NASHVILLE, Tenn., May 22, 2006 -- A team of researchers has achieved a long-sought scientific goal: using laser light to break specific molecular bonds. The process uses laser light, instead of heat, to strip hydrogen atoms from silicon surfaces. This is a key step in the manufacture of computer chips and solar cells, so the achievement could reduce the cost and improve the quality of a wide variety of semiconductor devices.
The technique was developed by Philip I. Cohen at the University of Minnesota, working with Vanderbilt University researchers Leonard C. Feldman, Norman Tolk and Zhiheng Liu along with Zhenyu Zhang from Oak Ridge National Laboratory and the University of Tennessee. It is described in the May 19 issue of the journal Science.
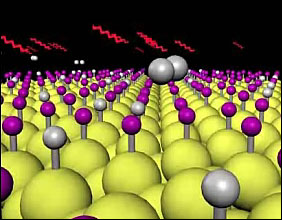
Laser light proves to be a gentle way to clear hydrogen from a silicon surface. (Photo: Brian Muller, Vanderbilt University)
"We live in the silicon age," said Tolk, a physics professor at Vanderbilt. "The fact that we have figured out how to remove hydrogen with a laser raises the possibility that we will be able to grow silicon devices at very low temperatures, close to room temperature."
Microelectronic devices are built from multiple layers of silicon. In order to keep silicon surfaces from oxidizing, semiconductor manufacturers routinely expose them to hydrogen atoms that attach to all the available silicon bonds. However, this process, known as passivation, means that the hydrogen atoms must be removed before new layers of silicon can be added. "Desorbing" the hydrogen thermally requires high temperatures and adds substantially to difficulty of process control because these temperatures create thermal defects in the chips and so reduce chip yields.
"One application that we intend to examine is the use of this technique to manufacture field effect transistors (FETs) that operate at speeds about 40 percent faster than ordinary transistors," said Cohen, a professor of electrical and computer engineering. "It should be possible to reduce the processing temperature of manufacturing FETs by 100 degrees Celsius. This should dramatically improve yields."
Vanderbilt, the University of Minnesota and Oak Ridge National Laboratory are filing a joint patent on the process and its potential applications.
In addition to a variety of potential applications, the discovery has important scientific implications. Since the invention of the infrared laser, chemists have been trying to use it to drive chemical reactions along nonthermal pathways. But, as Yale chemist John C. Tully said in an accompanying commentary in Science, "Molecules have not cooperated."
When a molecule is heated up, the weakest bond breaks first. Attempts to tune lasers to break stronger bonds have been thwarted repeatedly by the rapidity with which molecules convert the light energy into thermal energy. Describing the new findings as a "striking contrast" to previous studies, Tully said the researchers have "successfully accomplished a long-standing goal."
The research was carried out at Vanderbilt’s W. M. Keck Free-electron Laser Center. The free-electron laser is a special kind of laser which has the advantage that its beam can be tuned through a wide range of frequencies in much the same way that different frequencies can be dialed on a radio. Most lasers produce light only in a few distinct frequencies. The Vanderbilt FEL operates in the infrared portion of the spectrum, which is particularly valuable for probing the structure and behavior of materials. This allowed the researchers to use laser light tuned to the frequency at which the hydrogen-silicon bonds vibrate and polarized so the photon’s electrical field is pointed in the same direction as the silicon-hydrogen bonds.
In addition to applying this basic system to silicon surfaces covered only with hydrogen, they also tested it on surfaces covered with a mixture of hydrogen and its isotope deuterium. The researchers found that the technique can remove hydrogen atoms while leaving the deuterium atoms intact.
This degree of selectivity could provide a way to control the growth of nanoscale structures with an unprecedented degree of precision, and it is this potential that most excites Cohen, who said, "By selectively removing the hydrogen atoms from the ends of nanowires, we should be able to control and direct their growth, which currently is a random process."
Feldman, a professor of physics at Vanderbilt, said the process represents a significant advance in the ability to modify the surfaces of materials at the atomic level. "We have a new way to selectively interrogate and modify surfaces," he said. "If you stop to think about it, surfaces are where the action is. It is where the rubber meets the road. So, not only will this new technique allow us to create innovative new devices, it will also provide us with invaluable new knowledge about vital surface processes. In fact, some of the most advanced nanotechnology devices that have been envisioned, like quantum computers, require the level of control that atom-specific processes of this sort make possible."
Zhiheng Liu is a post-doctoral fellow at Vanderbilt, and Zhenyu Zhang is a condensed matter theorist at Oak Ridge National Laboratory and the University of Tennessee.
The project was supported by grants from the Department of Energy, the Defense Advanced Research Projects Agency and the National Science Foundation.
For more information, visit: www.nsf.gov