Daniel S. Burgess, Senior Editor
In the years since Bio-Rad Laboratories Ltd. of Hemel Hempstead, UK, licensed the right to commercialize multiphoton microscopy systems from Cornell University in Ithaca, N.Y., turnkey multiphoton workstations have appeared in labs worldwide. Researchers in Scotland are using adaptive optics to improve the performance of multiphoton microscopes for biological imaging, offering a glimpse of what future versions of the instruments might look like.
Multiphoton microscopy, developed in 1989 by Winfried Denk, James P. Strickler and Watt W. Webb, is an outgrowth of confocal scanning microscopy that uses less-energetic, longer-wavelength photons, tightly focused into a small excitation volume, rather than single, shorter-wavelength ones, to excite fluorescence in a specimen. The result is a significant reduction in phototoxic events within the sample, no photobleaching of fluorophores outside the focal volume and, therefore, the potential for long-term imaging of live samples. In addition, the longer-wavelength photons penetrate with less scattering, enabling imaging at greater depth.
John M. Girkin, Paul N. Marsh and David Burns of the University of Strathclyde’s Institute of Photonics in Glasgow, UK, are one of several teams investigating adaptive optics in multiphoton microscopes. In their recent work, their setup increased the imaging depth of the instrument, with submicron resolution, from 3.4 to 46.2 μm using an oil-immersion lens and a water sample medium, a common biological preparation.
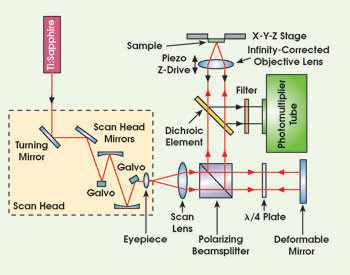
Researchers are using adaptive optics to improve the performance of multiphoton microscopes for biological imaging. A 15-mm-diameter deformable mirror in the scanning optics induces corrective distortions in the excitation light that optimize the response from the sample. Images courtesy of John M. Girkin.
Adaptive optics systems employ deformable mirrors — or other optical elements whose properties may be changed rapidly and repeatably — to introduce distortions that compensate for the aberrations caused by dynamic variations in the media through which an optical signal passes. Although they largely have been the purview of astronomy, microscopists are discovering that the approach also works to counter the aberrations in their systems, such as the spherical and higher-order ones due to the refractive-index mismatch between the immersion medium of a high-numerical-aperture objective lens and the specimen and the additional aberrations introduced by the specimen itself.
The Strathclyde researchers introduced a 15-mm-diameter mirror from Flexible Optical BV of Delft, the Netherlands, into the scanning optics of a multiphoton microscope. The output of a Coherent Mira Ti:sapphire laser, operating with 200-fs pulses of 750-nm light at a rate of 76 MHz, was directed into a Bio-Rad MRC 600 scan head. After the linearly polarized excitation light emerged from the scan head, it passed through a scan lens, polarizing beamsplitter and quarter-wave plate onto the silicon-nitride membrane mirror. From the mirror, it was reflected back through the wave plate and redirected by the beamsplitter through a dichroic element and into a Nikon 1.3-NA 40× oil-immersion objective lens to the sample. A photomultiplier tube collected the resulting fluorescence directly after the dichroic mirror in the return path.
Unlike adaptive optics systems in telescopes, which compensate for aberration in the incoming signal, the microscopy setup applies its correction to the excitation light to optimize its ability to generate bright, sharp responses at the desired depth in the specimen. This enables not only deeper imaging and higher resolution, but also the use of lower laser powers at shallower depths to avoid photodamage.
Ideally, the system would compare the characteristics of several images to establish the best setting for the mirror for imaging at that focal volume in the sample. Unfortunately, repeated stimulation, even by the less-invasive multiphoton technique, can lead to phototoxic effects in biological specimens. To establish the settings for the 37-actuator mirror, therefore, the researchers first optimized the point-spread function of the fluorescence response for various depths in nonbiological samples that simulated the biological ones. When imaging a real specimen, they then could select from a group of the settings for that depth and choose the best in only a few tries. Tests with artificial samples and with muscle from guinea pig bladder confirmed improved resolution and imaging depths in the system.
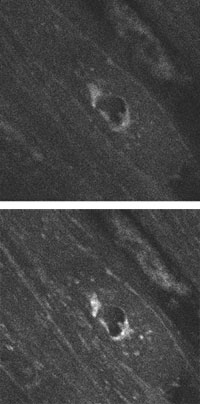
Images of a 32 × 32-μm slice of guinea pig bladder at an objective movement of 30.8 μm into the specimen without (top) and with corrections (bottom) illustrate the enhanced performance of a multiphoton microscope equipped with adaptive optics.
Achieving the optimum performance from adaptive optics will require a transition from the current, relatively complicated optical assembly. “Yes, it works, and it’s a microscope, and you can put biologically significant samples in there, but you still need a physicist holding the biologist’s hand,” Girkin said. A better system would be one that does not require the user to understand what is going on inside the box.
Contact: John M. Girkin, University of Strathclyde, Glasgow, UK; +44 141 553 4120; e-mail: [email protected].
References
P.N. Marsh et al (May 19, 2003). Practical implementation of adaptive optics in multiphoton microscopy. OPTICS EXPRESS, pp. 1123-1130.