
STED Microscopy: A New Chapter in Light Imaging
When Stefan Hell invented a technique that pushed beyond the diffraction limit of light for the first time, a new field of nanoscale imaging was born. Now laboratories around the world are using STED microscopy to smash through more boundaries in fundamental science.
Looking back over two decades of progress, the man behind the revolutionary light imaging technique says he is very satisfied with how STED (stimulated emission depletion) microscopy has evolved our understanding of biomolecules as well as transformed the way we think about how light behaves.
“As a result of this development, one is now able to unravel 3-D distributions of proteins and other biomolecules in [living] cells and tissues noninvasively, and with a resolution reaching down to a few [tens of] nanometers – i.e., almost to the size of the proteins themselves,” said Stefan Hell, inventor of STED and a director of the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany. “Two decades ago, this feat was considered plainly impossible.”
As the patent approaches expiration – Leica Microsystems GmbH of Wetzlar, Germany, is currently exclusively licensed to offer STED systems – Hell believes we are about to see a new wave of advances.
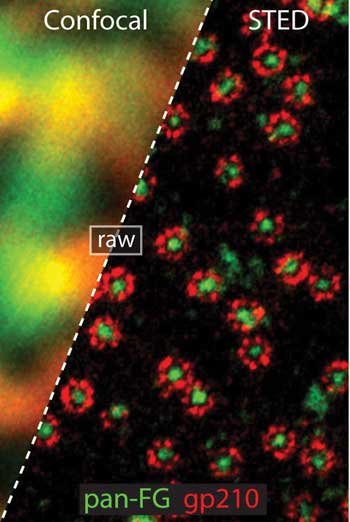
STED imaging showing two color colocalized recordings of nuclear pore complexes in amphibian
cells at resolution of 20 nm (red) and ~30 nm (green channel). The imaging is described in Biophys
J 105, L01 - L03, (July 2013). Göttfert et al.
“I anticipate the number of STED systems and STED users to grow very rapidly in the coming two years, as the basic patent on STED is expiring,” he said. “Although Leica has done a great job bringing STED superresolution to leading life-science laboratories around the world, competition between different companies will most naturally lead to improved performance in terms of speed, versatility and specified resolution, and make STED and STED-related microscopy methods standard in life-science laboratories around the world.”
Hell anticipates that STED systems will soon become available in all possible varieties: upright and inverted systems dedicated to live brain imaging, cell biology, correlation spectroscopy, the materials sciences and so on.
The popularity of STED microscopy comes from its incredible potential to not only image living cells but its speed, too; it is currently the fastest nanoscopy method, delivering time-lapse images of 60 to 80 nm in resolution at up to 80 fps.
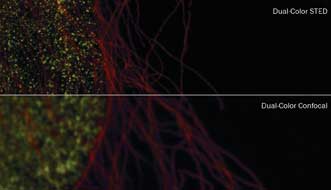
HeLa cells imaged by dual-color confocal (bottom) and dual-color STED image (top). Courtesy of Leica Microsystems CMS GmbH.
It is also remarkably flexible, having been applied to almost every area in which fluorescence plays a role, including the imaging of neurons in the brain of a living mouse.
Superresolution is used more and more as a standard tool in life-science research, with the majority of users interested in subcellular details, said Jochen Sieber, product manager of superresolution technologies at Leica Microsystems.
“Membrane research, cell [biology] and neurobiology are the major fields of applications. In my opinion, live-cell studies are not only the most interesting, but also the most challenging discipline of superresolution microscopy,” he said. “STED microscopy – with its ability to use common fluorescent markers like GFP [green fluorescent protein] and YFP [yellow fluorescent protein] as well as its capability to deliver superresolved images instantaneously without the need of image processing – [is] by far the most suited technology for this kind of study.”
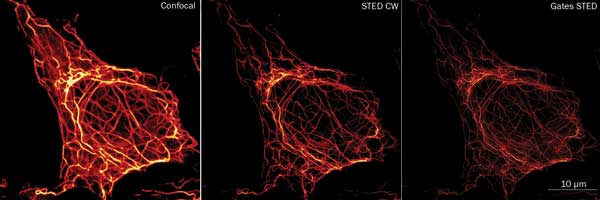
YFP-labeled keratin filaments in SW-13 cells: a comparison of confocal (left), STED CW (middle) and gated STED CW imaging. Sample courtesy of Dr. Reinhard Windoffer, RWTH Aachen University, Institute of Molecular and Cellular Anatomy, Aachen, Germany. Courtesy of Leica Microsystems CMS GmbH.
Christian Eggeling is a principal investigator at the MRC Human Immunology Unit and scientific director of the Wolfson Imaging Centre Oxford at the Weatherall Institute of Molecular Medicine of the University of Oxford in England. Among the advantages of STED microscopy, Eggeling highlights its targeted (scanning) readout, which delivers direct images.
“You see what you get without any possible bias by image analysis. In contrast, PALM [photoactivated localization microscopy]/STORM [stochastic optical reconstruction microscopy] have to use potentially biasing image reconstruction,” he said. “Due to its targeted readout, it can straightforward be combined with (single molecule) spectroscopy tools such as fluorescence correlation spectroscopy (STED-FCS) to highlight nanoscopic diffusion and interaction dynamics of different molecules.”
At the imaging center, Eggeling is introducing STED microscopy to less-experienced biomedical researchers, and his group is using STED microscopy and other superresolution techniques to unravel nanoscopic changes at the molecular level in living cells following cellular immune responses.
Infectious agents such as the influenza virus, HIV, dengue fever virus or the hepatitis C virus often cause severe symptoms and thousands of deaths each year. Eggeling hopes that understanding the full pathway of viral infection down to the cellular and molecular level could lead to successful treatment and prevention of these infections.
RESOLFT to overcome disadvantages
Since a lot of laser light is needed to prepare the “off” states required for STED imaging, some of this light can be accidentally absorbed, causing damage to living cells.
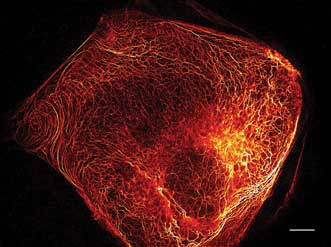
A RESOLFT image of a living mammalian cell expressing keratin plus a switchable GFP, as reported in the July 2013 issue of Nature Methods. It was recorded at low light levels using 116,000 doughnuts in parallel.
Hell’s Department of NanoBiophotonics came up with a way to reduce this problem by using time-gated STED microscopy. Gated STED, which has the ability to provide sharper images at lower power, was introduced in 2011 in a paper published in Nature Methods.
The group found that by applying pulsed excitation together with time-gated detection, the fluorescence on-off contrast in CW STED microscopy is improved. This means that finer details in fixed and living cells are revealed using moderate light intensities.
Leica Microsystems quickly harnessed this potential and developed a commercial implementation at high speed less than a year after publication of the first images.
But the ultimate solution and the next step forward in superresolution imaging could be RESOLFT (reversible saturable or switchable optical fluorescence transitions) microscopy, which uses a different photoswitching mechanism and therefore lower levels of light for scanning and/or gated detection, necessitating less laser intensity.
“Although this generalized STED concept called RESOLFT has been around for more than a decade, using cis-trans isomerization for spatially targeted on-off switching has become useful only recently, due to the advent of novel reversibly switchable fluorescent proteins, such as [redshifted GFP],” Hell said.
A major benefit of using cis-trans isomerization and metastable on and off states is that the intensity required to turn the fluorescence off (and to create an on-off state difference in space) is lower by five to six orders of magnitude compared with STED. In fact, even light from a compact CW laser of a few inches in size does the job.
“Thus, one obtains a STED-like method that is able to superresolve large fields of view (>100 x 100 µm2) within a second at intensity levels that are ~100 to 1000 times lower than in PALM/STORM and ~10,000 to 100,000 times lower than in STED,” Hell said. “We anticipate this concept to become extremely important in the near future, as it reconciles many of the goals of superresolution imaging.”
For information on lasers for STED microscopy, please see the article on page 30.
STED and the superresolution microscopy family
The birth of superresolution microscopy began in 1994 in a laboratory at the University of Turku, Finland, when Stefan Hell came upon a new theory for feature separation.
Hell realized that nanosized features can be effectively singled out from others residing within their diffraction proximity if the other features are kept dark at the time point of detection. In this case, the diffraction limit of light no longer restricts the resolving power of a microscope.
This principle of keeping feature molecules transiently dark – i.e., nonresponsive to the excitation light – turned out to be very fundamental and hence very general. It is now at the heart of an entire arsenal of superresolution approaches, including STED, RESOLFT, SSIM (saturated structured-illumination microscopy), and PALM, STORM and GSDIM (ground-state depletion imaging).
“If you think about it, you realize that all current methods rely on the same basic principle for feature separation,” Hell said. “The fluorophores of the tiny features in the near neighborhood are forced to stay ‘off.’ ”
Establishing “on-” and “off-” state fluorophores for STED, RESOLFT and SSIM is created by applying a pattern of light, such as a doughnut, arrays of doughnuts or standing waves. As the pattern is scanned across the sample, fluorophores of adjacent features are forced to assume the “on” and “off” states sequentially at controlled positions.
“In contrast, in the stochastic methods (PALM/STORM, GSDIM), the ‘on’ state is assumed molecule by molecule, at random and hence unknown positions in space,” Hell said. “Roughly speaking, only one molecule is allowed to fluoresce within the 200-nm range. The position of fluorescence is then found out using the flux of photons emitted by the individual molecule, allowing a precise centroid-fit, which is called localization.”
In short, STED microscopy locates the feature coordinate by illuminating the sample with photons, whereas in the stochastic methods, this is done with photons leaving individual molecules in the sample.
Published: September 2013