Advances in endoscopy technology are helping doctors get more information from images.
Hank Hogan, Contributing Editor
According to modern medicine, there is a grain of truth in the old adage: The way to a man’s heart is through his stomach. Dr. Michael B. Wallace, a gastroenterologist with the Mayo Clinic in Jacksonville, Fla., uses narrow-diameter and flexible endoscopes to reach internal organs and tissues via the gastrointestinal tract. Clinicians and researchers such as Wallace can reach almost any organ by threading these devices through one of the body’s natural orifices.
Such reach is now being combined with imaging techniques that include high-magnification white-light, autofluorescence, narrowband imaging and ultrasound. Doctors can examine patients from inside, take a needle biopsy of areas of concern and, in some cases, even deliver treatment using endoscopes. Other advances in endoscopy include swallowed cameras-in-a-pill that provide pictures of their journey. For those who suffer from knee injuries, advances in rigid endoscopes used for minimally invasive techniques provide their surgeons with a better view.
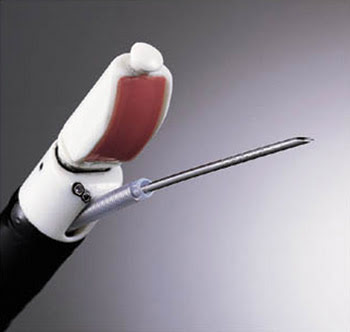
An ultrasound endoscope with a fine needle allows doctors to biopsy tissue deep within patients without the use of surgery. Courtesy of Dr. Michael B. Wallace and Dr. Jorge M.S. Pascual, Mayo Clinic.
There also are some significant advances in microendoscopy, which will be covered in a feature article in the July issue. What follows is a look at endoscopy above the cellular level.
Looking in both directions
Wallace was part of a team that investigated how to better determine the stage of suspected lung cancer. If confined to the lungs, the tumor can be removed surgically. Lymph nodes in the central part of the chest are the most common place for the cancer to spread. Once the disease is there, a combination of radiation and chemotherapy, with or without surgery,is used.
Thus, it is important to know whether the cancer has spread to these lymph nodes, which is traditionally determined via surgical removal of some of the nodes. As with all surgeries, it carries some risk and creates some discomfort. In a less invasive approach, doctors take a fine-needle biopsy of the lymph nodes, using an endoscope to place the needle in the right spot.
Endoscopes equipped with ultrasound can allow clinicians to see ∼5 cm into surrounding tissue with resolution of better than 1 mm. However, because air stops ultrasound and because of the location of the esophagus, doctors can use the technique on only half the chest. That is where advances in pulmonary endoscopes come to the rescue, said Dr. Jorge M.S. Pascual, a pulmonologist at the Mayo Clinic who also was part of the research team. Miniaturization of transducers makes it possible to insert an ultrasound imaging endoscope into the trachea to locate lymph nodes in the part of the chest that cannot be imaged through the esophagus.
As reported in the Feb. 6, 2008, issue of JAMA, the researchers combined the two views to get a complete image. They achieved an estimated sensitivity of 93 percent and a negative predictive value of 97 percent. This was better than either method alone and better than the traditional surgical technique.
The double imaging approach does have drawbacks. It requires, for instance, two specialists, although it may be possible to cross-train one person for both jobs. For now, the Mayo Clinic is using the method, with plans to see if it can be helpful when looking for the recurrence of lung cancer, Pascual said. In the future, he would like to see even smaller ultrasound transducers because the transducer size limits how far into the lungs doctors can go. Of course, further miniaturization still would have to maintain the ultrasound image quality.
Current pulmonary ultrasound endoscopes are 5 to 6 mm in diameter. This is smaller than the 13 to 14 mm of gastrointestinal endoscopes but still larger than the couple of millimeters needed to enter the smallest bronchial passages.
“With more miniaturization, we should be able to reach further in terms of the periphery of the lungs, and be able to detect things that are even smaller than we are detecting right now,” Pascual said.
In a different light
Researchers also are putting other technological advances to work. In a February 2008 GUT paper, a team from the Academic Medical Center in Amsterdam, the Netherlands; from Queen’s Medical Centre in Nottingham, UK; and from the Mayo Clinic looked at the effectiveness of trimodal endoscopic imaging in detecting a precursor to esophageal adenocarcinoma, a disease with a fast rising incidence. If caught early, the cancer is curable. If undetected cancer advances, however, the survival rate drops to less than 15 percent.
Clearly, screening for the precursor is important, but detecting it with standard endoscopy is difficult. Recent advances, however, have given clinicians new tools, including autofluorescence imaging, which exposes tissue to short-wavelength light. This elicits different responses from normal and abnormal tissue. Another advance is narrowband imaging, which squeezes down the normal green and blue spectral light output while increasing the intensity of the blue. Both techniques have shown promise but have drawbacks, such as a high false-positive rate in autofluorescence imaging.
The researchers also investigated how well a combination of imaging techniques detected early neoplasia in Barrett’s esophagus -- the precursor lesion to esophageal adenocarcinoma -- using a new endoscopy system from Olympus of Tokyo. The system combines white-light, autofluorescence and narrowband imaging in one unit. It can magnify up to 100× using white light and narrowband imaging. It has a CCD for white light and narrowband imaging and another CCD for autofluorescence imaging.
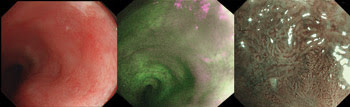
On the left is a white-light endoscopic image of the esophagus, with no clear lesion visible. In the middle is an endoscopic autofluorescence image, obtained by illuminating the area with blue light and capturing the resulting fluorescence from only 500 to 630 nm. Two suspicious areas that appear purple or brown can be seen, at 2 o’clock and 4 o’clock, or about 30° and 60° to the right of the midline. On the right is a narrowband illumination endoscopic image of the first suspicious area. The image shows irregular mucosal and vascular patterns consistent with dysplasia. Courtesy of Dr. Wouter L. Curvers, Academic Medical Center.
Switching between modes takes only a few seconds. One filter wheel enables rendering of white-light video from overlaid red, green and blue images, while another restricts the green to 530- to 550-nm and the blue to 390- to 445-nm narrowband imaging. A second filter system enables fluorescence imaging after blue excitation, with only 500- to 630-nm light reaching the CCD. The unit is not yet available for purchase in the US.
The researchers found that autofluorescence imaging identified all 16 patients who had early neoplasia and who also were picked up by high-resolution endoscopy. What is more, autofluorescence imaging detected an additional 11 patients not identified by the other technique. The false-positive rate of autofluorescence imaging was 81 percent, but using narrowband imaging cut this figure to only 26 percent.
Dr. Wouter L. Curvers, a gastroenterology research fellow at the Academic Medical Center who was part of the research team, predicts that such multimodal endoscopic imaging will become increasingly common and is likely to be combined with techniques that further classify or differentiate suspicious areas. “This will enable a more targeted sampling of the intestine. As we say: ‘Look longer, biopsy less.’”
He noted, however, that the most important tool is a good-quality white-light endoscope that can capture high-resolution and high-definition images, along with proper training. At the same time, he also would welcome a highlighting technique with better resolution and specificity than the current autofluorescence imaging provides.
A narrow view
Another example of the potential usefulness of narrowband imaging comes from a study done at Kyushu University in Fukuoka, Japan. There, a group used magnifying colonoscopy with narrow-band imaging to identify dysplasia in patients who had ulcerative colitis. It reported its results in a November 2007 paper published in Gastrointestinal Endoscopy.
For the study, the researchers used an Olympus colonoscope equipped with a prototype narrowband imaging system. Lead author Dr. Takayuki Matsumoto, a research associate at the university’s Graduate School of Medical Sciences, noted that the researchers used narrowband imaging at 415 and 540 nm because these are the absorption wavelengths for hemoglobin.
With this setup, they examined the large bowel of patients using standard colonoscopy and then narrowband imaging at magnifications of up to 703. They found that the highest incidence of dysplasia was in protruding lesions with a tortuous pattern, which was visible using narrow-band imaging but not when employing standard colonoscopy.
“I was surprised by my observation that I could see flat dysplasia by magnifying narrowband imaging colonoscopy,” Matsumoto said of the results.
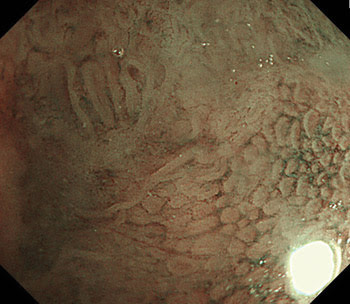
Narrowband endoscopic imaging reveals a tortuous pattern, which could be used as an indicator of dysplasia in patients with ulcerative colitis. Courtesy of Takayuki Matsumoto, Kyushu University. Reprinted from the May 2008 issue of Stomach and Intestine, with permission.
He added that a multicenter study is under way to confirm their findings. It also will serve to test interobserver variations and to allow a comparison of the value of magnifying narrowband imaging colonoscopy with conventional multistep biopsy, and will help study the value of magnifying narrowband imaging for cancer surveillance.
A different angle
While researchers are investigating new methods of endoscopy, companies are working to improve well-established techniques. For example, the privately held manufacturer Avantis Medical Systems Inc. of Sunnyvale, Calif., has developed the Third Eye Retroscope, an auxiliary endoscope system. With a distal tip diameter of only 3.5 mm, the disposable device fits through the instrument channel of the standard endoscope used in a colonoscopy.
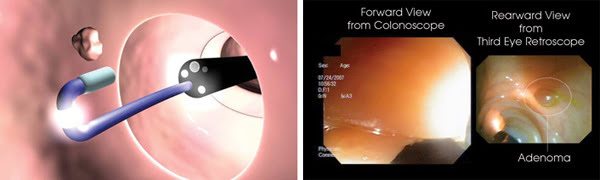
An auxiliary endoscope that provides a reverse view of areas behind folds during a colonoscopy has been developed (left). On the right is an image taken with a colonoscope’s forward view on the left and with the retrograde view on the right. In this case, a polyp behind a fold was hidden from the colonoscope’s view. Courtesy of Avantis Medical.
“Our whole goal is to allow physicians to better visualize the colon and get better surface coverage,” said director of product development Lex Bayer. Once inside the patient, the unit turns around, with an LED light source and a CMOS sensor capturing the rear view.
Because of folds in the colon, some polyps are hidden in shadows and behind tissue so are not visible when examined with only a forward-looking camera and light. Imaging in the reverse direction can reveal the hidden polyps, allowing physicians to see and remove potential precursors to colon cancer.
Tests on anatomic models of colons indicate that as many as 80 percent of hidden polyps are found using retrograde viewing, compared with only 12 percent of the same polyps discovered using standard colonoscopy alone, according to a January 2007 study in Gastrointestinal Endoscopy. A multicenter test of the retrograde-viewing system from Avantis is under way, and initial results were scheduled for release last month. Bayer said the device was available commercially at limited locations beginning in April.
This rearward look requires an appropriately sized sensor and light source that can both fit into a standard instrument channel and yet can provide images of high enough quality. The sensor for the Avantis device comes from Sunnyvale-based OmniVision. According to senior director of marketing Todd Koelling, OmniVision makes only CMOS sensors.
“The key with the medical applications is to make them small, to be as least intrusive as possible,” he said.
Other important attributes include low power, the ability to maintain high image quality in low-light conditions and support for long cabling. The sensor manufacturer achieves this through sensor and pixel design, which also helps ensure that the device is as small as possible.
Historically, the company’s main focus has been on large markets, including cell phone camera and security applications as well as toys and games. Turning endoscopy into a large market requires that the devices be disposable, with a new one used on every patient. For doctors, the benefits of this approach are that there is no need to sterilize instruments and no chance for cross-contamination, an advantage for patients as well.
However, a disposable endoscope must be inexpensive, which places further constraints on the imager. What is more, a disposable endoscope also will require doctors to change their usage model, which may take some time.
Easy to swallow
Flexible endoscopes are not the only areas of innovation and research. Another is capsule endoscopy, where patients swallow a small device. In one version of the technique, the capsule transits the gastrointestinal tract, wirelessly transmitting images of its surroundings. Such capsules must incorporate a light source, an image sensor, a wireless transmitter, associated electronics and, often, a battery, into something small enough to be swallowed. Several commercial versions of these devices exist, with some 10 by 20 mm in size. Capsules are useful for imaging the small intestine, which is difficult to reach with other forms of endoscopy.
However, another type of capsule endoscope has been developed by investigators from the University of Washington in Seattle. They used a single optical fiber connected to a miniature scanner for illumination. Around this, they formed six light-collecting optical fibers within a 6.4-mm-diameter pill that a patient is expected to swallow with a sip of water.
The device is wired, with a 1.4-mm tether trailing behind the capsule. Team leader Eric J. Seibel, a research associate professor of mechanical engineering at the university, volunteered to be the first human to test the device. He reported that it was like swallowing a pill and that the tether presented no problem.
The wire adds some big benefits. For one thing, it allows a physician to control the capsule, backing it up if necessary to review something. More importantly, however, the tether allows the capsule to scan its surroundings. When an electric current flows through the capsule, the scanner moves, and the fiber bounces around so that it sees everything, one pixel at a time.
The key to pulling off this feat lies in how the scan is done and in the microscanner devised by the team. “We have developed a subhalf-millimeter-diameter scanner that scans a circular optical fiber in a spiral pattern using amplitude-controlled resonant scanning,” Seibel explained.
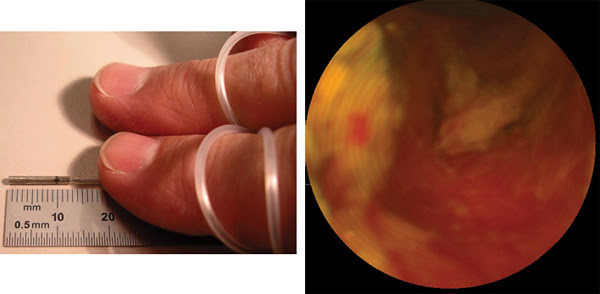
Thanks to a microscanner that rotates an optical fiber around, a capsule-size endoscope (left) images a pig’s bile duct (right) at video rates, capturing the image one pixel at a time. Courtesy of Eric J. Seibel, University of Washington.
While the microscanner rotates the light-collecting fibers, the illumination fiber sequentially is beaming out red, green and blue laser light at 635, 532 and 442 nm, respectively. The light and imaging are synchronized so that a video frame color image is captured, with a resolution of better than 20 μm when zoomed. The field of view is 100°.
The device has been successfully used for in vivo imaging of a pig bile duct, a finding Seibel reported on in a paper presented at SPIE Photonics West in January. Tests showed that the scanner could operate for billions of cycles without breakage.
Pentax Corp. of Tokyo has licensed the technology from the University of Washington, presumably with plans to commercialize it. For his part, Seibel wants to extend the technique from looking to doing. When asked about where he sees the research headed, he replied, “Integrated imaging, diagnosis and therapy in millimeter-sized catheterscopes.”
Higher definition
Finally, there are innovations appearing in rigid endoscopes, the imaging instruments used during laparoscopic or minimally invasive surgery. Such devices typically have an external light source and imaging system, with a rigid tube directing light to the site of the surgery. The same tube collects the reflected light and conducts it to a camera.
As in consumer video, the trend is toward high definition, with 1080i-resolution systems being introduced in the past few years. Gary Pitre is eastern regional sales manager for the imaging systems division of Toshiba America Information Systems of Irvine, Calif. Toshiba does not make laparoscopic systems, but it does make a 1080i high-definition camera that is used in such devices. Pitre reported that, at industry trade shows, high-definition systems are now prevalent, whereas before, they couldn’t be found.
The use of 1080i ups the resolution to 1920 × 1080 pixels, a significant improvement over the 720 × 480 pixels of the old NTSC standard. However, obtaining the best image quality also requires a display with a resolution of 1920 × 1080, Pitre said.
At OneLight Corp. of Vancouver, British Columbia, Canada, President Nick Mac-Kinnon is pushing another rigid endoscope innovation. The company makes digital illumination systems that can be used in a number of areas. One of these could be in rigid endoscopes, which today have very expensive, high-performance imaging systems that require high-quality optics, making the endoscopes expensive. This means that they must be reused, which involves cleaning to prevent cross-contamination. The image quality justifies the drawbacks, however.
Inexpensive sensors exist that could be mounted on the tip of the endoscope nearest the patient, potentially cutting costs and, possibly, making the devices disposable. However, they lack dynamic range. What is more, they face a size-related performance constraint. Sensors small enough to fit on the endoscope tip are monochromatic because that is the only way to get the required resolution in the space available.
These problems could be solved if the light source could be manipulated rapidly enough and with fine enough spectral control. For example, color imaging without the use of a resolution-decreasing filter can be achieved by outputting red light and capturing the response for the red channel, with the same pairing taking place for green and blue. And the dynamic range can be extended several orders of magnitude by adjusting the intensity of the light source, MacKinnon said.
“By combining the image data with the illumination data and creating hyperimage data, then you have a lot more to work with,” he explained. “You can get very accurate color rendition. You can get fairly fast readouts because you don’t have big data sets. By putting the smarts in the box, you’ve now enabled very low cost endoscopes.”
OneLight has just begun shipping the product, with the first systems due out at the end of this month. Thus, there isn’t an endoscope system currently available that puts these ideas to use, but one could soon appear.
Whether or not digital illumination takes off, it is clear that endoscopic advances mean that surgeons and other doctors can now see more than they ever could before. Such information is likely to provide some significant benefits, perhaps proving that it is not just the heart that can be reached through the stomach, but also health.