state-of-the-art analog-to-digital converter (>multigiga samples per second) as well as high-performance computing processors, e.g., a field-programmable gate array or graphical processing unit – permitting the access and processing of enormous amounts of digital data in real time. Depending upon the targeted applications, tailored designs in both hardware (analog-to-digital converter and processors) and software (novel signal process algorithms) are necessary.
Meet the author
Dr. Kevin K. Tsia is an assistant professor at the University of Hong Kong in the department of electrical and electronic engineering, and in the medical engineering program; email: [email protected].
References
1. D.A. Basiji et al (2007). Cellular image analysis and imaging by flow cytometry. Clin Lab Med, Vol. 27, pp. 653-670.
2. J. Chou et al (2008). Real-time spectroscopy with subgigahertz resolution using amplified dispersive Fourier transformation.
Appl Phys Lett, Vol. 92, p. 111102.
3. D.R. Solli et al (2008). Amplified wavelength-time transformation for real-time spectroscopy. Nat Photonics, Vol. 2, pp. 48-51.
4. K. Goda et al (2009). Serial time-encoded amplified imaging for real-time observation of fast dynamic phenomena. Nature, Vol. 458, pp. 1145-1149.
5. K. Goda et al (2012). High-throughput single-microparticle imaging flow analyzer. PNAS, Vol. 109, pp. 11630-11635.
6. A.M. Fard et al (2011). Nomarski serial time-encoded amplified microscopy for high-speed contrast-enhanced imaging of transparent media. Biomed Opt Express, Vol. 2, pp. 3387-3392.
7. A. Mahjoubfar et al (2011). High-speed nanometer-resolved imaging vibrometer and velocimeter. Appl Phys Lett, Vol. 98, pp. 101107-101109.
8. W. Wieser et al (2010). Multi-megahertz OCT: High quality 3D imaging at 20 million A-scans and 4.5 GVoxels per second. Opt Express, Vol. 18, p. 14685.
9. K. Goda et al (2012). High-throughput optical coherence tomography at 800 nm. Opt Express, Vol. 20, 19612-19617.
10. T.T.W. Wong et al (2012). Optical time-stretch confocal microscopy at 1 μm. Opt Lett, Vol. 37, pp. 3330-3332.
11. Y. Qiu et al (2012). Exploiting few mode-fibers for optical time-stretch confocal microscopy in the short near-infrared window. Opt Express, Vol. 20, pp. 24115-24123.
12. C. Wang et al (2012). Dispersive Fourier transformation in the 800 nm spectral range, in CLEO: Applications and Technology, OSA Technical Digest, paper ATu2G.2.
13. C. Zhang et al (2011). Serial time-encoded amplified microscopy (STEAM) based on a stabilized picosecond supercontinuum source. Opt Express, Vol. 19, pp. 15810-15816.