Extends the Reach of Far-Field Microscopy
Nancy D. Lamontagne, News Editor
By taking advantage of adaptive optics technology, confocal and multiphoton microscopy could achieve greater depth and resolution, to the benefit of biological studies.
In confocal and
multiphoton microscopy, light travels to the focal plane and excites fluorescence
in the sample, and the fluorescence travels back through the optics to a detector.
During this trip, the light meets different surfaces that introduce aberrations
into the image. In far-field techniques, these aberrations can reduce both the axial
resolution and the signal, placing severe limits on image quality and depth. Researchers
have found that adaptive optics may be able to dynamically correct for such aberrations.
Microscopes induce a limited number of aberrations
caused by the changes in the indices of refraction between the objective and immersion
fluid, the coverslip and the specimen, and even within the specimen itself as light
penetrates deeper. Researchers have tried to circumvent these errors using static
correction, but this method doesn’t work with multiple specimens.
Adaptive optics, invented for astronomy
to correct for the error induced by Earth’s atmosphere, may hold promise for
microscopy. The technique can compensate for aberrations by measuring the error
introduced by a specific sample — seen as a bend in the light’s wavefront
— and applying the opposite error to the wavefront.
Although there are several approaches
to finding the ideal shape to oppose the error, a deformable mirror is key to all
approaches. Such a mirror can form different shapes because it consists of a membrane
lying above many electrodes. Each computer-controlled electrode can pull the membrane
to change the mirror’s shape on the fly. Now that such mirrors are readily
available in a compact form for the relatively affordable price of around $8000,
researchers are trying to integrate them into microscopes.
Confocal microscopy
A group led by Tony Wilson at Oxford University
in the UK recently created such a system by using adaptive optics to correct for
aberrations in a confocal microscope. In this kind of microscope, the excitation
light and the emission light travel through the same optics, so both can be corrected
by inserting one membrane mirror. The researchers’ system used the focal point
of the light as the source of measured wavefronts, much as an artificial guide star
is used in astronomy.
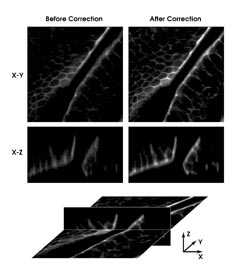
Adaptive optics used with confocal imaging allowed researchers at
Oxford University to improve x-z scans of mouse intestine. © 2002 National
Academy of Sciences, USA.
They have also applied the technique
to multiphoton microscopy. Martin Booth, a team member, sees the primary role of
adaptive optics as ensuring that a microscope performs optimally for all specimens.
For correction, the system requires
only a deformable mirror, such as one the scientists tested from OKO Technologies
in Delft, the Netherlands, and some lenses. They chose the mirror, made of an aluminized
silicon-nitride membrane above 37 hexagonal electrodes, because of its compactness
and low cost and because, it was one of the first few to appear on the market, Booth
said.
A number of combinations of optics
can produce the same results. “The configuration used in the PNAS paper
[April 30, 2002] is a tidy implementation which could be easily incorporated into
an existing microscope with the minimum amount of hardware,” he said. They
are now testing other optical setups to increase the speed of correction.
The researchers used their system to
detect and apply the correction needed for astigmatism, coma, trefoil and spherical
aberrations in a single field of view. Because each correction required two scans,
they turned down the laser power during the nonimaging scans to prevent photobleaching.
They found that the method shortened the point-spread function by a factor of 1.8
in an image of a 200-nm polystyrene bead. For a section of fluorescent-labeled mouse
intestine, the corrected images exhibited more contrast and sharpness than did the
uncorrected images.
They have demonstrated correction of
a few planes, and Booth said that once they understand the aberrations introduced
by biological specimens, it may be possible to measure the aberrations from a few
planes at different depths and use this data to calculate the correction for the
entire 3-D volume. Such an approach would reduce the number of scans needed to correct
for the entire image. They are working with a microscopy company to integrate their
system into a confocal microscope, which Booth thinks could be available in two
or three years.
Two-photon microscopy
Adaptive optics has an even greater potential
to improve two-photon microscopy, because the intensity of the two-photon signal
is quadratically dependent on the incident light. This means that aberrations make
the signal level fall drastically. Researchers led by Theodore Norris at the Center
for Ultrafast Optical Science at the University of Michigan in Ann Arbor used adaptive
optics to increase the resolution of multiphoton imaging. They hope that one day
they will be able to apply the technique to image deeply into tissue for the study
of drug delivery systems.
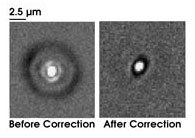
Researchers at the University of Michigan in Ann Arbor used adaptive optics to increase the resolution
of multiphoton imaging. They captured images of two-photon fluorescence at a focus depth of 600 μm before and after correction. Reprinted with permission from the Royal Microscopy Society, Journal of Microscopy.
They also used the OKO deformable mirror
to correct for various aberrations, including both off-axis and on-axis. Instead
of directly measuring the wavefront, they used a genetic learning algorithm in conjunction
with nonlinear excitation from ultrashort pulses to find the best correction, because
they knew that the optimum shape of the mirror would produce the brightest signal.
They hoped this approach would avoid the loss of power that comes with direct measurement.
They first tried to correct for depth-induced
spherical aberration by applying an opposite spherical aberration. In a solution
of the dye coumarin and water — which has the same properties as a biological
sample — they achieved an optimum solution in 10 generations (three minutes)
that increased the scanning range from 150 to 600 μm. By applying a defocus
bias to correct the same error, they were able to increase scanning range to 800
μm. Although the defocus bias produced better results, it suffered from a 50
percent loss of energy, which, the researchers say, would not be suitable for multiphoton
imaging.
One drawback — that the mirror
can be shaped in only one direction (pulled but not pushed) — prevents the
mirror from being formed into the shapes necessary to correct for more complex aberrations.
However, the mirror can be used to correct most aberrations in a practical microscope
system, said team member Jing Yong Ye. Next, the group plans to test the device
on real biological systems.
John Girkin and Paul Marsh at Strathclyde
University’s Institute of Photonics in Glasgow, UK, are also using adaptive
optics with the purpose of producing improved multiphoton images from deep within
samples. Because multiphoton microscopy works by producing only a limited excitation
volume, controlling this volume is crucial to the overall performance of the system.
With a well-confined spot, every fluorescent photon can be employed to build up
the image.
Rather than mathematically calculating
the best corrections, the scientists are focusing on producing the best image. They
have inserted a deformable mirror, again from OKO Technologies, into a point-scanned
two-photon microscope. Although this reduces the potential field of view, it promises
higher-quality images at depth.
According to Girkin, in a final practical
system, users are more concerned about the quality of the images than about knowing
the exact correction applied by the mirror. However, the researchers had to ensure
that beam manipulation was not creating artifacts of the image. Therefore, to keep
the images accurate as they delve deeper, they make sure that the structures remain
consistent.
Protect the sample
Their method corrects for the average errors in
a plane, but as they image more deeply, errors increase, and they also would like
to correct for each point. However, Girkin said that the time required to change
the mirror to correct for 256 x 256 points might damage the sample too much. “You
sometimes have to compromise the ultimate optical performance in favor of producing
biologically significant images,” he said.
Potentially, though, adaptive optics
could do more than allow deeper imaging. For instance, because multiphoton imaging
produces only sectioned images, finding the area of interest can be difficult and
time-consuming, which can result in photo-toxic “damage” to the sample.
But, Girkin said, with adaptive optics’ potential to rapidly alter the beam
shape, it could be used to quickly find the region of interest by changing the beam’s
shape into an ellipse (longer vertically than horizontally) so that the planes would
become thicker. Once the image appeared, the beam could be quickly changed back
to the ideal shape.
In multiphoton microscopy studies,
protecting the biology is especially important because, once damage begins, it tends
to continue at a very fast rate, Girkin said. “The farther you can stay from
this threshold of biological damage, the better.” Adaptive optics allows
imaging using lasers with lower average powers because the users can maintain a
small excitation region.
Typically, Ti:sapphire lasers with
average powers of around 500 mW are used in multiphoton microscopes, though only
a small fraction of the power is used for most imaging applications. The extra power
can be required when the user wishes to go deeper into the sample, but adaptive
optics can allow the use of less power because the excitation photons are used more
efficiently. Less power means lower-cost sources and less damage to the sample.
“This technology could potentially
reduce the cost of multiphoton microscopy and open up new biological systems for
study,” Girkin said.