From microscopy of ocular diseases to imaging in neurosurgery to tomography in patients with skin cancer and brain tumors, fluorescence lifetime imaging microscopy is the versatile partner that can be teamed with a host of other imaging techniques to produce striking results.
Fluorescence lifetime imaging microscopy (FLIM) emerged in the late 1980s with the development of pulsed lasers. But it was not until the 1990s when, thanks to the introduction of two-photon femtosecond laser scanning microscopes, FLIM gained significant popularity.
Today, FLIM has become an important add-on tool for fluorescence microscopy. It is favored for binding studies and molecule characterizations, occupying a secure position in both life and materials science for more than a decade.
Human skin, stained with pH-sensitive dye. Fluorescence lifetime represents pH. Courtesy of Wolfgang Becker, Becker & Hickl, Germany.
“The term FLIM is defined as fluorescence lifetime imaging [microscopy] where the fluorescence lifetime is depicted with spatial resolution. FLIM images are typically generated with microscopes and nanoscopes, endoscopes and tomographs, but can be also acquired on a macroscopic level,” said Karsten Koenig, professor at Saarland University in Saarbrücken, Germany, and CEO of the laser medicine company JenLab GmbH in Jena, Germany.
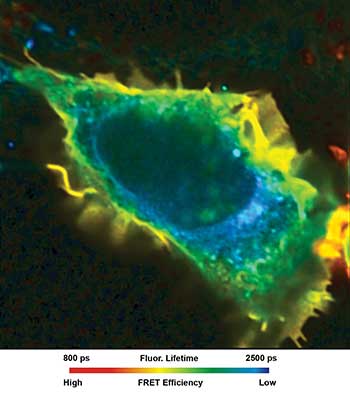
By detecting either intrinsic or extrinsic fluorophores in a specific nano- or micro-environment, FLIM can be used to probe intermolecular interactions within a 10-nm distance, such as bindings, by Förster resonance energy transfer (FRET) and anisotropy measurements. FLIM is also applied in animal studies, artwork, microfluidics and in forensic science.

In vivo 4 × 3-mm² wide-field image on human skin (forehead). (Green: autofluorescence; Red: SHG). Courtesy of JenLab GmbH.
“FLIM images are often false-color coded where the colors reflect values. FLIM images can be taken in the frequency mode (measurement of phase shift and demodulation with a continuous or pulsed laser), as well as in the time mode using time-gated cameras, streak cameras and time-correlated single photon counting (TCSPC) units,” Koenig said.
In fact, it is TCSPC-based FLIM that is currently being used to explore one of the most challenging and exciting applications: in vivo clinical imaging of patients. In this approach, one-photon excitation comes courtesy of picosecond laser diodes in the visible spectral range while two-photon excitation comes from the application of near-infrared femtosecond Ti:sapphire lasers.
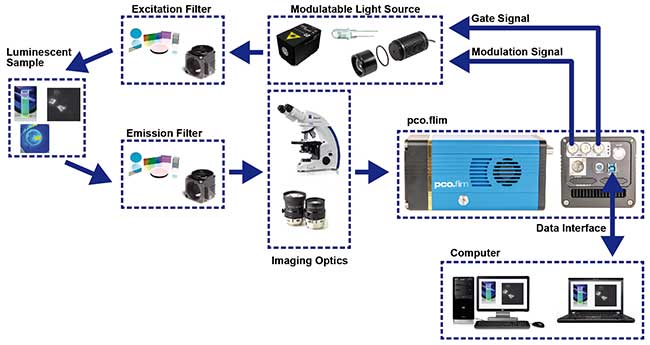
Structural overview of a suggested setup for luminescence lifetime imaging using a PCO FLIM camera system. Courtesy of PCO AG, Kelheim.
Thanks to the focus being on measuring the fluorescent lifetime of a fluorophore (the time a fluorophore stays in the excited state), rather than its intensity, FLIM is independent of a whole host of factors that otherwise afflict traditional fluorescence microscopy techniques.
“It is independent of experiment parameters, such as illumination intensity, light path length, detection efficiency, scatter and photobleaching,” said Klaus Weisshart, product manager at Carl Zeiss Microscopy GmbH in Jena, Germany. “It is thus able to provide contrast between different fractions of the same fluorescent species in different molecular environment[s], or between fluorescence species with identical or strongly overlapping emission spectra.”
As well as being independent of internal instrument settings, such as laser intensity or detection efficiency, FLIM allows many types of fluorescent markers to be used to assess different processes simultaneously.
When it comes to data analysis, any background emission due to autofluorescence from tissue can be easily removed by using the fluorescence lifetime information. This results in higher detection efficiency and more accurate marker localization.
“In biological systems like cells, the fluorophore concentration often cannot be accurately determined and compared amongst different cells,” said Sandra Orthaus-Müller, technical sales and support specialist for microscopy at PicoQuant GmbH in Berlin. “Thus, FLIM allows [users] to compare results from different cells without numerous control/calibration measurements and to quantify the data (most important for detection of protein interactions).”
But for all of its advantages, there are challenges that imaging specialists are working hard to overcome. One of the main limitations is the elaborate equipment that has to be used; there has to be supplementary electronics: a fast photodetector that is able to count single photons and additional analysis software.
What is more, TCSPC is hard to apply with high imaging speed, and quantifying lifetime using an intensity measuring system such as a camera is tricky, resulting in data that is complicated to interpret.
FLIM microscopes, tomographs and endoscopes remain expensive and relatively bulky, limiting their adoption to a specialized market. But with the availability of ultracompact, low-cost femtosecond laser sources within the next few years, Koenig predicts that FLIM tools will become increasingly available and clinical applications will begin to emerge.
While medical FLIM is still considered early stage, image resolution has dramatically increased over the last decade. FLIM in the time domain is getting faster and therefore proving a more popular method compared with fast frequency domain FLIM.
“Large tissue areas or a large number of cells can now be imaged in the same experiment, and under exactly identical experimental conditions,” said Wolfgang Becker, co-founder of Becker & Hickl GmbH in Germany. “FLIM has been combined with STED to obtain superresolution data of cellular structures. The ability to record large amounts of data has led to techniques that record temporal variations of molecular parameters, at millisecond resolution.”

Endogenous fluorescence of pig skin. Courtesy of Wolfgang Becker, Becker & Hickl, Germany.
When it comes to clinical applications, FLIM can be used to study the molecular environment within cellular compartments or changes thereof including ion concentration, pH, oxygen and membrane polarity. It also is a powerful method to study interaction or conformation of proteins by using FRET techniques, or to probe the positions of molecules within a macromolecular complex.
An important class of applications takes advantage of FLIM’s ability to derive metabolic information from the fluorescence of endogenous fluorophores, such as nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotide (FAD). Measuring NADH in its free and bound form provides information on oxidative stress, which is of interest to the cosmetic and pharmaceutical industries.
In fact, JenLab’s DermaInspect and MPTflex is currently being used to measure the efficacy of anti-aging drugs to fight against oxidative stress in skin and to trace components in situ in deep tissue.
Cancer research is also a hot topic; previous studies have shown that the fluorescence lifetime changes the more a cell becomes dedifferentiated in the course of cancer progression. Therefore, FLIM might be involved in diagnostics.
Instrument advances
Improvements in detectors are among the most important technological advances. For example, the development of sCMOS (scientific CMOS) image sensors with a charge-swing per pixel allows for a fast demodulation up to 40 MHz per pixel.
“This enabled us to develop a CMOS camera with 1008 × 1008 pixels with which we can measure pixelwise phase angles, modulation index and intensity to convert this with a proper reference measurement into fluorescence lifetime distributions,” said Gerhard Holst, who heads up the science and research department at scientific camera specialist PCO AG, based in Kelheim, Germany. “The FLIM measurement was possible before, but not with the same frame rate at 1 MP of 22 frames/s.”

The pco.flim camera system. Courtesy of PCO AG, Kelheim.
Holst believes that key to success is making FLIM easier to apply by nonexperts. “If the market is interested and the companies see economic success, it’ll be likely that they continue to improve the special CMOS image sensors,” he said. “Further, it would be nice to have a better sensitivity (higher quantum efficiency) and maybe faster modulation capabilities.”
At Leica Microsystems GmbH, the Leica HyD SMD is a sensitive and rapid detector for single molecule applications. It is fully integrated into the spectral detection system of the Leica TCS SP8 confocal microscope, and therefore, FLIM experiments can be performed spectrally and the detection range can be optimally adapted to the fluorescence properties of the fluorophore. In combination with a supercontinuum laser source, which can be tuned in the entire visible spectrum, a microscope can be adapted to the sample, and excitation and emission can be optimized for the imaged dyes.
“In addition, it is fully integrated into the spectral detection system of the Leica TCS SP8,” said Irmtraud Steinmetz, application manager for the life science division at Leica Microsystems in Mannheim, Germany. “Thus, FLIM experiments can be performed spectrally and the detection range can be optimally adapted to the fluorescence properties of the fluorophore.”
At PicoQuant, complete and turnkey upgrade kits are available for standard laser scanning microsopes (LSMs), making this method accessible to a broader scientific audience. Another interesting development lies in the combination of FLIM with other characterization methods, such as spectral, dynamic or even topological information from atomic force microscopy (AFM).
Such combined measurement methods open promising prospects by accessing different information types from the same sample space in a single experiment.
“For example, acquiring topological information from AFM and molecular behavior as detected by FLIM was previously limited to correlative experiments, requiring large amounts of statistics especially for heterogeneous biological samples,” said PicoQuant’s Orthaus-Müller. “With a combined FLIM-AFM set-up, as can be realized by interfacing the MicroTime 200 microscope to an AFM, the data are acquired in a simultaneous and correlated manner.”
Extending the spectral range of FLIM into the deep ultraviolet is also of great interest since many biological molecules feature naturally occurring chromophoric groups, such as tyrosine or tryptophane.
“By using UV lasers and adequate optical elements, one can exploit this native fluorescence, reducing the need to label such molecules with dyes,” Orthaus-Müller said. “In contrast to intensity imaging, FLIM enables [users] to differentiate the (auto)fluorescence in UV spectral range based on its different contributions.”
With further improvement in instrumentation, along with advances in techniques, methods and fluorescent labels (including new types of fluorescent proteins), there will certainly be new discoveries in cell biology in FLIM’s future.