The threat of chemical and biological terrorism and the infusion of hazardous materials into inhabited areas have created the demand for two field-deployable analytical techniques.
Jay Thomason, Ocean Optics Inc.
With important advances in the miniaturization, cost and performance of optoelectronic devices and sampling accessories, techniques such as fluorescence spectroscopy and laser-induced breakdown spectroscopy have found new applications in environmental monitoring, in chemical and biological warfare detection and in other security areas. Fluorescence spectroscopy is the more familiar standard, but it is being used today to address new research challenges. The relatively new laser-induced spectroscopy technique also is stepping up to the plate in novel instrumentation and application areas.
The old tried and true
Fluorescence spectroscopy, a mainstream analytical technique that is solidly rooted in the laboratory environment, is finding roles in an important new security application — the real-time, on-scene detection of anthrax.
Field-deployable fluorescence systems that cost less than $5000 can instantly detect endospores in powders suspected of containing anthrax. These portable units weigh less than 10 pounds and are now available to first responders. Indeed, the creation of miniature, portable fluorescence systems has enabled optical sensing applications that apply new assays, such as lanthanide reagent chemistries developed at the US Army Research Laboratory, to examine the photoluminescence of bacterial endospores, including substances believed to be concentrated forms of anthrax.
Using portable systems in the field begins with testing for the presence of a major spore component, which is extracted from the endospores when added to an Army-developed reagent in a disposable test vial. Only two genera of bacteria are known to produce endospores — Clostridium and Bacillus, the latter of which includes Bacillus anthracis (anthrax).
Several specific technological advances have made it possible for portable fluorescence systems to perform rapid, laboratory-quality, multiwavelength analysis across a broad spectral range. Among the advances are the development of filtering techniques and the availability of sensitive time-gated detectors, which have dramatically improved the performance of modular spectrofluorometric devices. In particular, mass-market, low-cost detectors have been improved in such key performance parameters as sensitivity, which is critical for fluorescence and other low-light techniques. This gives researchers the advantages of miniature fiber optic spectroscopy without the size and cost limitations associated with the scientific-grade detectors common to laboratory fluorescence instrumentation.
Not only have low-cost detectors with greater sensitivity made high-performance miniature spectroscopy possible, but they also have inspired additional advances. For example, innovative filtering technologies, such as Ocean Optics Inc.’s linear variable filters, have added a dimension of versatility to modular fluorescence platforms. The filters can be combined to provide dynamic wavelength and bandpass selection, tasks previously accomplished only through the use of multiple discrete filters, or to spectrally shape excitation sources, providing excellent separation of excitation and emission energy and eliminating the need for external scanning monochromators. Fluorescence analyses that once were restricted to the laboratory can now easily be performed in situ.
Another advance in fluorescence instrumentation is the installation of silver-coated mirrors in the spectrometer’s optical bench, which has proved to increase system sensitivity — for example, to the picomolar range for fluorescein.
Variable-delay time-gated detection also is useful for measuring compounds such as lanthanides, which have long emission lifetimes. By gating the spectrometer, light energy from the excitation source and shorter-lived fluorescence emissions do not interfere with the desired emission spectrum.
The new(er) test on the block
Laser-induced breakdown spectroscopy is a laser spark analytical technique used to determine the elemental composition of various solids, liquids and gases (Figure 1). A high-power laser pulse is focused on a sample to create a plasma or laser spark. The atoms and ions in the plasma emit light energy that is collected by a lens or fiber optic cable and analyzed by a time-gated spectrometer. The atomic spectral lines can be used to determine elemental composition (and concentrations in some systems). One benefit of the technique is that little or no sample preparation is required, making it field-portable.
Its use in chemical analysis dates to the early 1980s; today its applications range from art restoration to complex metals analysis to the more sobering task of identifying hazardous materials, and chemical and biological warfare agents.
Researchers use the technique to ablate samples with a high-power, short-pulse laser to create microplasma. Temperatures in the microplasma continuum can reach 20,000 K. As it cools, the microplasma’s atomic components emit light energy, providing discrete spectral fingerprints that can be captured and resolved with optical fiber and a spectrometer with a CCD array detector.
This type of spectroscopy is excellent for analyzing trace amounts of materials, making it practical for use as a scanning tool. For instance, chemical and biological warfare agents and other hazardous materials can be concealed in many types of containers, such as parcels and luggage. Traces of chemical and biological materials might be attached to the surface of these containers when the package is prepared. In standoff-mode, the spectroscopy method is used to rapidly interrogate large volumes of containers (airline luggage or mail on a conveyor belt) or to quickly examine single items, such as a personal carry-on item in commercial air travel.
In applications such as biological warfare agent detection, laser-induced breakdown spectroscopy can be used to differentiate between closely related Bacillus species, as shown in the spectra of anthrax surrogates (Figure 1).
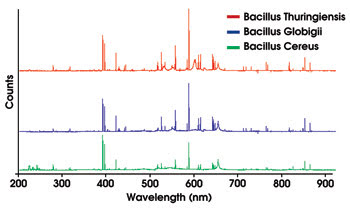
Figure 1. These spectra of anthrax surrogates were taken with the Ocean Optics LIBS2000+ laser-induced breakdown spectrometer. Courtesy of the US Army Research Laboratory and the US Army Soldier and Biological Chemical Command.
Recent advances in component instrumentation have played a key role in the refinement of these spectroscopy systems. For example, broadband (200 to 980 nm), high-resolution spectrometers enable the characterization of molecular and biological samples in a single laser pulse; detection of parts-per-billion and picogram quantities of elements can be resolved at a resolution of 0.1 nm (full width half maximum). By incorporating high-speed data transfer technology in these systems, such as USB 1.0, full spectral scans can be sent to a PC’s memory in less than 100 ms, raising elemental qualification to computer speed.
Progress in the miniaturization of components such as compact high-power lasers and technological improvements in CCD-array spectrometers, sampling probes and batteries, will make possible the further development of lightweight, portable systems for the field. These systems will enable the fast and reliable detection of explosives, biological pathogens and chemical agents.
Meet the author
Jay Thomason is marketing communications manager for Ocean Optics Inc. in Dunedin, Fla.; e-mail: [email protected].