The optical properties of these materials fulfill the requirements for LED packaging.
Dr. Ann Norris, Dow Corning
Solid-state lighting, such as LEDs, is an emerging platform that, in some cases, will replace conventional incandescent and fluorescent lighting. This technology has many benefits: It will increase energy savings, is more reliable and environmentally friendly, and has a smaller footprint, a longer life and more versatility.
But for LEDs to be competitive with existing lighting options for the general illumination market, they must not only increase light output and efficiency, but also achieve an overall decrease in cost. High-brightness LEDs offer a means of reaching these objectives.
Many of the currently available high-brightness LEDs operate with a junction temperature of at least 125 °C and must maintain their intensity for about 100,000 hours. These conditions are quite demanding for the packaging materials used in the device construction.
The encapsulant/packaging material is not only what protects and surrounds the LED (semiconductor substrate), but also the path through which the light is extracted from the device. Therefore, it’s critical that the encapsulant material maintain its optical clarity throughout the device’s lifetime.
Siloxanes have a number of inherent properties that make them an attractive choice over other organic polymers for high-brightness applications, among them excellent clarity and thermomechanical stability.
Many options
Silicones can be formulated to provide dispensable, very low modulus soft gels and elastomers. This is an important property for relieving stress on the package components. They also can be formulated to very rigid resins that can be transfer- or injection-molded; in this form, they are very tough and provide mechanical protection. This wide range of material options results from the many ways in which the basic silicone precursor molecules, consisting of linear (D), branched (T and Q) and terminal (M) monomer units, can be combined in a silicone composition (Figure 1).
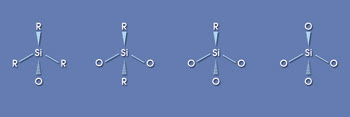
Figure 1. There are various siloxane structures that can be synthesized. The R group is sequentially replaced by oxygen atoms leading to a more rigid material.
Siloxane materials can be made with very high transparency and a wide range of refractive indices, and they offer a number of cure options, depending upon the application requirements. The refractive index of today’s siloxane commercial products ranges from 1.38 to 1.55. The nature of the R groups attached to the silicon atom (shown in Figure 1) controls the index. Experimental materials have been developed with refractive indices as high as 1.65. The refractive index of the encapsulant material is one contributor to defining the maximum light output (brightness or extraction efficiency) of the LED device.
With a higher refractive index, less light is lost from internal reflection. To improve light extraction and, therefore, brightness, it is critical to be able to control the material’s refractive index so that it more closely matches that of the LED’s semiconductor substrate.
Silicons have another important property: high thermal stability, which is associated with the inherent stability of the silicon-oxygen polymer backbone. Silicones have been shown to withstand very high temperatures in multiple applications without exhibiting significant changes in key properties. They also have excellent stability in high flux at wavelengths from about 350 to 800 nm because of the absence of absorptions in this region (Figure 2).
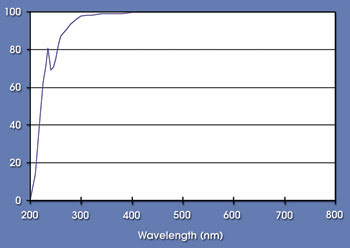
Figure 2. In this representative absorption spectrum for polydimethylsiloxane, the percentage of transmission is shown as a function of wavelength through a 1-mm-thick material.
In one study, no change in the refractive index or transparency of samples was detected after exposure to accelerated aging conditions of high heat and humidity for up to 2000 hours. In another study, silicone gels and commercial epoxies were exposed to 370-nm light at 150 °C long enough to induce significant reductions in the transparency of the epoxies but not to cause measurable change in the silicone’s transparency. The results of these studies speak to the material’s excellent photothermal stability.
Meet the author
Ann Norris is Associate Scientist in the Electronics & Advanced Technologies Group at Dow Corning in Midland, Mich.; e-mail: [email protected].