
Improving the safety of cancer drugs
Novel approach shrinks tumor blood vessels by approximately 70 percent.
Gary Boas
In the early 1990s, David A. Cheresh and colleagues at the University of California, San Diego, in La Jolla observed that a particular protein called integrin anb3 (pronounced alpha-vee-beta-three) is found mostly in blood vessels of tumors. They have published several papers since then suggesting that the abundance of this protein in tumors could be exploited for imaging or therapy. Doctors could use it to identify tumors earlier or to treat patients without as many harmful side effects as conventional chemotherapy produces.
Now the researchers are preparing to demonstrate what they had discussed previously only in theory. They recently received funding from the National Cancer Institute, which had begun a large initiative to support development of nanotechnology for diagnosis and treatment of cancer.
“This prompted us to consider further whether we could take a drug with known toxicity problems and improve upon its anticancer activities while reducing side effects,” Cheresh said. The investigators reasoned that they could combine a known anticancer drug with molecules that target αvβ3 nanoparticles, and that the drug would be safer and would accumulate mostly in tumors.
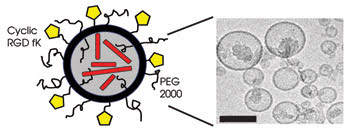
Researchers have described a nanoparticle that can provide up to a fifteenfold improvement in drug delivery efficacy by targeting tumor vasculature expressing integrin αvβ3. Shown here are a schematic (left) and a transmission electron micrograph (right) of the nanoparticle. Cyclic RGDfK 5 targeting peptide. PEG 2000 5 part of the nanoparticle safety coating. Images reprinted with permission of PNAS.
In the July 8, 2008, issue of PNAS, Cheresh and the others reported the design and characterization of a nanoparticle that incorporates molecules of the drug doxorubicin, a safety coating and, on the surface of the coating, peptides that target αvβ3.
Although doxorubicin has been shown to control the spread of both pancreatic and kidney cell cancer in mice, the dosage typically is limited because the drug is toxic to cells. With the new nanoparticles, the researchers demonstrated that they could decrease the level of the drug by as much as fifteenfold while still achieving a strong anticancer effect.
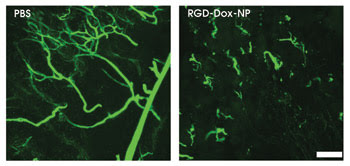
Injecting mice with nanoparticles carrying the cancer drug doxorubicin showed significant reduction in tumor vasculature compared with that in mice treated with the control, PBS, inhibiting angiogenesis by roughly 70 percent (scale bar: 100 μm).
Before assessing the nanoparticles’ potential for drug delivery, they tested them for targeting to the tumor vasculature by injecting mice with melanoma cells and allowing the cells to grow. After one week, they injected both targeted and nontargeted molecules and imaged them as well as the vasculature itself using a Nikon spectral confocal microscope. This confirmed that the αvβ3-targeting molecules accumulated in the tumor vasculature, while the nontargeting nanoparticles did not.
They then tested the efficacy of the nanoparticle with two sets of mice. In one set, they injected pancreatic tumor cells directly into the pancreas, and, in the other set, they injected kidney cancer cells into the left kidney.
After allowing the tumors to grow, they determined the effect of the targeted nanoparticle on tumor blood vessel growth, injecting both sets of mice with doxorubicin-carrying nanoparticles and measuring blood vessel growth after one week by labeling the vasculature with a fluorescein-tagged protein. As anticipated, animals treated with the drug exhibited significant vascular pruning with respect to animals that were not, with blood vessel growth inhibited by approximately 70 percent.
They found that the molecule is much more effective at preventing cancer from spreading than it is at reducing the size of an original tumor, with a fifteenfold increase in antimetastatic activity with respect to animals treated with the free drug. They offered several possible explanations for this effect. First, the blood supply was still being established at the metastatic sites, whereas at the primary tumors, this typically had occurred already. Second, the investigators showed that the doxorubicin-carrying nanoparticles cause cells to die in the αvβ3-expressing tumor blood vessels, which might prevent the tumor from spreading. The cells died by apoptosis, a type of suicide mechanism that cells have when they realize that their machinery has gone awry.
Finally, in the cancer models used in the study, treatment resulting in reduced size of the primary tumor also appeared to lead to less cancer spreading. The researchers noted that further investigation of the antimetastatic effect of the nanoparticles could contribute to the development of other targeting strategies.
Having determined the efficacy of the method in animal models, the researchers are looking toward clinical testing. They are exploring the possibility of partnerships in the private sector. Cheresh offered that the technique likely would be “of interest to companies that have drugs with potency but maybe not the safety profile,” for example. By lowering the dosage needed to be effective, he said, companies might be able to reintroduce drugs that failed initially.
Much of the preclinical testing already has been completed. The next step is to perform Phase I clinical testing, which is done with healthy human volunteers. “If it does as well as it did in animal models, then I think there will be a place for it,” Cheresh continued. “You can imagine that, if you need only a three- or fourfold improvement in drug delivery to achieve significant improvements with respect to toxicity,” then a fifteenfold improvement would be especially welcome.
Published: September 2008