A new paradigm in spectral encoding extends the multiplexing capabilities of flow cytometry for cell and molecular analysis.
Dr. John Nolan, La Jolla Bioengineering Institute
In flow cytometry, which performs high-speed optical analysis of single cells, suspensions of cells are hydrodynamically focused single file through a laser beam (Figure 1). The light scatter provides information about gross morphological features (size and granularity), and fluorescence from labeled antibodies or other probes reports molecular features of cells that provide information about cell identity and function.
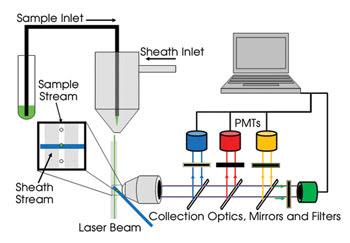
Figure 1. Conventional commercial flow cytometers measure light from cells in discrete wavelength ranges through dichroic mirrors and bandpass filters with photomultiplier tubes (PMTs). Four or five colors of fluorescence can thus be measured simultaneously using a single laser. Multilaser systems extend this approach to enable the measurement of nearly 20 different fluorescence labels simultaneously.
Over the past two decades, the number of fluorescence signals that can be measured simultaneously has increased from two or three to nearly 20, thanks to the use of multiple lasers and detectors. This expansion in measurement capabilities has revolutionized our understanding of the immune system and has enabled new applications for flow cytometry in cell and molecular analysis.
Further expansion of the number of parameters measurable would enhance these efforts, but the spectral features of available fluorescence probes, light sources and detectors are a limiting factor. Because the emission spectra of commonly available small organic fluorophores are relatively broad (50 to 75 nm full width half maximum), only four or five probes can be measured with widely used 488-nm lasers and photomultiplier tubes (Figure 2). Adding 405-nm and 633-nm lasers can increase the number to nine to 12 probes. Semiconductor quantum dots, with their slightly narrower emission spectra and broad absorption bands, enable as many as 17 fluorescence probes to be analyzed simultaneously. Expanding the usable electromagnetic spectrum beyond the visible range will require the adaptation and development of new light sources, detectors, optics and probes. Or, alternatively, the currently accessible region of the spectrum could be used more efficiently, and Raman spectroscopy is one way to accomplish this.
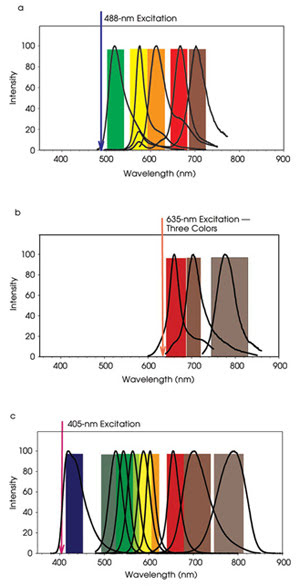
Figure 2. In a conventional benchtop flow cytometer, four or five different fluorescence probes can be measured with 488-nm excitation. The emission spectra of FITC, PE, PE-Texas Red, PE-Cy5, and PE-Cy7 are shown as an example (a). The addition of excitation from a red laser (635 nm), enables the detection of additional fluorophores, such as allophycocyanine (APC), APC-AlexaFluor660, and APC-AlexaFluor750 (b). Semiconductor quantum dots are efficiently excited by a violet laser (405 nm) and have been used to expand the total number of fluorophores measured to 17. For example, the emission spectra of Cascade Blue, QD525, QD545, QD565, QD585, QD605, QD655, QD705 are shown in (c).
Bright signals
Raman scattering results from the inelastic interaction of light with matter, producing an optical spectrum that is characterized by narrow linewidths corresponding to specific chemical bonds. The technique is attractive because it produces much more information from a small region of the visible spectrum, providing the potential for discriminating among many molecular probes.
Although intrinsic Raman scattering is an inefficient process that produces low signal intensities, several variants of the technique enhance the Raman scattering signals, providing stronger signals that, in some cases, rival the brightness of fluorescent probes. Surface-enhanced Raman scattering (SERS) has attracted attention for sensitive detection applications. Metal nanoparticle-based SERS probes are being developed that exhibit bright and characteristic Raman spectra, demonstrated in several bulk- and image-based detection applications.
To exploit the potential of Raman scattering-based detection, we developed a flow cytometer that rapidly measures Raman scattering spectra from a single particle.1 From a flow cytometry standpoint, the key challenge is to provide spectral resolution that is much higher than is traditionally achieved using optical filters and discrete detectors such as photomultiplier tubes. From a Raman spectroscopy perspective, the primary challenge is to perform these measurements on submillisecond time scales at rates of hundreds of particles per second.
The flow cytometry and Raman spectroscopy challenges were addressed by using high-efficiency light-collection optics, a Kaiser Optical Systems high-throughput imaging spectrometer and an Andor back-thinned electron-multiplied CCD camera triggered by detection of Rayleigh light scatter from single particles on a Thorlabs photodiode. The resulting instrument shares design features with instruments previously used to measure fluorescence spectra in flow (Figure 3).2,3 The instrument can measure Raman spectra from particles bearing as few as approximately 200 Raman probes and can make measurements with integration times as short as 100 μs. Results with the instrument show that it can detect more probes in a spectral range than traditional systems can.
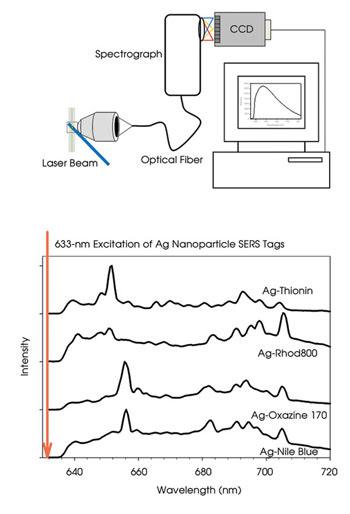
Figure 3. Spectral flow cytometers (top) often use a grating to disperse collected light across an array detector such as a CCD, thus measuring the spectrum continuously with higher spectral resolution than conventional flow cytometers. This allows the relatively narrow spectral features of SERS-based reporter tags to be identified (bottom), enabling more labels to be distinguished within a given region of spectral space.
Apart from requiring relatively little spectral “space,” SERS nanoparticles can be engineered to exhibit a further enhancement, called resonance Raman, at selected wavelengths. This makes the prospect of developing dual fluorescence/Raman instruments quite feasible. The development of robust reagents is an important challenge. Unlike small organic fluorophores or fluorescent proteins, which can be prepared in high purity, nanoparticle-based SERS systems tend to be relatively heterogeneous. Researchers have made SERS systems reproducibly, but the process requires special care.4,5 Thus, engineering improved nanoparticle SERS systems remains an important objective.
The analysis of Raman spectra from thousands of particles presents some new challenges for flow cytometry data analysis. Although conventional flow cytometry experiments typically involve fewer than 20 parameters, each corresponding to a single fluorescence probe, the Raman flow cytometry data involves more than 200 parameters, representing both conventional flow cytometry probes and the spectral data.
The current flow cytometry standard (FCS) data file format can accommodate these large data sets, but commercial flow cytometry software is not well suited to the analyses commonly used in spectroscopy and multispectral analysis. We used a combination of available flow cytometry data analysis software and multivariate statistical tools to perform classification based on principal components analysis as well as spectral unmixing based on a linear least-squares algorithm.
The integration of these capabilities into flow cytometry data analysis software will be an important next step to making Raman flow cytometry a practical tool for biological researchers.
The Raman Flow Cytometry project is funded by a Bioengineering Research Partnership grant to La Jolla Bioengineering Institute from the National Institute of Bioimaging and Bioengineering of the National Institutes of Health and involves collaborators at the National Flow Cytometry Resource at Los Alamos National Laboratory and at the National Institute for Nanotechnology at the University of Alberta in Edmonton, Canada.
Meet the author
John Nolan is a professor at La Jolla Bioengineering Institute in California; e-mail: [email protected].
References
1. D.A.Watson et al (February 2008). A flow cytometer for the measurement of Raman spectra. CYTOMETRY A, Vol. 73, pp. 119-128.
2. G. Goddard et al (Aug. 1, 2006). Single particle high resolution spectral analysis flow cytometry. CYTOMETRY A, Vol. 69, pp. 842-851.
3. J.P. Robinson (April 2005). Multispectral cytometry of single bio-particles using a 32-channel detector. PROC SPIE, INT SOC OPT ENG, Vol. 5692, pp. 359-365.
4. L.O. Brown and S.K. Doorn (March 18, 2008). A controlled and reproducible pathway to dye-tagged, encapsulated silver nanoparticles as substrates for SERS multiplexing. LANGMUIR, pp. 2277 -2280.
5. L.O. Brown and S.K. Doorn (March 4, 2008). Optimization of the preparation of glass-coated, dye-tagged metal nanoparticles as SERS substrates. LANGMUIR, pp. 2178-2185.