A hybrid sensor technology combines the best attributes of CCDs and CMOS technology
Dr. Antoinette O’Grady, Princeton Instruments/Acton
Spectroscopists continue to push the boundaries of technology as they seek to learn more about life’s building blocks. From understanding how the human body works in minute detail to discovering and analyzing new planets, the limits of current instrumentation and scientific detectors are constantly challenged.
One key area of modern-day research is single-molecule detection, which pushes technology to its absolute limit. Recent technical advances in detectors and data manipulation have made single-molecule detection a reality and have resulted in a growing interest in associated spectroscopic and imaging methods.
The interest in single molecules spans many scientific disciplines, from cell biology to materials science. In cell biology, the study of single molecules in their natural physiological environment enables understanding of their behavior and interactions within a living cell. This allows researchers to understand and manipulate the behavior and mechanistic involvement of biochemically relevant cellular components.
Until recently, such experiments were limited to bulk, or “ensemble,” samples that averaged information from many molecules. This provided limited information and, thus, limited understanding of individual molecular properties. Single-molecule techniques also are used in materials sciences because changes in the vibrational structure of the molecule reflect transformations in its chemical and physical environments. Experiments in this area can reveal the mechanical properties of the material surrounding the molecule. For example, the method can be used to study complex systems — such as condensed matter — that have heterogeneous properties because of local environments or conformational states.
Although fluorescence imaging and emission techniques allow the researcher to gather a lot of interesting information, spectroscopic analysis can provide additional information for single-molecule and more conventional ensemble experiments. Biological systems and physical materials absorb, emit and scatter light in a way that gives rise to distinct spectral fingerprints. These fingerprints can help researchers distinguish between healthy and diseased species, track changes within cells, and identify the location, concentration and interaction of intracellular components.
For example, surface-enhanced Raman spectroscopy has been used recently for single-molecule research. Enhanced Raman spectra of single molecules and nanoparticles adsorbed on silver colloids have been recorded. The method has provided a large enhancement factor and a signal much more intense and stable than single-molecule fluorescence.
All single-molecule experiments require a high detection limit and a fast spectral rate. Most biological systems are prone to photodegradation, so experiments must be done before this occurs. Because many interactions are extremely fast, sensitivity and spectral acquisition speed are of the essence.
Some traditional spectroscopy applications, such as pump-probe techniques, suffer from the constraints of speed and detection limits in current detector technology. Coherent anti-Stokes Raman spectroscopy (CARS) is a pump-probe technique that can be used to monitor vibrational dynamics in systems of biological interest in gaseous, liquid and solid states, but it can take hours to acquire the necessary data.
In this method, two simultaneous femtosecond pulses — a pump and a Stokes pulse — generate coherent vibrations in a system. Then, using a variety of set time delays, a third pulse with the same wavelength as the pump pulse monitors the generated molecular dynamic and generates an anti-Stokes signal. A CCD records many broad CARS spectra in the anti-Stokes region for each time-delay setting. Fourier transform of the measured transients in these thousands of acquired spectra allows a high spectral resolution, even though very broad spectral pulses have been used for their generation. Thus, a detector that would enable shorter exposures and faster readout times, combined with a high detection limit, would be of great benefit in reducing the overall experimental acquisition time.
These are just some applications that would benefit. Any investigation of the kinetics of transient events that requires an extremely high sustainable spectral rate would also profit from a scientific spectroscopy detector that can deliver thousands of spectra per second while maintaining high sensitivity.
Trade-offs
Today’s spectroscopy CCD detectors have excellent sensitivity for low-light applications. They have 100 percent fill factors and good spectral resolution, they are quantitative in nature, and back-illuminated models have quantum efficiencies of up to 95 percent. Their main limitation is that the readout noise rises with increased readout speed, limiting the detection capability at faster readout speeds. Current detectors offer continuous acquisition of hundreds of spectra per second at megahertz speeds with relatively low readout noise and with extremely good detection limits (and up to a 1000 spectra per second by utilizing subregions of the CCD).
However, some applications require faster spectral rates combined with high sensitivity. Electron multiplying CCDs maintain high sensitivity at high readout rates by amplifying the signal over the readout noise. They are most beneficial for overcoming noise when operated at high readout rates and are not as helpful when used at rates of only a couple of megahertz. Their sensitivity is limited by their inherent noise sources, and they are not truly quantitative detectors.
The ideal spectroscopy detector is a true quantitative device that combines high readout speeds with real subelectron readout speeds and high sensitivity.
Princeton Instruments/Acton has been working on what it calls hybrid sensor technology, which combines the benefits of CCDs and CMOS detectors. It is being used to develop a dedicated spectroscopy detector that can exhibit a quantum efficiency of up to 95 percent while maintaining actual readout noise below 1 electron at readout speeds of more than 1000 spectra per second.
As is well-known, in a conventional CCD, photons are converted to electrons and stored in a 2-D pixel array. The charge in each pixel is shifted vertically in a controlled manner through vertical shift registers into a horizontal shift register. In spectroscopy applications, the array is essentially divided into columns, and all the charge in each column is shifted simultaneously (binned) into the horizontal shift register. The charge from each pixel is then shifted horizontally to an output amplifier, where the charge signal is converted to a voltage signal.
CMOS imagers capture photons in the same manner as do CCDs, but their readout architecture differs significantly. CMOS devices have circuitry at each pixel that makes back illumination impractical. The presence of this circuitry also reduces the fill factor of these devices and limits the overall sensitivity. On the other hand, having electronic circuits at each pixel provides some benefits, such as random access and nondestructive readout.
Another advantage of CMOS is that both analog and digital electronics can be incorporated on the same chip as the detector. The detectors can be designed to operate very fast because their readout and processing electronics can be duplicated many times, with each circuit connected to a subregion of the sensor. As a result, all of the subregions with their associated electronic circuits can be operated in parallel to attain very high frame rates.
Hybrid detector
Hybrid sensor technology combines the quantum efficiency benefits of CCD devices with the analog and digital signal processing capabilities of CMOS devices. The CCD is used to collect and convert photons into electrons in potential wells, in the same manner as conventional CCDs. It can be either front- or back-illuminated, providing much higher sensitivities than traditional CMOS devices. The charge from each pixel is transferred through vertical and then horizontal shift registers, as with a conventional CCD. However, this is where the similarities end.
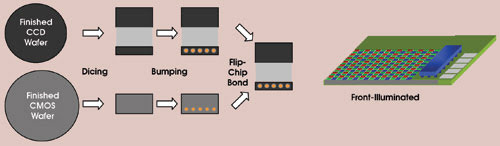
Figure 1. A bump-bonding technique bonds CMOS processors to CCD sensors.
Patented technology eliminates the need for the CCD to have an on-chip amplifier; instead, the charge is transferred through a unique “bump-bonding” technique from the CCD silicon to a CMOS silicon chip (Figure 1). Once transferred, the charge is amplified by a very low noise charge amplifier. The CMOS amplifier is operated at kilohertz speeds to produce very low noise. However, because the CCD can be divided into many subregions, each with its own output connected to a separate CMOS amplifier circuit (all these outputs can be operated in parallel), the net effect provides an extremely high net charge throughput rate (Figure 2). A proprietary sampling technique reduces the amplifier noise even further and confers subelectron readout noise to the device.
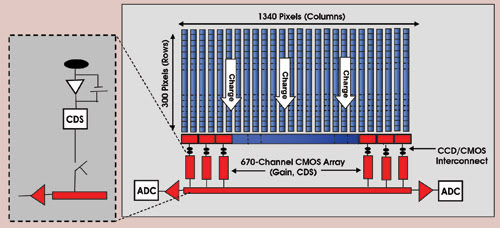
Figure 2. This schematic shows the parts of a spectroscopy hybrid sensor technology detector (ADC = analog-to-digital converter, CDS = correlated double sampling).
Current spectroscopy detectors provide excellent detection limits and sensitivity and are more than adequate in terms of spectral rates for the majority of applications. However, a growing number of applications, particularly in the area of single-molecule spectroscopy, CARS and time-resolved spectroscopy of transient events, could benefit from a spectroscopy detector with the high sensitivity of traditional CCDs combined with high spectral rates and unparalleled detection limits.
Hybrid CCD/CMOS technology meets the needs of these applications and provides researchers with the technology they need for breaking barriers and achieving scientific breakthroughs.
Meet the author
Antoinette O’Grady is a chief scientist at Princeton Instruments/Acton in New Jersey; e-mail: [email protected].