A new technique that uses light to activate chemotherapy drugs in specific cells shows promise as a way to improve the effectiveness of cancer therapies while preventing severe side effects.
By making existing cancer drugs sensitive to light, this approach could be used to treat a range of tumors with a new level of precision, according to researchers at Ludwig Maximilian University of Munich.
"We hope that our compounds will one day be used in medicine to deliver a killer blow to many types of localized cancer tumors, without producing side effects, thus improving on standards of care and also providing chemotherapy options for currently untreatable tumors," said postdoctoral researcher Oliver Thorn-Seshold.
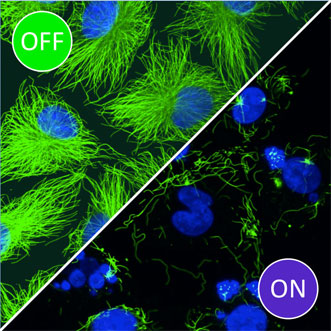
Incubated human cancer cell lines in cell culture, treated with photostatin. Cells kept in the dark, left, are compared with those exposed to brief pulses of blue light, right. Courtesy of Malgorzata Borowiak/Ludwig Maximilian University.
Presently, some of the most successful and widely used chemotherapeutic drugs are inhibitors that interfere with the function of microtubules — cellular components that play a key role in cell proliferation, migration and survival. As these drugs don't specifically target cancer cells, they do interfere with the function of normal cells, leading to side effects such as heart and nerve damage. Consequently, microtubule inhibitors often are limited to low doses, which do not provide optimal therapeutic benefits.
Thorn-Seshold and colleagues have developed a method for optically controlling microtubule inhibitor drugs currently in clinical trials. The strategy involves identifying a fixed structural element, which is required for a drug's biological activity, and then replacing that element with a flexible hinge that swings open or shut in response to blue light.
"We can then use light to switch the hinged drug on and off, where and when we want, with single-cell precision," said professor Dirk Trauner. "The upshot is that our compounds retain the powerful anticancer effects of existing microtubule inhibitors but add the bonus of tissue-specific localization."

The team demonstrated that these modified compounds, called photostatins, are effective in inhibiting the proliferation and survival of cells targeted by light, without affecting neighboring cells. Because they can be switched on and off hundreds of times, photostatins are suitable for long-term applications in both the clinic and laboratory. Additionally, they can be used to study or treat a range of organisms or processes, with the potential to address human diseases, as well.
"We imagine that, since they eliminate systemic side effects, our compounds will be able to be used in medicine at dosages that are truly therapeutic in tumors, thus achieving a much more effective therapy than currently possible," Thorn-Seshold said.
In future clinical settings, patients could wear blue-tinted glasses to treat eye cancer or other eye diseases, while individuals with skin cancer could wear a bandage that delivers light to tumors, the researchers said. Internal tumors might be treated with an implantable network of tiny LEDs that blink every few minutes in order maintain the chemotherapeutic effect only in the enclosed tumor site.
The researchers intend to refine their technique with more precise and effective compounds, while concurrently working to refine the technique for treating tumors in mice. Because the field of photopharmacology is very young, it may take some time for pharmaceutical companies to realize the value of these compounds, Thorn-Seshold said.
"Yet, if our ongoing studies are successful, we will have a convincing proposition for further preclinical development and we are committed to getting as far into real therapy as we can," he said.
The research was published in Cell (doi: 10.1016/j.cell.2015.06.049).
For more information, visit www.uni-muenchen.de.