Dr. David Zhang, Brimrose Corp.
Covert IR taggants represent a strong photonic tool in identifying key personnel and materials in both military and civilian applications.
Covert infrared taggants are embedded markers that can emit coded optical signals outside of the visual spectrum. Their main purpose is to track and identify. They can find multiple uses in commercial, homeland security and military applications, such as developing authentication measures against counterfeiting goods and document fraud, tracking explosives and radioactive materials, and in locating and identifying personnel and assets.
Optical taggants take many forms. They can be fully integrated into marked objects in the form of paints, coatings, fillers and fibers; they can also be used as stand-alone devices such as patches and buttons. The types of covert optical taggants are generally determined by three major factors: the nature of the optical materials/devices, the detection technique and the application platform.
Materials and devices
Certain materials can emit IR light through chemiluminescence (the emission of light as the result of a chemical reaction), photoluminescence (light emission that occurs following the absorption of photons) or electroluminescence (which takes place when a material emits light in response to an electric current or field).
The three general groups of IR-emitting materials are IR-emitting organic dyes, lanthanide IR emitters and IR-emitting quantum dots.
IR-emitting organic dyes: Many pure organic dyes have been developed, especially for NIR biomolecular imaging. The use of NIR fluorophores will eliminate background noise caused by the auto-fluorescence of biosubstrates. Common organic NIR dyes include cyanine, oxazine and rhodamine dyes. The fluorescence maxima of these dyes are between 700 and 850 nm.
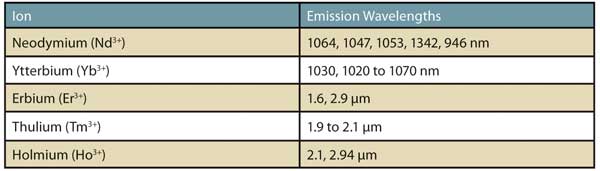
Figure 1. IR emission wavelengths of lanthanide ions.
Lanthanide IR emitters: Organic dyes with fluorescence maxima extending to the far-NIR and into the short-wave IR (SWIR) can be achieved by the formation of metal ion complexes. The most notable group of metals whose ions are capable of narrowband IR emission is the lanthanide series with atomic numbers between 57 and 71 (lanthanum to lutetium). The photoluminescence of lanthanide cations is due to their abundance of 4f–4f and 4f–5d transitions. Well-known IR emission wavelengths from lanthanide ions are listed in Figure 1.
It is also well-known that several trivalent lanthanide ions possess possible emission transitions in the mid-wave IR (MWIR) spectral region.1-7 Figure 2 shows several of the rare-earth ions with potential MWIR (3 to 5 µm) emission transitions.
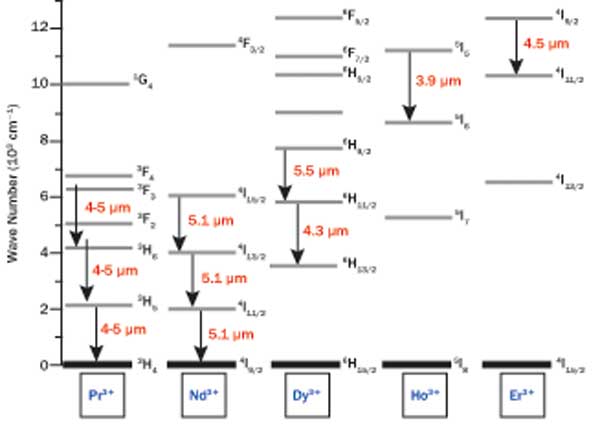
Figure 2. Rare-earth ions with possible MWIR (3-5 µm) emission transitions.
Lanthanide IR phosphors can also be hosted in inorganic matrices, including fluoride and oxyfluoride optical glasses such as NaYF, SiO2-Al2O3-NaF-YF3, and in oxide glass/ceramics, including SiO2, ZrO2, Y2O3 and Y3Al5O12 (yttrium aluminum garnet, or YAG). They are generally optically transparent, especially in the IR spectral region.
IR-emitting quantum dots: Quantum confinement occurs when the size of a semiconductor crystallite is reduced below its exciton Bohr radius. These small semiconductor crystallites, commonly referred to as quantum dots, have properties between those of bulk materials and molecules. For smaller quantum dots (usually below 10 nm), quantum confinement dominates the electronic properties, leading to highly discrete energy levels.
In this region, the bandgap and splitting of energy levels are highly dependent on the size and shape of the quantum dot; in general, the bandgap is inversely related to size. Therefore, the electronic and optical properties of quantum dots can be easily tuned by varying the size during synthesis. The high versatility of quantum dots for wide-spectrum IR emission is also due to the availability of many types of narrow-bandgap semiconductors, with bulk bandgap energies spanning from negative to close to 1 eV with potential for IR emission (1 µm => 1.24 eV) (Figure 3).

Figure 3. Bulk bandgap energies of narrow-bandgap semiconductors and approximate bandgap ranges for ternary alloys.
Compared with quantum well structures grown either with molecular beam epitaxy or chemical vapor deposition, colloidal synthesized semiconductor quantum dots are much cheaper to make. This low cost factor, combined with the capability of highly efficient, narrow-wavelength photon emissions spanning a broad spectrum, means that quantum dots have great promise for revolutionizing many optical applications, including photovoltaic cells, LEDs, laser diodes, medical imaging and quantum computing.
Active IR-emitting devices
Passive IR taggants with embedded dyes/pigments require outside stimulation such as photoluminescence, while self-activating taggants can be based on chemiluminescence or electroluminescence. Chemiluminescence is the mechanism for light sticks in both the visible and NIR regions.
Electroluminescence is probably the most efficient way to produce light from an IR emitter. LEDs and laser diodes are typical electroluminescence devices. Both are commercially available for IR light emission at wavelengths from the NIR to MWIR (1 to 5 µm).
LEDs and laser diodes both have advantages and disadvantages. Compared with LEDs, laser diodes produce much narrower band emissions, but they may require cooling features such as heat sinks, especially when operating at high-energy densities, while no cooling is needed for LEDs. Light emissions from LEDs and, especially, laser diodes are highly directional. This not only raises concerns about eye safety, but also limits the effectiveness for large-area illumination. Therefore, light-scattering/
-diffusing media are often integrated with LEDs and laser diodes. Examples of these optical media include side-emitting fibers/strips and light-scattering lenses/coatings.
Taggant encoding, detection
Optical taggant emissions can be excited with various light sources, including lasers, LEDs, lamps and sunlight. The encoding of optical signals generated by taggant materials is a crucial step for achieving high levels of encryption against reverse engineering and producing a rich collection of distinct markers. Basic encoding techniques can rely on the manipulation of the population makeup of emitter mixtures, emitter-host material relation/interaction, taggant excitation methods, the choice of detectors and detection techniques, and the use of logical analytical tools such as computers.
For taggant markup manipulation, materials with different emissions and excitation wavelengths can be blended into multiwavelength compound emitters or strategically distributed into patterns/layers to produce sophisticated signals and codes that can be fully identifiable only to taggant designers/manufacturers. Photon emission from one component of a compound taggant system can also excite other components of the same system. A simplified example of the compound taggant system can be illustrated by the excitation/emission behaviors of a two-component taggant mixture, Nd-doped ZrO2 and Er-doped ZrO2, as shown in Figure 4.
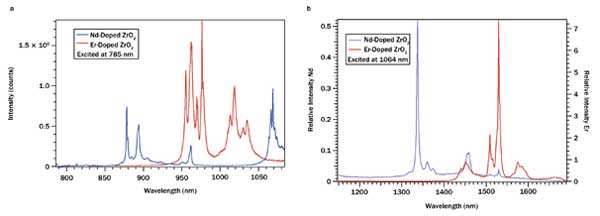
Figure 4. (a) Photon emissions of Nd- and Er-doped ZrO2 excited at 785 nm. (b) Photon emissions of Nd- and Er-doped ZrO2 excited at 1064 nm.
The effect of matrix materials on taggant emission and performance can also be used to benefit taggant system design. Normally, a fully inert matrix such as an IR-transparent ceramic or polymer is preferred so as not to interfere with taggant excitation/emission.
However, materials with simple and specific IR absorption peaks, such as polyethylene, can also be used as taggant matrices or top coatings to purposely block certain taggant emission wavelengths to add additional layers of sophistication. Certain optical dyes, such as IR-transparent UV absorbers, can also be incorporated into matrix/coating materials to prevent the excitation of taggants with commonly available light sources such as UV lamps.
Taggants based on electrically powered emitters such as LEDs and laser diodes can be encoded with sequenced flashing algorithms to achieve stroboscopic effect. Electroluminescence and photoluminescence effects may also be incorporated into flexible/printable electronic substrates/circuits for optical taggant applications.
The design of taggant emission spectra can also be tailored for specific detector/imager systems. Most IR detectors/imagers are only effective with certain wavelength ranges. Therefore, taggant systems emitting signals that cannot be fully detected with a single type of detector are more covert.
Hyperspectral tunable optical filters, such as acousto-optic tunable filters (AOTFs), can greatly enhance an IR detector’s capability for detecting and deconvoluting complex optical signals such as clustered emission peaks. In an AOTF, a piezoelectric transducer bonded to a crystal of tellurium dioxide or quartz generates high-frequency acoustical compression waves that alter the refractive index of the crystal in a periodic pattern.
This leads, in effect, to the generation of a diffraction grating so that an orthogonal beam of polarized and collimated light incident at the Bragg angle is diffracted with high efficiency into the first-order beam. Variations in the frequency of the acoustic waves alter the spacing pattern in the crystal and, therefore, the wavelength of the diffracted light.
Likewise, altering the amplitude of the acoustic wave determines its relative intensity. Unlike other optical filter systems, AOTFs do not suffer from the mechanical constraints, speed limitations, image shift or vibration associated with rotating filter wheels, and can easily accommodate multiple wavelengths. They are mechanically robust and lightweight, and are ideal for field and handheld applications (Figure 5).

Figure 5. An integrated IR camera/acousto-optic tunable filter hyperspectral imaging system can activate optical taggants beyond the normal vision capabilities.
Commercial applications
Covert optical taggants for commercial applications must first address safety and environmental concerns. Low-toxicity materials, such as lanthanide-based emitters, are highly preferred over organic dyes and semiconductor nanocrystal taggants, especially for food and pharmaceuticals. The packaging and integration of taggants into marked objects should be tailored to particular application platforms and be as seamless and unobtrusive as possible.
The covert taggant implantation process can emulate the traditional marking and tracking techniques such as labels, logos, serial numbers and scannable identification numbers and bar codes. Computerized IR optical scanners and imagers with integrated photon-exciting light sources must be developed.

Figure 6. An optical taggant displayed as part
of a soldier’s sleeve is activated in the second photo using a
hyperspectral imager and optical source.
Tracking and identifying using covert IR taggants can become an important technique for asymmetric warfare conducted by the US military against unconventional enemies such as terrorists. Through “tagging, tracking and locating” with various forms of taggants and tagging technologies, the military aims to identify, track and locate enemy combatants and terrorist groups that often blend in with the general population. Illicit materials and assets, such as smuggled weapons, cash and narcotics, can also be tracked in helping to reveal and locate terrorist networks. For such purposes, passive and unobtrusive IR taggants can have significant advantages over other methods, such as radio-frequency tags.
Covert optical taggants can also benefit friendly forces by providing battlefield identification and communication to aid tactical operations, prevent friendly fire casualties and facilitate rescue missions. Figure 6 shows an activated taggant patch identifying a friendly soldier in clouded conditions.
Optical taggants emitting in the NIR range have been developed for battlefield operations. However, with the wide availability of 3G IR goggles capable of seeing up to 1 µm, they lose their effectiveness. Future taggants should operate in the
longer IR wavelength, which is a powerful future direction.
Meet the author
Dr. David Zhang is senior scientist at Brimrose Corp. of America in Hunt Valley, Md.; email: [email protected].
References
1. K. Binnemans (September 2009). Lanthanide-based luminescent hybrid materials. Chem Rev, Vol. 109, pp. 4283-4374.
2. R. Paschotta (May 2013). Laser gain media: A diverse family of materials. Phot Spect, pp. 57-60.
3. J. Kido, Y. Okamoto (June 2002). Organo lanthanide metal complexes for electroluminescent materials. Chem Rev, Vol. 102, pp. 2357-2368.
4. S. Keuleyan et al (Oct. 19, 2011). Synthesis of colloidal HgTe quantum dots for narrow mid-IR emission and detection. J Am Chem Soc, Vol. 133, pp. 16422-16424.
5. S. Andergassen et al (July 9, 2010). Charge transport through single molecules, quantum dots and quantum wires. Nanotech, Vol. 21, p. 272001.
6. S.V. Kershaw et al (April 7, 2013). Narrow bandgap colloidal metal chalcogenide quantum dots: Synthetic methods, heterostructures, assemblies, electronic and infrared optical properties. Chem Soc Rev, Vol. 42, pp. 3033-3087.
7. E.C. Scher and S.A. Empedocles (March 22, 2011). Nanocrystal taggants. US Patent No. 7,912,653 B1.