New donor/acceptor pair improves protease FRET assays.
Cecilia L. Po and Dr. Vera Rakhmanova, AnaSpec Inc.
Proteases are enzymes that cut precursor proteins to generate proteins that can be useful or harmful for an organism. The harmful proteins generated can cause certain diseases including cancer, arthritis, Alzheimer’s, hypertension, hepatitis and AIDS.
Since the discovery of chemical compounds that inhibit HIV protease activity — and thus are effective in treating early-stage HIV infection — much research in the pharmaceutical industry has focused on understanding these enzymes and on screening for drugs that act on them. Assays that provide a continuous reading of activity are homogeneous (don’t require separation of bound from unbound label) and extremely sensitive and can accelerate drug discovery.
Spectrophotometric methods are favored for detecting results from assays because of their speed, accuracy and ease of use. Of assays used with spectrophotometry, fluorogenic ones are generally several orders of magnitude better than the chromogenic type because they have a wide linear dynamic range and offer good reproducibility. In recent years, Förster resonance energy transfer (FRET) assays have found extensive use in high-throughput screening.
Briefly, FRET is the transfer of excited-state energy from a donor to an acceptor, with the donor typically emitting at shorter wavelengths that overlap with the acceptor’s absorption (Figure 1, right). FRET occurs when a donor (fluorophore) and an acceptor (another fluorophore or quencher) are within a specified distance, usually 10 to 100 Å. The distance at which energy transfer equals 50 percent is called the Förster radius. Within this distance, when a donor transfers its resonance energy to an acceptor, a decrease in the donor fluorescence can be detected.
How it works
To use FRET for a protease assay, an enzyme substrate is constructed using a peptide sequence derived from a known protease cleavage site. Upon enzyme hydrolysis, FRET efficiency falls dramatically as the donor-acceptor distance exceeds the Förster radius, resulting in the recovery of the donor’s fluorescence (Figure 1, left).
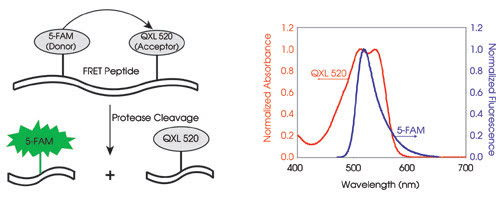
Figure 1. On the left is a schematic representation of a FRET peptide. When 5-FAM and QXL 520 are in proximity, quenching occurs; upon cleavage, fluorescence is recovered and is linearly related to substrate hydrolysis. The absorption spectrum of QXL 520 perfectly overlaps with the emission spectrum of 5-FAM (right).
Recognizing an urgent need for more sensitive protease assays where colorimetric or low-wavelength fluorometric or FRET assays existed, AnaSpec Inc. of San Jose, Calif., developed a series of longer-wavelength FRET assays for proteases. Traditional FRET assays have employed shorter-wavelength pairs. However, their short absorption wavelengths and low extinction coefficients create some limitations for inhibitor screening. To circumvent these limitations, a novel FRET pair — 5-FAM/QXL 520 (excitation = 490 nm, emission = 520 nm) — was used.
Efficient quenching occurs because the absorption spectrum of QXL 520 overlaps with almost the entire emission spectrum of 5-FAM, which results in a decrease in background signal. This quencher is also hydrophilic, rendering the peptide substrates more soluble. Additionally, the green fluorophore 5-FAM is bright and has a high extinction coefficient. Its longer wavelength reduces interference from autofluorescence and absorbance of drug candidates and cellular components.
Ease of use, minimal hands-on time, better signal-to-background ratio and reproducibility make the assays good for high-throughput inhibitor screening or for detecting protease activity. For the HCV NS3/4A protease, the new do-nor/acceptor pair gives a lower detection limit of 10 pM and a Km of 3.2 μM. And using the pair for a renin assay allows detection of only 20 pM of renin, which is 40 times better than an EDANS/DABCYL substrate and 10 times more than an Amp/Dnp substrate (Figure 2).
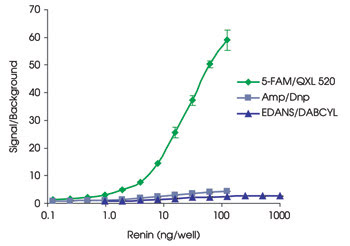
Figure 2. Renin detection is shown using three different FRET substrates.
In addition, 62.5 pM of the human recombinant MMP-7 with a linear range of up to 4 nM has been detected with one of the 5-FAM/QXL 520 substrates. Matrix metalloproteinase (MMP) assays are notorious for problems with cross-recognition of substrates, but they can be made more specific by pulling down a particular MMP by using capture monoclonal antibodies. When this step was used with the long-wavelength assay, sensitivity was enhanced from 100 to 6 pM.
Meet the author
Cecilia L. Po is in the technical support department at Anaspec Inc. in San Jose, Calif.; e-mail: [email protected].
Dr. Vera Rakhmanova is senior scientist at Anaspec.