Recent developments in the production of synthetic diamond abrasives have resulted in an array of varieties suitable for optical finishing applications.
Dr. Nicholas J. Tumavitch, Warren/Amplex Superabrasives
Monocrystalline diamond abrasives, both natural and synthetic, are used widely in the lapping and polishing steps of optical component manufacturing, but inherent structural defects will limit their application in the fabrication of next-generation products.
Recently, diamond manufacturers made some morphological refinements to compensate for the inadequacies of the material. As a result, engineers are achieving higher removal rates and finer, scratch-free surfaces.
The functional utility of all types of diamond abrasives increases with decreasing particle size distribution, and nanoparticle technology will play a role in their synthesis for optical manufacturing applications. To put the relative size of these materials in context, consider that one carat (200 mg) of 100-nm diamond abrasive contains slightly more than 100 trillion pieces (Table 1). Because the number of pieces per carat is proportional to the cube of the radius, the mass of an equivalent number of pieces of 3-μm diamond is 100,000 carats, or roughly 44 pounds. The mass of an equivalent number of pieces of 30-μm diamond, a size typically used in cutting wheels, is 22 tons.
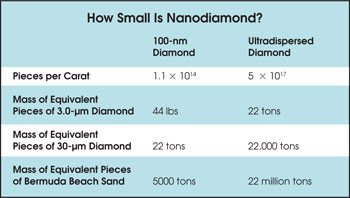
One carat of another type of nanodiamond, ultradispersed diamond, contains about 100,000 trillion pieces approximately 5 nm in diameter (Figure 1). The mass of an equivalent number of pieces of 300-μm Bermuda beach sand is 22 million tons. That much sand would fill 550 miles of 50-yard-wide beach to a depth of 1 foot, or roughly 10 times the perimeter of Bermuda. A carat of ultradispersed diamond thus may be the source of the old adage, “as much as all the stars in the sky or grains of sand on the beach.”

Figure 1. Ultradispersed diamond (top) often is referred to as “nanodiamond,” but it more accurately is a class of nanodiamond. One carat — i.e., 200 mg — of the material contains approximately 1017 particles, with average diameters from 2 to 10 nm. The mass of an equivalent number of 300-μm-diameter pieces of Bermuda beach sand (bottom) is 22 million tons.
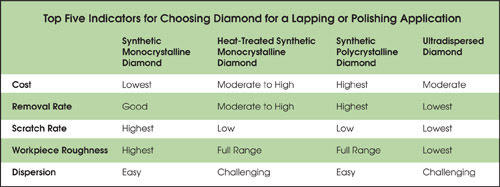
With a variety of types of diamond abrasives now available to the optical component manufacturer, it is helpful to understand how they differ. Each has its advantages and disadvantages, and there is a fine line to walk between optimum performance and cost-effectiveness. The summary in Table 2 is offered as a guide to selecting a diamond abrasive for new or next-generation finishing processes.
Note, however, that the performance of diamond also may be controlled with the slurry chemistry. A slight change in dispersion or slurry viscosity can reduce the scratch rate and/or increase the material removal rate. Whether one is considering process improvements or new state-of-the-art finishing applications, approach the descriptions that follow with the assumption that all other manufacturing conditions — including machinery, work pressures and slurry — are constant.
 Monocrystalline diamond
Monocrystalline diamond
Monocrystalline diamond is a highly ordered crystalline solid. The carbon atoms are linked in a regular fashion. Each atom shares one of its outer shell electrons with four other carbon atoms in an equally spaced tetrahedral environment.
The material, in both natural and synthetic forms, was the primary abrasive in the finishing industry before the turn of the millennium. Today, synthetic monocrystalline diamond is used 30 times more by volume than natural diamond because of its advantages in terms of cost and supply.
Artificial synthesis requires pressures exceeding 60 kilobars to emulate the conditions that produce diamonds in nature. The first attempts to convert graphite to diamond at low temperature and high pressure were unsuccessful. The reaction speed, for example, was too slow for the process to be considered cost-effective. To increase the reaction kinetics, higher temperatures were needed, but they, in turn, required even higher pressures. This dilemma is attributable to the positive slope along the graphite/diamond stability line on the pressure-temperature phase diagram (Figure 2).
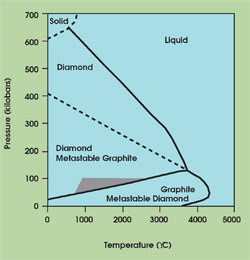
Figure 2. Early attempts to artificially synthesize diamond were unsuccessful, as researchers confronted the positive slope along the graphite/diamond stability line. Increasing the reaction kinetics demanded higher temperatures and, in turn, even higher pressures. Today, all production of synthetic monocrystalline diamond is performed at pressures and temperatures within the gray region of the phase diagram, using nickel catalysts to lower the temperature to about 2000 K and the pressure to 60 kilobars.
The transition conditions influenced the evolution of the modern diamond press, which has witnessed 50 years of refinements since its birth at General Electric Co. in the 1950s. Today, presses use nickel catalysts to lower the temperature to about 2000 K and the pressure to 60 kilobars. Mechanically, the presses differ slightly throughout the world, but all synthetic monocrystalline diamond production falls within the gray region of the phase diagram. Production factories may have hundreds of presses, because the reaction chamber is quite small. One synthesis run may produce fewer than 100 carats, and the raw diamond then must be crushed, milled, cleaned and graded.1
In terms of performance, synthetic monocrystalline diamond abrasive has a good material removal rate. Workpieces finished with this form of diamond, however, are the roughest compared with the others described in this review because the edges of the material are large in relation to the total crystal size, and are sharp and not friable (Figure 3).

Figure 3. Being relatively large, synthetic monocrystalline diamond (top) offers a high material removal rate. A disadvantage of the material is the existence of slivers (bottom) that can cause subsurface damage during finishing.
Its toughness enables micron- and submicron-size synthetic monocrystalline diamond to retain some of the shape characteristics of the parent crystals, such as pyramids and trigons, after the crushing and milling process.
Synthetic monocrystalline diamond is resistant to acids and to hot caustic solutions, so it can be cleaned to very low impurity levels. The batch-to-batch quality from suppliers thus is very consistent.
Futhermore, the cost per carat is the lowest of the types of diamond described here. Prices are currently 25 to 75 cents per carat, depending on the cleanliness and the standard deviation of size.
A disadvantage of synthetic monocrystalline diamond is the inclusion of slivers (needlelike pieces) and plate structures (thin, flat pieces) in feed batches, which form when the 011 crystal plane cleaves during the crushing process. If these elongated structures are oriented perpendicular to the workpiece during finishing, the excessive pressure may produce subsurface damage. Irregular finishing patterns and lower removal rates also can occur if a plate glides horizontally across the workpiece.
Slivers a challenge
Reducing excessive slivers and plates from a feed batch presents a considerable challenge to diamond suppliers. The plates and slivers are hydrodynamically equivalent to smaller, more regularly shaped pieces. During elutriation, the slivers gather momentum from the added frictional component and are carried over into the graded batch. The phenomenon is similar to dropping two pieces of paper, one crumpled and one flat, from shoulder height to the ground. The crumpled paper will hit the ground faster because the frictional force of air “floats” the flat paper.
Slivers and plates may be removed in milling if the energy selected is above the cracking energy of the thin pieces and below the cleaving energy of regular-shaped pieces. The smaller fragments then will be removed in the grading process. But extra-tight distributions add significant production time and costs.
Synthetic polycrystalline diamond is produced by explosive shock synthesis. Several tons of explosives generate about 250 kilobars of pressure on the graphite feed.
Each piece of synthetic polycrystalline diamond contains smaller “microcrystallites” whose planes are oriented in different crystallographic directions every 10 to 50 nm, regardless of the particle size distribution of the parents. The hardness of an individual microcrystallite is comparable to that of synthetic monocrystalline diamond.
The difference between these materials is discernible in scanning electron microscopy, however, and transmission electron microscopy clearly reveals the microcrystallite structure (Figure 4).
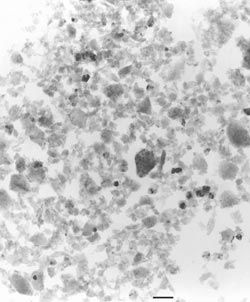
Figure 4. Synthetic polycrystalline diamond abrasive has a material removal rate up to 10 times higher than synthetic monocrystalline diamond because it has more cutting edges and a higher surface area. Each piece of synthetic polycrystalline diamond contains smaller “microcrystallites” that are visible under a transmission electron microscope.
Synthetic polycrystalline diamond abrasive has a material removal rate up to 10 times higher than synthetic monocrystalline diamond because it has more cutting edges and a higher surface area. Multiple edges and corners are in simultaneous contact with a workpiece, reducing the probability of subsurface damage from excessive pressures.
Its most obvious disadvantage is the sticker shock it can produce. On average, it costs 10 times as much as synthetic monocrystalline diamond, at $2.10 to $8 per carat. But the potential user should consider that the need to use less diamond in the finishing process easily recovers these higher up-front costs and that the higher material removal rate results in additional savings in terms of labor.
In comparison with synthetic monocrystalline diamond, batch-to-batch consistency is more challenging for synthetic polycrystalline diamond suppliers for two reasons. First, unlike synthetic monocrystalline diamond, the edge sharpness and crystallinity can vary with milling conditions. Undermilling tends to produce a rougher diamond surface with a relatively higher scratch rate (that is, in comparison with better-quality synthetic polycrystalline diamond; its scratch rate still is far lower than that of synthetic monocrystalline diamond). On the other hand, overmilling dulls the edges of the pieces, lowering the material removal rate.
Impurity
Secondly, impurity, in the form of pieces of transitional carbon, can reach unacceptable levels. Transitional carbon may be thought of as synthetic polycrystalline diamond oriented in one direction. This impurity can result in a lower material removal rate. It can be spotted using a scanning electron microscope, and analysis by transmission electron microscopy is required to control and select batches with consistent edge definition.
Heat-treated synthetic monocrystalline diamond is manufactured by heating well-graded and -cleaned material in an inert atmosphere to about 1500 K. Under these conditions, the powder darkens from light gray to black, a color change that represents a reorganization of the surface into a more disordered, graphitelike layer (Figure 5).

Figure 5. Heating well-graded and -cleaned synthetic monocrystalline diamond in an inert atmosphere to about 1500 K induces a color change. This is indicative of the reorganization of the surface into a more disordered, graphitelike layer that lowers the scratch and defect rate.
The particle size distribution of heat-treated diamond often is tighter than that of synthetic monocrystalline or polycrystalline diamond. Unwanted pieces on the fine end of the distribution tend to fuse into larger clusters or to disappear in the heat-treating process.
The graphite sheets provide a lubricating region between a sharp diamond edge and the workpiece. This shell coating lowers the scratch and defect rate compared with synthetic monocrystalline diamond, with its difficult-to-remove slivers. Because the batch is engulfed in the high-temperature oven, the heat-treating process shapes and lubricates the slivers that might otherwise have gone on to scratch or damage a surface.
Although its material removal rate is comparable to that of synthetic polycrystalline diamond, heat-treated synthetic monocrystalline diamond has a distinct advantage: reduced diamond embedding in the workpiece. Customer feedback indicates that workpieces show 33 percent less embedding when using heat-treated synthetic monocrystalline diamond rather than other diamond sources.
Great care must be taken when preparing dispersions of this diamond, however. The heat-treating process decreases the surface polarity, resulting in a tighter stability region. In fact, heat-treated diamond must undergo a proprietary surfactant soak to add polarity to the surface, or else the material will settle clear in an aqueous environment in a matter of minutes.
The cost is about 10 percent less than comparable synthetic polycrystalline diamond. Once again, the benefits can more than compensate for the price.
Ultradispersed diamond is produced in the diamond stability region at a temperature of 3000 K and a pressure of 100 kilobars. One difference in process distinguishes it from other diamond types: The feed, explosives and product are contained within the explosion chamber.
The chamber and explosives must be oxygen-deficient (<6 percent) to reduce the incidence of unwanted by-products and of conversion of the ultradispersed diamond back into the more thermodynamically stable graphite. The product, unlike other diamond types, contains many surface functional groups, such as methyl, nitrile, COH, COOH, CO and C6H6. It is composed of carbon (87 percent), oxygen (10 percent), nitrogen (2 percent) and hydrogen (1 percent). The functional groups reduce its density to about 2.8.
Ultrasmall particles
As mentioned earlier, ultradispersed diamond particles are extremely small, so one carat of the material contains approximately 1017 pieces. The diameter of the pieces falls within a range of 2 to 10 nm, with the predominant sizes being 3 to 7 nm.
This incredibly small size range has led to some inconsistencies in nomenclature. Ultradispersed diamond often is referred to as “nanodiamond.” However, the term actually encompasses all types of diamond with a particle diameter of 100 nm or less, and such nanosize diamond is readily available from suppliers of synthetic monocrystalline, synthetic polycrystalline or heat-treated synthetic monocrystalline diamond. Ultradispersed diamond simply is a class of nanodiamond.
Another term for this material that is gaining popularity is “cluster diamond.” This is unique to ultradispersed diamond so long as it reflects a clustered diamond and not nanocluster diamond. The reason for this distinction is that ultradispersed diamond is composed of primary and secondary particles. The primary particles cannot be broken down into smaller sizes and exist in suspension as clusters. The size of a cluster can range from a few primary particles to a few microns.
Work in progress
Because ultradispersed diamond is relatively new to the market, quantifying its performance is a work in progress. However, it is expected to be the most tested type of diamond abrasive in 2005, so the data gap will close quickly.
Initial results indicate that its material removal rate is relatively low, which is to be expected when finishing to angstrom- or subangstrom-level roughness with nanoparticles. The finishes it yields are relatively scratch-free because rogue slivers are not produced in the manufacturing process.
Another advantage of this material is that the edges are not subject to the attrition variability inherent in all other diamond types. Ultradispersed diamond particles are produced directly in the explosion process — unlike all other types of diamond, which are chips from parent pieces. The control of cluster size appears to be the primary parameter for ultradispersed diamond products, as the finishing quality will change with cluster size.
Reference
1. Ron Abramshe (January/February 2004). Grading techniques, analysis, and standardization of micron materials. ABRASIVE MAGAZINE, pp. 10-14.
Meet the author
Nicholas J. Tumavitch is the manufacturing and development chemist at Warren/Amplex Superabrasives in Olyphant, Pa. He received an undergraduate degree in chemistry from The University of Scranton in Pennsylvania and a PhD in physical chemistry, specializing in interfacial thermodynamics, from State University of New York at Buffalo; e-mail: [email protected].