Ultrahigh-resolution optical coherence tomography (UHR-OCT) enables micron-scale, cross-sectional and 3D-imaging capabilities. Supercontinuum sources offer the necessary optical bandwidths, spatial coherence and optical power densities — all of which are key UHR-OCT parameters. Within the biomedical world, such imaging techniques are relevant for robust tissue analysis.
Thomas Feuchter, NKT Photonics A/S
OCT is a noninvasive imaging technique. In contrast to high-resolution optical microscopy — an invaluable tool for a wide range of applications but one that only permits surface-level or shallow-layer imaging of biological samples — OCT methodologies allow for cross-sectional or 3D imaging.
The use of OCT for imaging was first demonstrated in 1991 by Huang et al.,1 and a thorough description of principles and applications has been provided by Drexler and Fujimoto in “Optical Coherence Tomography: Technology and Applications.”2 Over the past 20 years, OCT has developed into an essential imaging tool in ophthalmology, with particular emphasis on detailed analysis of the retina and the surrounding tissue.
However, OCT applications are not limited to ophthalmology — an increasing amount of research is being carried out using OCT in many other areas of study, such as in the pulmonary, dermatology and cardiovascular fields.
As part of an optical measuring technique, the performance of the light source is crucial for OCT imaging. Supercontinuum sources offer a combination of features — extreme optical bandwidth that allows for selectable center wavelengths, excellent spatial coherence and high optical power density. All of these attributes are important parameters for OCT and enable a multitude of applications. The number of OCT applications using supercontinuum sources (both in research and commercial uses) has increased rapidly, which was well-illustrated at the 2015 SPIE BiOS/Photonics West conferences, where more than 10 presentations discussed research results obtained by using supercontinuum sources for OCT techniques.
This article discusses how supercontinuum sources now are being used for OCT applications, enabling ultrahigh spatial resolution along with a variety of other measurement modalities to reveal detailed supplementary sample data.
OCT basics
OCT is based on white-light interferometry, where light reflected or scattered off of the sample being investigated interferes with light from a reference arm (typically in a Michelson interferometer), as is shown schematically in Figure 1. The light from each arm originates from the same light source, and hence the two beams interfere with each other when the path length difference of the two arms is within the coherence length of the optical signal. This allows the detection system to discriminate reflections from reflectors, which are separated by a distance corresponding to the coherence length of the source, and thus enables high-resolution imaging along the optical axis. Scanning the beam position on the sample in either a line or a 2D raster scan enables cross-sectional or volumetric imaging of reflectors within the sample.
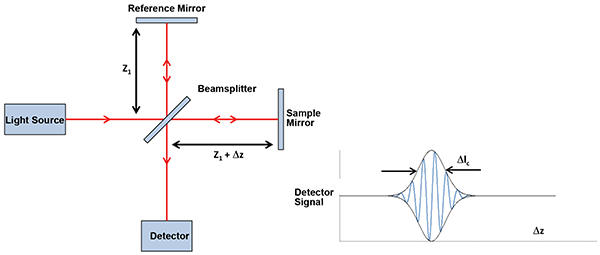
Figure 1. Principle drawing of an OCT setup (left), interference signal on detector (right).
Measurement sensitivity through the use of OCT can be very high, which enables even the weak signals originating from subsurface reflections to be detected. Cross-sectional imaging of the test sample can be performed in a way similar to that used for ultrasound but with a much better resolution and with image depths of several millimeters.
Practical realizations of OCT
There are several practical implementations of OCT.2 The most commonly used are:
• Time-domain OCT (TD-OCT), where the reference mirror is movable and thereby enables coherence gating at
different depth positions in the sample arm. TD-OCT was the first realization of OCT and still remains relevant. For
example, it is used for full-field OCT, where the interference pattern for a full 2D area of a sample is detected simultaneously by a 2D detector array (e.g., a CCD or CMOS sensor).
• Spectral-domain OCT (SD-OCT), which also is known as Fourier-domain OCT (FD-OCT), is used when the reference mirror is in a fixed position and the interference pattern is detected spectrally and subsequently converted to spatial information by the Fourier transformation. SD-OCT can be subdivided into the following:
• Spectrometer-based OCT (Sp-OCT), where a broadband source is used to generate the interference spectrum and thus is detected with a high-speed spectrometer, typically with several thousand pixels and with subnanometer optical resolution.
• Swept-source OCT (SS-OCT), where a tunable source rapidly scans the relevant spectral range. The spectral response of the interferometer is detected by using either a single or a balanced detector.
Supercontinuum sources can be used in all of these realizations — even for SS-OCT via time-stretching the individual supercontinuum pulses in a dispersive fiber to effectively sweep the center wavelength.3 Most supercontinuum sources, though, have been applied to SD-OCT based on Sp-OCT.
Axial resolution of OCT
The axial resolution defines the ability to distinguish between reflectors at different depth positions and is related to the coherence length of the light source.2 In Figure 2, the theoretical value of the coherence length, and hence axial resolution lc, is shown for a Gaussian-shaped spectrum with a center wavelength of λc and a full width half maximum bandwidth of Δλ.
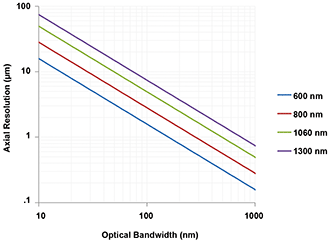
Figure 2. Axial resolution as a function of the spectral bandwidth and the center wavelength of the optical signal shown for center wavelengths of 600, 800, 1060 and 1300 nm.
Figure 2 also shows that the theoretically achievable axial resolution strongly depends on the optical bandwidth and the center wavelength. It is evident that an axial resolution of 1 µm requires an optical bandwidth of 100 nm or more, and that the highest axial resolution is obtained by operating at the shorter center wavelengths.
Supercontinuum sources for OCT
Supercontinuum sources have optical advantages — output spectra are continuous and can cover a very wide optical range from less than 400 nm and up to 2400 nm. This allows the center wavelength to be selected to match the requirements of the application. Furthermore, the typical output power density of 0.5 to 5 mW/nm is significantly higher than alternatives like superluminescent diodes (SLEDs).
In addition to the optical parameters of supercontinuum sources, NKT Photonics’ SuperK technology offers several very useful features like portability, high reliability, maintenance-free operation and instant-on operation with no need for warm-up or careful alignment. These features are beneficial in daily work and become increasingly important as the applications move from optical laboratories and into medical labs and clinics.
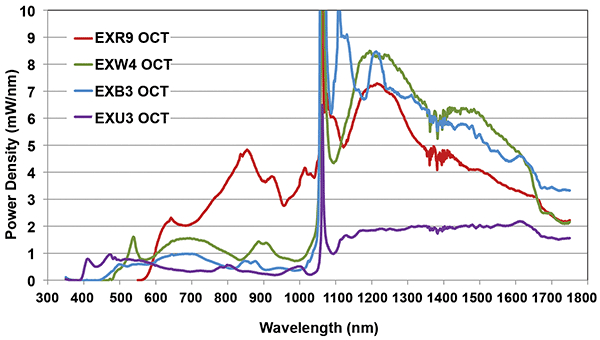
Figure 3. Spectral power density of customized versions of NKT Photonics’ SuperK, optimized for OCT.
Noise properties
The quality of an OCT image is highly affected by the signal-to-noise ratio (SNR) of the detected interference signal, which limits the lower level of reflections that can be detected and imaged. This is particularly important for samples with low reflections, such as tissue samples. The noise level of the detected signal depends on the detector noise (i.e., read noise and thermal noise), the shot noise originating from the finite number of photons detected, as well as the relative intensity noise (RIN) originating from the light source.
Supercontinuum generation of the large optical bandwidth is a partly stochastic process, causing increased RIN compared with, for example, SLED sources. However, the development of highly stable optical seed lasers and RIN-optimized nonlinear fibers for supercontinuum generation has resulted in a reduction of the RIN noise to a level where it is lower or similar to the other noise sources of the system. Furthermore, high-output power density enables the ability to measure reflections from very weak reflectors.
The combined effect of the improved RIN properties and signal power results in sensitivities and SNR values that are comparable to those of OCT systems based on other light sources, but with the added increase in axial resolution and the free choice of a center wavelength within the available wavelength range. The noise performance of different supercontinuum sources has been evaluated and, compared with that of SLEDs,4 shows the improvement of SNR obtained with SuperK technology. As for any OCT system, though, the impact of RIN depends on the realization of the system — particularly on the optical and electronic bandwidths of the detection system — and thus must be evaluated independently for each configuration.
UHR-OCT
UHR-OCT allows for the visualization and analysis of living tissue on a micron scale, which can reveal essential information about the progression of diseases, and for the evaluation of the beneficial effects of potential treatments. Two recent examples of applications that require micron-size axial resolutions are: 1) The evaluation of pulmonary diseases, such as cystic fibrosis,5,6 where the oscillatory motion of the micron-sized cilia (marked with “a” in Figure 4a-b) is monitored and quantified, and 2) The interaction between macrophages and cholesterol crystals that may act as precursors for atherosclerosis in the human coronary artery.7
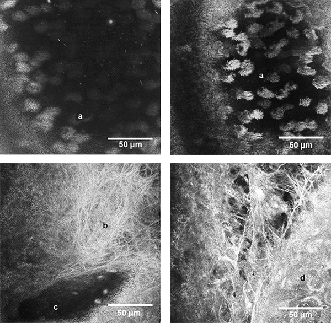
Figure 4. En-face images of mouse trachea at different depths showing cilia (a), fibrous structure (b), vessel (c) and single cell (d). Courtesy of Gereon Hüttmann et al./BMO Lübeck.
Researchers at Johns Hopkins University, located in Baltimore, are investigating UHR-OCT in a variety of implementations using different light sources.8 As shown in Figure 5, they have investigated the esophagus of a guinea pig by endoscopic OCT using the described technology, thereby enabling in vivo analysis of the detailed structures.
UHR-OCT also can be applied in a variety of nonmedical applications, especially within nondestructive analysis and testing where the detailed information on subsurface features is evaluated. One application is for analyzing the layers of paint on old master paintings,9 where the individual layers of paint can be measured and visualized.

Figure 5. Endoscopic OCT imaging of a guinea pig esophagus in vivo (SC: Stratum Corneum; EP: Epithelium; LP: Lamina Propria; MM: Muscularis Mucosae; SM: Submucosae; MP: Muscularis Propria). Courtesy of Scott W. Yuan, Jessica Mavadia and Xingde Li/Johns Hopkins University.
Combining OCT with other modalitiesV
Large optical bandwidth combined with high optical power density makes the described technology suitable for systems that implement UHR-OCT together with other measurement techniques within a single setup, also known as multimodal imaging techniques.
Other functionalities may use the same optical band as used for OCT, providing additional data other than the purely morphological information. Such is the case for spectroscopic OCT, where the different spectral components of the SD-OCT signal are analyzed independently and can provide spectral information on, for example, the absorption or reflection properties of the sample. One such application has been reported by Chong,10 where the blood vessels in the retina of a rat were analyzed using OCT in the wavelength range of 500 to 650 nm. Spectroscopic analysis of OCT data enabled estimation of both the hemoglobin concentration as well as the oxygen saturation in these vessels. Similarly, researchers at Northwestern University in Evanston, Ill., have demonstrated noninvasive measurements of the oxygen saturation in a mouse ear by using OCT in the wavelength range of 520 to 630 nm in combination with speckle contrast analysis.11
The technology’s wide, continuous spectrum also can be split into subbands, from which one spectral band can be used for OCT, and the other spectral bands can be used for other measurement modalities. This enables simultaneous detection of various properties of the sample being investigated. One example of this multifunctional imaging modality is the combined measurement of the morphology by OCT at 480 nm and the autofluorescence detection of lypofuscin in the retina by implementing highly sensitive autofluorescence microscopy at 515 nm, as was recently demonstrated by Jiang.12 Combined with the autofluorescence imaging of rhodopsin — the light-sensitive protein in the eye — as reported by Liu,13 such techniques are likely to become important tools in ophthalmology and other fields.
The outlook for OCT
Broadband light sources are becoming increasingly important, not only for realizing UHR-OCT but also for combining different imaging and measurement techniques. The performance of the described sources for these demanding applications has been successfully demonstrated in a number of different applications.
The performance, ease of operation and reliability of these sources are important features that facilitate the transition from research labs into commercial applications. Combined with the large number of applications and functions enabled by the large optical power and bandwidth, these sources are well-suited to serve as key components in various optical instruments for medical and nonmedical applications.
Meet the author
Dr. Thomas Feuchter, PhD, is senior research scientist and OCT product manager at NKT Photonics A/S in Birkerød, Denmark; email: [email protected].
References
1. D. Huang et al. (1991). Science. Vol. 254, No. 5035, pp. 1178-1181.
2. (2008). Optical Coherence Tomography: Technology and Applications. W. Drexler and J. Fujimoto, eds. Springer Berlin Heidelberg.
3. K. Goda et al. (August 2012). High-throughput optical coherence tomography at 800 nm. Opt Express, Vol. 20, Issue 18, pp. 19612-19617.
4. W.J. Brown et al. (2014). Noise characterization of supercontinuum sources for low-coherence interferometry applications. J Opt Soc Am A, Vol. 31, Issue 12, pp. 2703-2710.
5. H. Schulz-Hildebrandt et al. (2015). Fast volumetric imaging with subcellular resolution by Fourier domain optical coherence microscopy with numerical refocusing. Presented at SPIE Photonics West BIOS.
6. L. Liu et al. (January 2013). Method for quantitative study of airway functional microanatomy using micro-optical coherence tomography. PLOS ONE, Vol. 8, Issue 1.
7. M. Kashiwagi et al. (July 2014). Feasibility of the assessment of cholesterol crystals in human macrophages using micro optical coherence tomography, PLOS ONE, Vol. 9, Issue 7, e102669.
8. J. Xi et al. (2014). Diffractive catheter for ultrahigh-resolution spectral-domain volumetric OCT imaging. Opt Lett, Vol. 39, Issue 7, pp. 2016-2019.
9. C.S. Cheung et al. (2015). Ultra-high resolution Fourier domain optical coherence tomography for old master paintings. Opt Express, Vol. 23, Issue 8, pp. 10145-10157.
10. S.P. Chong et al. (2015). Quantitative microvascular hemoglobin mapping using visible light spectroscopic optical coherence tomography. Biomed Opt Express, Vol. 6, Issue 4, pp. 1429-1450.
11. Ji Yi et al. (2014). In vivo functional microangiography by visible-light optical coherence tomography. Biomed Opt Express, Vol. 5, pp. 3603-3612.
12. M. Jiang et al. (2014). Simultaneous optical coherence tomography and lipofuscin autofluorescence imaging of the retina with a single broadband light source at 480nm. Biomed Opt Express, Vol. 5, pp. 4242-4248.
13. T. Liu et al. (2015). In vivo imaging rhodopsin distribution in the photoreceptors with nano-second pulsed scanning laser ophthalmoscopy. Quant Imaging Med Surg, Vol. 5, Issue 1, pp. 63-68.