
Explaining Why Oil and Water Don’t Mix
Michael D. Wheeler
It's a principle most seventh-graders know: Oil and water don't mix. Of course, there is more to the story. They do interact at some level; for example, oil spreads evenly over water, rather than beading. So, if the bonding between water molecules is extremely strong and the bonding between oil molecules is relatively weak, what type of bonding causes the oil to spread rather than to dissolve?
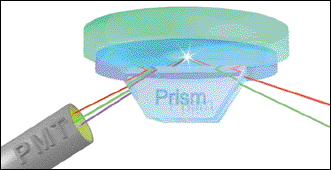
Researchers have used surface-specific vibrational spectroscopy to investigate the behavior of water and hydrophobic liquids. Solutions of water and oil are placed on a prism, and a photomultiplier tube collects the sum frequency for analysis. Courtesy of Lawrence F. Scatena, University of Oregon.
The question has baffled researchers for nearly four decades. Now a group at the University of Oregon has used surface-specific vibrational spectroscopy to investigate the behavior of water and hydrophobic surfaces at the molecular level.
The technique involves shining two pulsed laser beams at the interface, one a tunable infrared beam and the other a fixed visible beam. In this case, the research team used an Nd:YAG laser to deliver 1 to 2 mJ of infrared and 4 mJ of frequency-doubled 532-nm light.
The beams induce a polarization in the molecules at the interface, which produces a weak signal that reflects off the surface and that corresponds to the sum of the incident beams. Because this is a second-order optical process, it is inherently restricted to the surface and displays a surface specificity superior to other forms of molecular probing, such as x-ray and neutron scattering.
The researchers conducted experiments with CCl4 and hexane, probing the interface between these hydrophobic liquids and water. Having charted the vibrational sum frequency spectra, their findings were in stark contrast to generally accepted models of how water next to fluid hydrophobic surfaces behaves.
"We were able to see that there are weak interactions between the oil phase and the water phase," explained Geraldine L. Richmond, a professor of chemistry at the university. "Many have believed that there is little, if any, interaction. This weak interaction causes water molecules to orient at the interface in ways that were previously not anticipated."
Beyond theory
Given these strong interactions at the interface, the question arises as to why organic molecules do not immerse themselves in water and surround themselves with favorable water attractions.
"For this to happen, more bonds between water molecules in the bulk liquid would need to be broken, and this is energetically unfavorable," Richmond said. Most oil molecules thus stay out of the water's bulk, while those at the surface bond to the water molecules at the surface discontinuity that are not tetrahedrally coordinated.
The study goes a long way toward settling the debate over how the hydrogen in water bonds to other water molecules at these interfaces. "Because there have not been ways of measuring this, the field has largely been dominated by theorists, and they have presented a wide range of conflicting ideas," Richmond said.
The work has implications in macromolecular assembly, protein folding and oil recovery. Details of the experiment appeared in the May 4 issue of Science.
Published: September 2001