Nanoscale antennae improve optical imaging, nanostoves speed up DNA analysis, and nanocontainers could deliver drugs and imaging agents.
David Shenkenberg, Associate News Editor
In the recent past, biology has shifted from the microscale to the nanoscale, from the level of the cell to that of viruses and of cellular constituents such as proteins, nucleic acids and most organelles. By extension, tools for the investigation of these constituents and viruses must operate on the same scale. Therefore, scientists have turned to nanotechnology for imaging biological processes, for analyzing biological materials and for fighting disease.
In many cases, scientists use light to stimulate or to characterize the nanoparticles employed for these applications. Some have used nanoparticles of elements such as gold or iron for biomedical imaging. Gold nanoparticles also have been heated with a laser to destroy cancer cells. Those of semiconductors, such as cadmium selenide, can serve as fluorophores with superior brightness and lifetimes. Coating chemotherapy agents with silica can reduce the exposure of healthy cells in the human body. Such coatings often are characterized by some form of spectroscopy to ensure uniformity and the ability to release the drug.
Nanoantennae
In another example of the utilization of gold nanoparticles, researchers from the University of Rochester in New York have used a gold nanoparticle on the tip of an optical fiber as an antenna to enhance the fluorescence of labeled proteins in the cell membrane.
Without enhancement, fluorescence microscopy cannot resolve anything below ∼200 nm because of the diffraction of light waves. Therefore, nucleic acids, most proteins and viruses either cannot be detected or appear as indiscriminate blobs. The resolution of fluorescence microscopy has been improved in various ways. The highest resolutions have been achieved by turning photoswitchable probes on and off and reconstructing an image, but this requires acquisition times longer than most biological processes. In contrast, the nanoantenna increases the resolution of fluorescence microscopy in real time.
Alternatively, atomic force microscopy can profile the surfaces of living cells with atomic resolution by contact with a metal probe. When the probe is chemically coated, it can yield chemical information. However, this technique typically cannot achieve the chemical sensitivity and specificity of fluorescence microscopy. For example, it can characterize proteins loosely as hydrophobic or hydrophilic, whereas fluorescent labels can tag specific proteins. Because the nanoantenna works with fluorescence microscopy, it can deliver precise chemical information and reveal surface topology, said principal investigator Lukas Novotny.
He explained that the nanoantenna used by his research group works similarly to a conventional antenna that transmits and receives radio waves. It receives incident laser radiation and localizes it onto each protein in a cell membrane, one at a time. Likewise, the antenna helps to release and direct fluorescence emitted by the fluorescent labels.
As reported in the February issue of Nano Letters, the researchers used the technique to study proteins in the membranes of red blood cells that transport calcium and that hydrolyze ATP.
For this purpose, the antenna was attached to a quartz piezoelectric tuning fork that was placed over the cell membrane of one blood cell on the stage of a Nikon inverted microscope (Figure 1). The antenna was raster-scanned over the membrane surface, remaining close to the surface as it did so. The tuning fork controlled the antenna’s movement in a manner analogous to tapping-mode atomic force microscopy: It measured the resonance shift as the antenna moved and adjusted the antenna’s position accordingly.
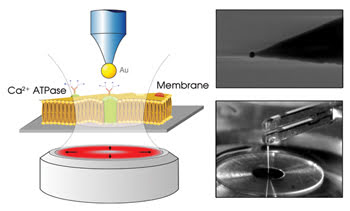
Figure 1. The nanoantenna in the electron micrograph (upper right) consists of a gold nanoparticle attached to an optical fiber. The scheme (left) and photograph (bottom right) show that this antenna is attached to a quartz-crystal piezoelectric tuning fork positioned over an inverted microscope. Reprinted with permission of Nano Letters.
The researchers excited the antenna with a Thorlabs 633-nm HeNe laser, a wavelength chosen according to an antenna scaling law, which states that the excitation wavelength must be redshifted with respect to the plasmon resonance of the gold nanoparticle. The radially polarized excitation laser beam was focused by a high-numerical-aperture oil-immersion objective, crucial for focusing the radiation onto the antenna, according to Novotny. The resulting fluorescence was collected by the same objective and measured with a PerkinElmer single-photon-count-ing avalanche photodiode.
With the antenna, the investigators achieved an ∼eightfold more intense fluorescence signal than without it and a resolution of ~50 nm (Figure 2). Each fluorescence spot corresponded to a single protein molecule. The antenna reduced photobleaching and enabled the use of intensities 10 times lower than without the antenna.

Figure 2. The middle panel is a higher-magnification portion of the image in the left panel, which is blurry because the nanoantenna was not used. On the right, the nanoantenna was used to achieve a much clearer image of the same area. Reprinted with permission of Nano Letters.
Changing the antenna geometry and materials could enable 10-nm resolution. Novotny said that the investigators plan to experiment with elongated particles similar to 10-nm-diameter gold or silver nanorods, which will behave like classical dipole antennae. He also said that they will attempt to distinguish among the various forms of the membrane protein. In parallel research, the scientists are investigating adhesion proteins in endothelial cell membranes.
As it turns out, gold nanoparticles can act not only as antennae, but also as stoves. A team of researchers led by Jochen Feldmann from Ludwig Maximillians Universität in Munich, Germany, has used gold nanoparticles as stoves to heat DNA during melting analysis.
Nanostoves
The bases that comprise DNA as well as the degree to which the strand sequences match can influence how easily two strands separate, or “melt,” during heating. Thus, melting curves can reveal information about the content of the DNA under study.
Thomas A. Klar, a professor on the team that developed the DNA melting assay, said that these melting curves have broad applications, from biotechnology to medicine. For example, a melting curve of genetic material from blood samples can determine the specific type of pathogen in an infected patient’s bloodstream.
The novel technique works in less than 1 ms, as compared with up to one hour for the conventional method of using a thermal cycler — a tabletop machine that contains a heat block — to warm test tubes containing DNA in solution. “Within a short time frame, you can have a huge number of tests,” Klar said.
To perform the assay, the research-ers attached template DNA to the surface of the gold nanoparticles using a thiol (-SH) linker. When a complementary sequence of DNA binds to the template strand, the gold nanoparticles aggregate, causing a broadening and a redshift of the extinction spectra of the surface plasmons. Using a laser to heat a gold nanoparticle, in turn, heats the DNA and causes the strands to separate, resulting in a reversal of these spectral changes (Figure 3). The changes show how fast the DNA separates.
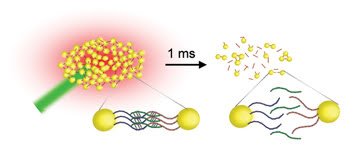
Figure 3. Laser heating separated not only the DNA but also the gold nanoparticles. The nanoparticle separation resulted in measurable spectral shifts. Courtesy of Ludwig Maximillians Universität Munich.
To heat the stoves, the researchers used a single 300-ns pulse from a Spectra-Physics 527-nm Nd:YLF laser. Then for spectroscopy, they used a 650-nm continuous-wave laser as the probe beam and detected the surface plasmons with a Thorlabs photodiode connected to a 1-GHz LeCroy oscilloscope, which provided the readout. The detector had a temporal resolution of 20 ns, which Klar said was important for performing the assay quickly.
The technique enabled the investigators to heat the DNA gently and reversibly. “We can adjust the temperature in steps of less than one degree,” Klar said. It also could distinguish between a perfectly matching strand of DNA and a strand containing a single mutation in less than 1 ms, even with a 1:1 mixture. Klar said that the conventional method cannot analyze mixtures so easily. These results are detailed in the February issue of Nano Letters.
The scientists plan to test more complex mixtures and to develop this technique into a kit or an instrument. The kit would contain a gold nanoparticle with the complement of the DNA the user wants to analyze. The user would then mix the sample genetic material with the kit and perform the analysis.
Although gold nanoparticles have proved useful for various purposes, they are toxic to the human body. On the other hand, silica nanoparticles are nontoxic and easily can be modified chemically for targeting tumors or pathogens.
By carrying chemotherapy drugs directly to tumors, silica nanoparticles could minimize the harmful effects of those drugs on the body. “We want to cut their exposure to the human body not by the hundredth, not by the thousandth but by the millionth,” said Sir J. Fraser Stoddart, now a professor at Northwestern University in Evanston, Ill. He and colleagues from the University of California, Los Angeles, have developed silica nanoparticles for this purpose.
Nanocontainers
The research began a few decades ago, when Stoddart’s group discovered movable molecules called rotaxanes. When Stoddart moved from the United Kingdom to Los Angeles, he began a collaboration with Jeffrey I. Zink, whose laboratory at UCLA had developed a solid support that could serve as a base for these moving molecules. Together, their groups have developed many silica nanocarriers, activated by light, pH, metal ions and electrochemistry.
Their latest design is a container with a rotaxane-containing snap-top lid that opens upon exposure to a specific enzyme. Zink said that, because certain tumors overexpress certain enzymes, the silica nanoparticles could travel to a tumor overexpressing an enzyme that opens the lid, releasing an anticancer drug right inside the tumor. He also said that nanoparticles of a certain size range have been shown to be absorbed at a greater rate by tumors than by normal tissue. Stoddart and Zink believe that their design is different from that of other silica nanoparticle carriers because of the snap-top lid and the precise control over it.
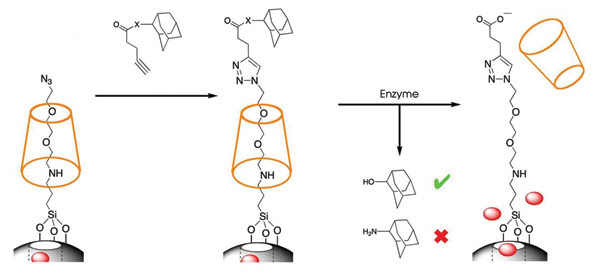
Figure 4. An enzyme opened the snap-top lids of the nanocontainers, releasing the particles inside. Reprinted with permission of the Journal of the American Chemical Society.
In a proof-of-principle experiment, the researchers loaded the nanocontainers with rhodamine and monitored the enzyme-mediated release of the fluorophore by luminescence spectroscopy with a Coherent argon-ion laser at 514 nm for the probe beam and with a Princeton Instruments CCD camera as the detector. They found that the enzyme indeed caused the release of the rhodamine, and that using this type of spectroscopy enabled them to quantify the rhodamine molecules released from the containers over time (Figures 4 and 5). The experiment was published in the Feb. 27 issue of the Journal of the American Chemical Society.
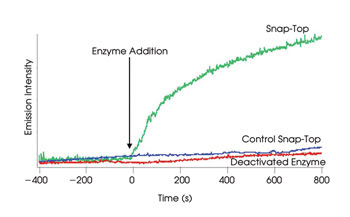
Figure 5. The enzyme released the rhodamine molecules in the nanocontainer but did not have an effect in control experiments with a snap-top lid not responsive for that enzyme or with a deactivated enzyme. Reprinted with permission of the Journal of the American Chemical Society.
Both Zink and Stoddart said that using the nanocontainers to deliver chemotherapy drugs in human patients may take some years to develop. They also are developing the containers for more immediate applications, such as for delivering imaging agents.