Dr. Gerhard Holst, PCO AG, and Dr. Markus Engstler and Dr. Tim Krüger, University of Würzburg
Researchers seek side-effect-free treatment for African sleeping sickness, and fast imaging technology plays a vital role in the effort.
It’s an age-old epidemic, and it’s terrifyingly deadly: African sleeping sickness, or trypanosomiasis, kills 30,000 people every year, in addition to entire herds of buffalo and cattle. Existing medications are out of date and, worse, they kill not only the pathogens but also the patient – in one case in 10. Biologists at the University of Würzburg have taken on the task of researching leads for the development of better medications. A fast, high-sensitivity camera plays an important part in their research efforts.
Trypanosomes, which are responsible for this tropical disease, live in their host’s blood, a hostile environment to which they manage to adapt perfectly. With corkscrewlike movements, they wriggle through the red blood corpuscles and swim incessantly, always in the same direction. But why do they not fall victim to the antibodies that constantly attack them in large numbers? And why do they swim without interruption, always in the same direction, apparently with no destination? These questions were the starting point for the work of the team of biologists headed by Markus Engstler, chairman of the Department of Cell and Developmental Biology at the University of Würzburg. In 2007, the research led to a hypothesis that caused a sensation among experts.1
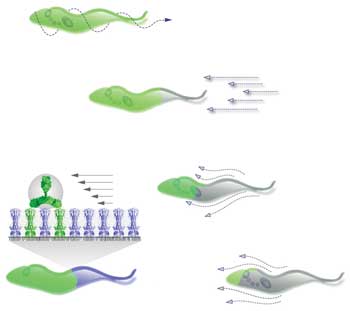
This pattern illustrates the binding of antibodies to the surface of trypanosome and the infuence of the hydrodynamic flow. On the right is the course of an experiment in which the antibodies (green) with directed movement are drawn to the posterior end. Images courtesy of Dr. Gerhard Holst, PCO AG.
The hypothesis was as simple as it was provocative: The pathogens consume the antibodies. According to this theory, the constant swimming fulfills the purpose of pushing the antibodies toward the end of the cell. The motion of the trypanosomes creates a current that spreads over the protozoans’ very smooth surface. If antibodies adhere to this surface, they offer resistance to the current and are pushed back by the “wind,” exactly in the direction of the cell mouth, where they disappear and lose communication with the immune system. The results of the research were initially called into question in a publication by researchers from Los Angeles in 2009. They proposed a mode of swimming that could not be reconciled with the “sailing hypothesis.”
With the help of the pco.edge, a scientific CMOS (sCMOS) camera from PCO of Kelheim, Germany, the biologists set about analyzing the swimming behavior of the flagellates more closely, to substantiate their hypothesis and also to find a new therapeutic approach focusing on the motility of the pathogens.
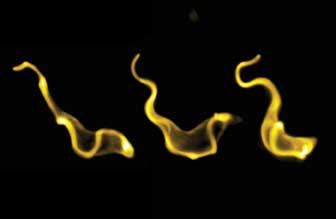
To minimize any external influence on the flagellates, the cell membrane was marked with a fluorescent dye to observe the trypanosomes in a fluid environment on the specimen slide of an inverse digital microscope. The flagellum of the organism beats 20 to 30 times a second; the camera’s imaging capabilities, with 400 frames per second in fluorescence, made it possible for the first time to create high-resolution image sequences of the self-propulsion of the low-fluorescent trypanosomes.

In the “blue series,” single images were compiled from a high-speed series of images taken with the pco.edge. Resolution in the millisecond range enables the images to show the position of the flagellum and of the waves produced on the cell.2
The images confirm the hypothesis first put forth in 2007: that the parasites literally swim for their life. The next step is to use this information to develop a suitable therapy. The goal will be either to slow down the trypanosomes or to reverse their movement – in other words, to make them swim backward. In pursuit of this goal, the Würzburg researchers are now searching for genes that slow the movement of the trypanosomes.
Systematic, large-scale genetic analyses identify the cells involved in the swimming movement. The camera exactly measures the change in swimming behavior; through systematic screening of substance libraries, it will then be attempted to achieve the same specific changes. This approach makes it possible to identify lead compounds in combination with mechanisms of action.
During the screening, the sCMOS cameras detected minimal deviations in the swimming movements. The goal was to change the movements to prevent elimination of the antibodies.
It has already been shown that this principle works. By creating a special RNA in the pathogens, Engstler’s team reversed the motion of the trypanosomes. The result: The pathogens swam backward, and the human antibodies were confronted with the front end of the pathogens instead of the cell mouth. Because the antibodies were not consumed, they could disable the trypanosomes.
This image shows high-resolution 3-D models of the trypanosomes from fluorescence images (taken with a different camera, but also possible with the pco.edge). With the help of high-speed images, Engstler and colleagues were able to correlate the morphology of the cells with the movement mechanism.2
The pco.edge camera was provided to the researchers by the manufacturer before it was available on the market; later, the camera was purchased by the Department of Cell and Developmental Biology. “We would not have been able to record these images with a rival product,” said Engstler, who has worked with PCO on past projects. “The short time intervals between consecutive images of a series make it possible to determine exactly how these flagellates move in three-dimensional space. The light signals correspond at the brightest areas to about 100 photons, which of course is a very weak signal.”
Currently, the cameras are being used on infected animals in Nairobi. “We are trying to re-examine the results obtained in the ivory tower,” Engstler noted.
The main focus is on finding a medication against trypanosomiasis that has no side effects. “African sleeping sickness is a disease of the poorest people in the world, and our results will certainly not be of interest to the major drug manufacturers,” Engstler added. Migration and expulsion have ensured that the disease constantly and unexpectedly flares up at new locations; the target group is economically uninteresting for global pharmaceutical corporations. Doctors Without Borders and other nonprofit organizations have emphasized the need for more research in the field of sleeping sickness, and the researchers in Würzburg and their partner lab in Africa receive additional support from the German Research Institute (DFG).
Meet the authors
Dr. Gerhard Holst works in science and research at PCO AG in Kelheim, Germany; email: [email protected]. Dr. Markus Engstler and Tim Krüger are cell biologists at the University of Würzburg.
References
1. M. Engstler et al (Nov. 2, 2007). Hydrodynamic flow-mediated protein sorting on the cell surface of trypanosomes. Cell, pp. 505-515 (doi: 10.1016/j.cell.2007.08.046).
2. N. Heddergott et al (2012). Trypanosome motion represents an adaptation to the crowded environment of the vertebrate bloodstream. PLOS Pathogens (doi: 10.1371/journal.ppat.1003023).