Matthias Schulze and Wallace Latimer, Coherent Inc.
A comprehensive analysis reveals that lasers provide both superior performance and lower total cost in comparison with LEDs for life sciences instrumentation.
Biotech instrumentation manufacturers are currently developing products that feature increased miniaturization and lower costs per use, in part to support worldwide trends in health care. For instruments based on fluorescence, this has opened a debate as to whether the laser or the LED represents the best choice for a light source. The superior performance of lasers is well known, but it may not be obvious that lasers nearly always also represent a lower-cost option.
True cost
Many instruments that analyze biochemical samples and reactions use fluorescent probes. Examples include devices with applications in cytometry, genetic sequencing, hematology, polymerase chain reaction, high-throughput drug screening and microarray scanners. Several trends, in both the Western and Chinese markets, are driving a need for miniaturization, automation and greater specificity; this includes developing compact, dedicated instruments configured for simplified and repetitive use in common tests, in contrast to the multifaceted flexibility of a typical flow cytometer.
The key to successful operation in all these instruments is the ability to maximize signal-to-noise ratio. This, in turn, is accomplished by delivering the necessary amount of usable excitation light (that is, light that actually excites the fluorescent probe) into the required illumination sample region, while minimizing the amount of light that ends up lost or focused outside of this region. For current commercial instruments, these target spot sizes can range from tens of microns in diameter to several millimeters in size (Table 1).
In general, the unique optical characteristics of the laser, particularly its spectral and spatial brightness, make it a much more efficient source for accomplishing this task than the LED, especially at smaller focused spot sizes. So, although the LED source itself might be much less costly than a laser, all the ancillary measures that must be employed to correct for its inherent output deficiencies can end up making the total system cost greater than that of a laser-based instrument.
Output power vs. rated power
One important difference between lasers and LEDs is not in how they operate, but in how their output is traditionally specified. Lasers, originally developed by the photonics community, have always been specified by their output power. Simply stated, a 1-W laser delivers 1 W of light output.

The BioRay series, a new generation of economical continuous-wave visible lasers, was developed with output parameters tailored specifically to meet current and future trends in cost-sensitive biotech instrumentation.
In contrast, LEDs originated in the electronics industry and are specified by their power consumption. Since even the best LEDs rarely surpass 10 percent efficiency, a 1-W LED usually outputs 100 mW or less of light. Moreover, the maximum current and voltage specifications often apply to pulsed operation. Thus, a “1-W” LED might be continuously operable only at 750 mW or less, which translates into an optical output of just 75 mW. So, from this factor alone, we need to correct the raw cost per watt by a factor >10 in any comparison between LED and laser.
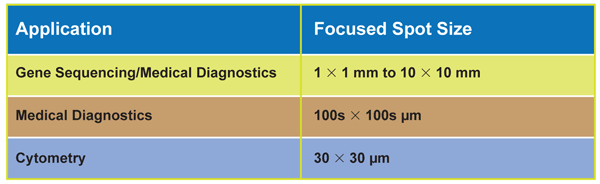
Probably the most significant inherent difference between lasers and LEDs is in the area of light collection and focusing efficiency. The output of a laser behaves as an ideal, true point source, meaning that virtually all the light can be collected and focused into the precise spot size needed by the application. Similarly, a laser beam can be efficiently coupled into a single-mode fiber (whose output can, in turn, be focused to a small, well-behaved spot).
On the other hand, an LED is a conventional extended light source, like a lightbulb. The ability to focus an extended source is defined by the size of the emitting surface area and the solid angle into which it radiates in a property called etendue. The important thing about etendue is that, in the best-case scenario, it is preserved across an optical system (and in a real-world optical system, it always gets a bit worse).

So, for a system with a perfect (Lambertian) emitting source (Equation 1), where the collecting numerical aperture (NA) is the smaller of either the source’s emission angle (i.e., source NA) or acceptance angle of the collecting optics (i.e., lens NA). Since real LEDs are never perfect Lambertian sources, this equation represents a theoretical best case. When the light is refocused, then Equation 2 applies.

To see the impact of etendue, let’s consider the case of a 100-mW LED with an active area of 1 mm2; that emits into a cone of π steradians corresponding to an NA = 0.5. If we wish to focus this output into a spot diameter of 100 µm, Equations 3-5 apply.

A well-corrected focusing lens with NA = 0.3 would already represent a fairly sophisticated and expensive optical system. (And the even higher cost of an increased NA would probably be prohibitive for clinical or point-of-care instruments.) In this instance, the highest theoretically possible NA on the focusing side is a value of 0.03. This means that only about 2 percent (2 mW) of the LED output is focused into the desired 100-µm spot (using NA ≡ n × sin θ) – i.e., <0.02 percent of the LED’s rated power, compared with >90 percent of the laser’s rated power. So now there is a 5000× factor in cost per watt, just for the source. The instrument builder also has to factor in the much higher cost of using corrected high-NA optics with the LED. And still the LED can’t deliver the focused intensity of a laser.
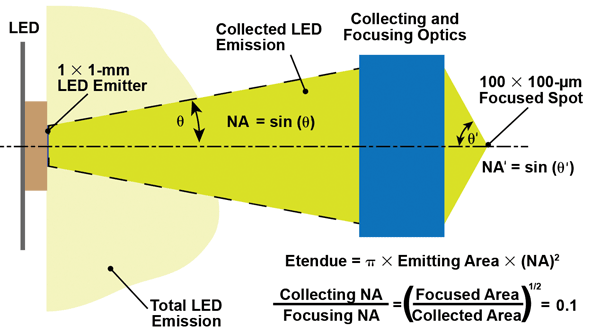
Etendue dictates that if a source is imaged at a magnification of 0.1×,
the collection NA must be 0.1 times the focusing NA. Since the highest
possible focusing NA is 1, the ability to collect all the output from
the source LED is severely limited.
This relatively low focused intensity can’t be avoided by combining multiple LEDs; this increases source size even further, making the problem even worse. What would help is brighter LEDs – that is, LEDs that produce greater light out-put without an increase in emitter size. At present, typical LED brightness is increasing by only 10 percent per year in response to the marketplace. And because most LEDs go into illumination or display applications that are many, many orders of magnitude larger than the bioinstrumentation market, biotech has no leverage whatsoever to change this situation. In contrast, laser manufacturers have long actively pursued the bioinstrumentation market with lasers offering a broad range of performance and costs.
Monochromatic lasers vs. broadband LEDs
Another major difference between lasers and LEDs is that the laser is a monochromatic source, whereas an LED emits over a much wider spectral range. With a laser, the only requirement is that the wavelength be somewhere near the broad excitation maximum for the target fluorochrome. As indicated in Figure 2, a longpass filter between the interaction zone and the photodetector effectively blocks all scattered laser light and ensures that the photodetector only “sees” the fluorescence, although it is far dimmer than the laser excitation. Thus, with a
laser, it is easy to get a high signal-to-noise ratio in the detection system.
But with an LED, the output has a broad spectrum with a long wavelength tail that usually overlaps with the fluorescence-emission spectrum. Moreover, the intensity of any scattered light in this overlap region can exceed the intensity of fluorescence, which can easily be as weak as 0.1 percent or less of the absorbed light. To minimize this problem, a shortpass filter must be included to block the long wavelength tail of these unwanted photons. The end result is higher system costs, due to the extra optic, and a reduction in signal-to-noise ratio because some of the LED’s output will be blocked by this optic – the direct cost of the useful LED photons gets pushed even higher. Also, the shape of the emission spectrum from LEDs of different batches (“bins”) can vary greatly, which generally requires changes in filter specifications. These discrepancies are beyond the control of the instrument manufacturer and even the LED vendor, meaning instrument builders need to buy a reel (typically thousands of identical LEDs) to cover the entire anticipated lifetime of any instrument, adding yet more indirect costs of using LEDs instead of lasers.
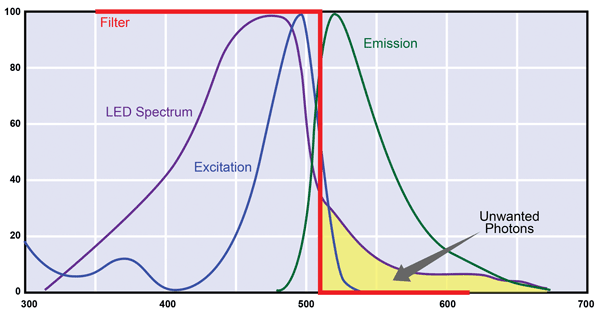
Every fluorophore is characterized by an excitation spectrum and a long-wavelength emission spectrum. With a single-wavelength laser, it is easy to separate scattered excitation light from the emission spectrum using a long-wave cutoff filter. But with a typical LED, the long wavelength tail of its output overlaps the fluorescence emission and must somehow be eliminated. Also, the size and shape of this tail can vary significantly among batches of LEDs.
The broadband output of LEDs is even more problematic in instruments targeting multiple fluorochromes. Extra front-end filters have to be carefully matched to both the excitation/emission spectra of the fluorochromes and the output spectrum of the LED(s), lowering optical efficiency still further.
Detector costs
An LED’s low focused intensity means that the signal will be correspondingly lower, compared to the use of a laser, particularly in applications with a small illuminated interaction zone. Achieving target speed and/or signal-to-noise performance may therefore dictate using a photodetector with higher gain and/or lower noise. For example, a particular instrument may require only a zero-gain photodiode or photodiode array when based on laser excitation, but may need a photomultiplier tube or a CCD array when used with an LED source. And where a laser-based instrument uses a room-temperature CCD array, an LED version of the same instrument might need an actively cooled CCD array.
Streamlined lasers for next-generation instruments
To maximize both the performance and cost advantages, manufacturers have developed streamlined lasers for the bioinstrumentation market – lasers that emphasize economy, compact packaging and simple integration over bleeding-edge performance. For example, BioRay lasers from Coherent deliver up to 50 mW to match the typical low-power needs of many fluorescence instrument types.
They are available at several visible wavelengths (such as 405, 450, 488, 520 and 640 nm) that match the optimum excitation of common fluorescent probes and genetically encoded markers.
Importantly, these application-specific products are based on laser diode technology, the simplest and lowest-cost method of generating continuous-wave laser output at these wavelengths and in this power range. Laser diodes also offer the highest efficiency, lowering the required power budget for the final instrument. Since edge-emitting laser diodes emit a highly divergent and asymmetric (elliptical) beam, optics are used in the laser head to produce a collimated, elliptical beam. To reduce the complexity of downstream beam-delivery optics, each head also has an adjustable output lens to enable smooth adjustment of the beam waist location. To further simplify integration of these compact lasers, the control electronics are miniaturized and incorporated within the laser head. All they require is a 24-V DC power source. Moreover, their closed-loop power stability minimizes the need for instrument recalibration, and their low noise output (<0.5% rms from 20 Hz to 20 MHz) delivers higher accuracy measurements at low intensities.
There’s certainly no question that it’s cheaper to buy an LED than a laser. But, when the total cost of design, complete bill of materials, service strategy and true cost of ownership are considered, lasers can be a more cost-effective option than LEDs for bioinstrumentation. This is even true for next-generation point-of-care systems, where component costs are paramount.
Meet the authors
Matthias Schulze is director of marketing for OEM components and instrumentation at Coherent Inc.; email: [email protected]. Wallace Latimer is product line manager for machine vision at Coherent in Wilsonville, Ore.; email: [email protected].