Device is effective for both liquid- and gas-phase observations.
Breck Hitz, Senior Technical Editor
By growing a porous material on the end of an optical fiber, investigators at the New Mexico Institute of Mining and Technology in Socorro have developed a versatile detector capable of in situ, real-time monitoring of chemicals in vapors and in aqueous solutions. The sensor works by measuring the reflectivity of the porous material, which changes when it absorbs the target chemical into its pores.
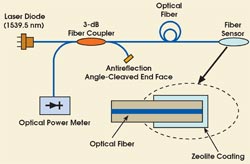
Figure 1. The intensity of light reflected from the coated fiber tip changes when the porous coating material absorbs the monitored chemical into its pores.
Joined for part of the work by scientists from Sandia National Laboratories in Albuquerque, N.M., and from the University of Illinois at Chicago, the institute researchers illuminated the fiber with a diode laser and monitored the light reflected from the coated fiber tip with an optical power meter (Figure 1). The coating material on the tip was a form of zeolite, a family of aluminosilicate crystals with nanometer-scale pores that can selectively absorb molecules based on their size and shape (Figure 2).

Figure 2. The ~4-μm-thick zeolite coating on the end of the fiber selectively absorbs molecules based on their size, shape or both.
Response time
In their gas-phase experiments, the investigators monitored concentrations of isopropanol vapor in nitrogen from 1.3 molecular percent down to 0.075 molecular percent (Figure 3). The detector had a fast response time (~1 s) when responding to the addition of isopropanol vapor to the mixture, but a somewhat slower response (~3 s) when the vapor concentration was decreased. Nonetheless, the signal was reversible and monotonic over the whole measurement range. They observed similar response from benzene and acetone vapors and believe that improvements in sensitivity and selectivity are achievable by choosing the zeolite structure and modifying its chemistry.
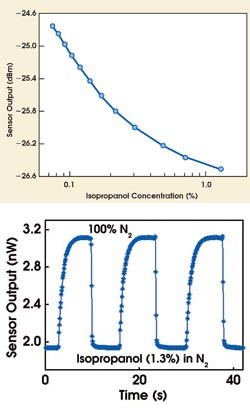
Figure 3. The reflected signal at the power meter decreased as the isopropanol concentration decreased, from 1.3 molecular percent to 0.075 molecular percent. The bottom shows the temporal response of the detector to a change in isopropanol concentration.
The observational techniques and results for the detector in aqueous solutions were similar to those for vapors. The scientists saw a measurable change in reflectivity as they changed the concentration of 2-propanol in deionized water from zero to almost 17,000 parts per million by volume, and they inferred that the detector’s limit could be less than 10 parts per million by volume. Mainly because the organic molecules diffuse more slowly in a liquid than in a gas, the response time of the detector in liquid (~10 to 12 s) was slower than the ~1-s response time in gas.
The researchers also measured the detector’s response to pentanoic acid, one of the major organic pollutants in oil-field water, and observed a much slower response time (~40 min) than with 2-propanol. They attributed the difference in speed to the diffusion rates for the two molecules, both through the liquid and into the zeolite pores, which relate to the molecules’ dissimilarities in mass, size and shape. By changing the chemical composition of the zeolite on the fiber tip, they increased the strength of the measured signal for a given pentanoic acid concentration, but the response time remained unchanged.
Optics Letters, June 1, 2005, pp 1270-1272; Langmuir, Sept. 13, 2005, pp. 8609-8612.