Hyperspectral imaging has evolved from a novel technique into a useful imaging method.
Alexandre Y. Fong and Elliot Wachman, ChromoDynamics Inc.
Multispectral and hyperspectral imaging techniques were developed for applications in remote sensing and astronomy but have slowly transitioned into use in many areas of the life sciences, including fluorescence microscopy and in vivo imaging. Unlike images taken with standard color (RGB) cameras, multispectral imaging information is not discernible to the human eye. The spectral information can be used for such diverse applications as food production quality assurance and monitoring of crop health.
In multispectral imaging, a series of images is acquired at many wavelengths, producing an “image cube.” Figure 1 illustrates the acquisition of images at individual bandpasses indexed by wavelength and compiled to provide spectral absorbance profiles for determination of oxygen saturation through a cranial window in a mouse.
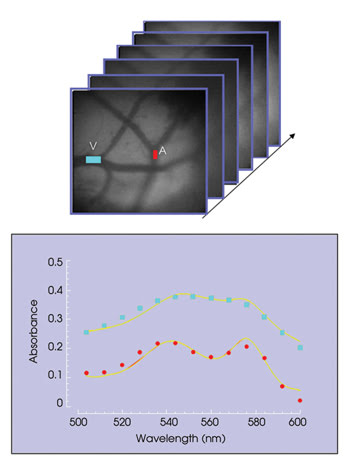
Figure 1. This image cube shows bandpasses indexed by wavelength and compiled to provide spectral absorbance profiles for determination of oxygen saturation through a cranial window in a mouse.
Increasingly, life sciences imaging involves simultaneous monitoring of multicolor labels such as fluorophores or quantum dots. Hyperspectral imaging enables the differentiation and quantitation of these labels and provides the basis for studies of not only cellular components but also of entire systems. Developments in nonlinear optics have introduced faster and more flexible, sensitive and precise hyperspectral systems, which will provide not only new insight for researchers but also more timely and accurate diagnostic tools for clinical use.
A typical wide-field fluorescence microscope is composed of an excitation source (often filtered), a sample stage, an objective magnification lens, a dielectric bandpass or “barrier” filter, a dichroic filter, a final magnification lens and an imaging detection plane. In its most basic form, multispectral imaging can be performed with a set of interference filters and a mechanical filter wheel incorporated into the optical imaging path of a microscope (Figure 2a). The sample can be a multilabeled slide, illuminated by an excitation source such as a mercury vapor lamp. The filter wheel’s output at each spectral bandpass is detected by a single or multielement detector, such as a CCD, and is collected via a computer. This approach confines the user to a limited number of fixed-wavelength choices — requiring knowledge of the sample’s spectral emission characteristics — and is mechanically restricted to changing wavelengths in ~50 ms.
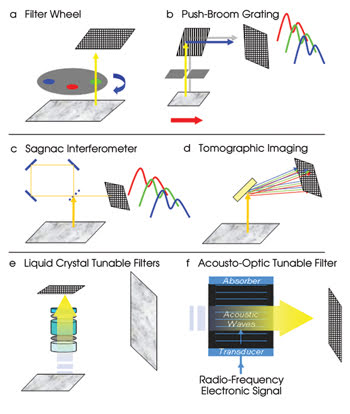
Figure 2. There are many methods for obtaining hyperspectral images.
Hyperspectral techniques
Many hyperspectral imaging systems employ a dispersive element such as a prism or a grating (transmissive or reflective). Grating or prism techniques often are referred to as “push broom” spectral imagers because the entire image is collected line by line by precise motion of the sample across the instrument’s optical path. By either physically moving the sample or by directing the beam and detector field of view, a spectral record of the entire sample image can be collected (Figure 2b).
The push-broom technique collects entire spectral profiles of each pixel of the sample image, which can be time-consuming in collection and in process and storage, but it has benefits in terms of data postprocessing. Such systems are limited to static or relatively slowly changing phenomena because they are restricted by the speed of motion of the mechanical stages used in moving the sample through the scan. Depending upon the size of the sample, this can range from seconds to minutes.
In Fourier transform spectroscopy, the entire image is captured using a Sagnac interferometer, and a spectral profile is constructed. This system provides excellent spectral resolution across the visible range (Figure 2c).
The technique, however, requires mechanical movement of one of the interferometer arms, and data processing requirements are considerable. Hence, its application to fast-moving dynamic phenomena is limited, and the acquired data is not immediately available for viewing.
Tomographic imaging is another approach to spectral imaging (Figure 2d). In this technique, the scene is dispersed off a diffraction grating or rotating prism, for example, and multiple orders of the spectrally distributed image are collected at once. The advantage is that full spatial and spectral information can be acquired at one time. However, spatial resolution is limited (because the CCD chip must capture all order images), and there are significant processing requirements, as with Fourier transform spectroscopy.
Liquid crystal tunable filters, also known as Lyot filters, are created using alternating layers and thicknesses of birefringent crystal and liquid crystal polarizers, which can be used to cull various bandpasses from a source spectrum, as shown in Figure 2e.
Lyot filters are band-sequential scanning systems with tuning capabilities from the visible through the infrared. Although they can be constructed with various bandwidths, once a bandwidth is fabricated, it cannot be changed. Wavelength switching times are on the order of 100 to 150 ms, and transmission throughput can be quite low, particularly in the blue and for narrower bandwidths.
Acousto-optic tunable filters (AOTFs) are crystals with optical properties that can be controlled by sending sound waves through them (Figure 2f). The sound waves are generated by applying a radio-frequency electrical signal to a specially designed transducer that is bonded to the crystal.
With these filters, both wavelength and bandwidth can be controlled electronically. As with tomographic imagers and liquid crystal tunable filters, there are no moving parts. Wavelength switching speeds are faster than other band sequential technologies, with random access selection in ~50 μs. Although AOTFs were introduced more than 30 years ago, their use has been limited because of difficulties in obtaining good image quality. This has been addressed recently, however, through the use of innovative transducer designs and long-interaction-length crystals.
Hyperspectral imaging techniques can be applied to conventionally stained slides viewed in transmission using a light microscope, and they have shown promise in detecting staining alterations (metachromasia) resulting from cancer-related changes in a cell’s macromolecular components and in quantifying light-absorbing chromophores (hemoglobin, bilirubin, etc.). Clinically, such approaches can be used to confirm and quantify the presence of abnormal tissue on a conventional pathology slide, to resolve “look-alike” questions or to clarify tumor margins in surgical pathology (Figure 3).
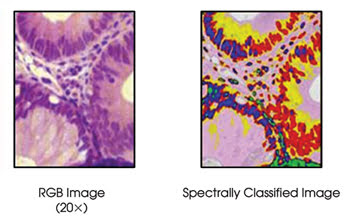
Figure 3. Spectral imaging can be used to detect metachromasia in conventionally prepared samples, as seen in these images of colon tissue treated with hematoxylin and eosin stain. Blue = normal nuclei; green = lymphocytes; pink = normal cytoplasm; red = abnormal nuclei; yellow = abnormal cytoplasm.
One of the most exciting areas for hyperspectral imaging is multicolor fluorescence imaging of cells and tissues, in which it has been applied to a number of research techniques:
• More colors on a single slide.
• Multicolor fluorescence in situ hybridization.
• Imaging of dynamic events within the neuron.
• Ratio imaging.
• Förster resonance energy transfer.
• Lifetime imaging microscopy.
• Spectral karyotyping.
With conventional multichroic interference filters, four fluorescence probes can be detected in a single cell for multi-parameter studies. As probes are added, overlap of the probe emission spectra makes distinguishing them very difficult. By using spectral imaging, multiple overlapping probes can be distinguished within a single filter passband (Figure 4). The figure illustrates this technique using an AOTF spectral imaging device to distinguish three closely spaced green fluorescence dyes in mouse endothelioma.
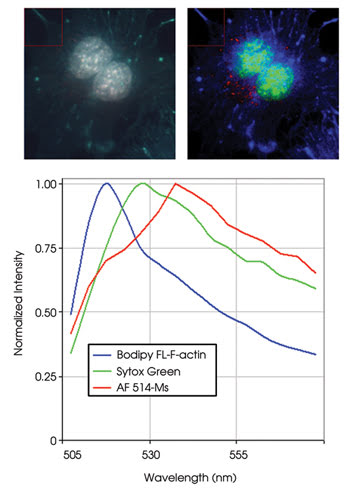
Figure 4. An acousto-optic tunable filter (AOTF) spectral imaging device distinguishes three closely spaced green fluorescence dyes in mouse endothelioma. On the left is the RGB image, and on the right is the classified AOTF image. The graph shows the quantitative spectral data for the fluorescence dyes. Courtesy of Molecular Expressions.
Unlike a color camera, spectral imaging provides detailed color information throughout the spectrum. By using techniques such as linear pixel unmixing, quantitative information can be obtained even when there is strong colocalization of the various probes. This technique also has been used to eliminate autofluorescence background from fluorescence samples.
Next steps
Hyperspectral technologies are becoming integrated with other life sciences imaging tools, including bright-field, fluorescence and confocal microscopes, and macroimaging setups. Spectral imagers can be used with other techniques to provide information not available with conventional microscopy and, in particular, to increase the number of imageable probes within a single sample.
Of specific interest for drug development are in vivo techniques, which permit real-time visualization of the takeup and effectiveness of therapeutic agents. Investigators must select from among the technology options for hyper- and multispectral imaging those that most closely address the event or phenomenon being observed. In the clinical sphere, automated imaging platforms incorporating conventional optical microscopes with hyperspectral imaging systems and intelligent software have the potential to transform diagnostic medicine as more and more diagnostically and therapeutically relevant targets are identified.
Numerous other applications are being investigated, and it is likely that their successful development and implementation will require interdisciplinary efforts involving physicists, engineers, life scientists and physicians.
Meet the authors
Alexandre Y. Fong is vice president of marketing and sales, and Elliot Wachman is vice president of research and development, both at ChromoDynamics Inc. in Orlando, Fla.; e-mail: [email protected].