BALTIMORE, Jan. 13, 2009 – Johns Hopkins researchers have invented dust-particle-size devices that can be used to grab and remove living cells from hard-to-reach places without the need for electrical wires, tube or batteries. Instead, these microgrippers are actuated by thermal or biochemical signals.
Mass-producible, they each measure approximately 0.1 mm in diameter. In lab tests, they have been used to perform a biopsylike procedure on animal tissue placed at the end of a narrow tube.

In this optical video snapshot, a tetherless microgripper grabs onto a clump of live L929 animal cells placed at the end of a narrow glass capillary tube. Images courtesy of TG Leong and CL Randall (Gracias Laboratory, Johns Hopkins University).
Although the devices will require further refinement before they can be used in humans, David H. Gracias, who supervised the project, said these thermobiochemically responsive, functional microtools represent a paradigm shift in engineering.
“We’ve demonstrated tiny, inexpensive tools that can be triggered en masse by nontoxic biochemicals,” said Gracias, an assistant professor of chemical and biomolecular engineering in Johns Hopkins’ Whiting School of Engineering. “This is an important first step toward creating a new set of biochemically responsive and perhaps even autonomous micro- and nanoscale surgical tools that could help doctors diagnose illnesses and administer treatment in a more efficient, less invasive way.”
Today, doctors who wish to collect cells or manipulate a bit of tissue inside a patient’s body often use tethered microgrippers connected to thin wires or tubes. But these tethers can make it difficult to navigate the tool through tortuous or hard-to-reach locations. To eliminate this problem, the untethered grippers devised by Gracias’ team contain gold-plated nickel, allowing them to be steered by magnets outside the body.
“With this method, we were able to remotely move the microgrippers a relatively long distance over tissue without getting stuck,” he said. “Additionally, the microgrippers are triggered to close and extricate cells from tissue when exposed to certain biochemicals or biologically relevant temperatures.”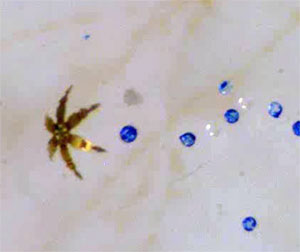
The microgripper can select and pick up specific beads (blue), as shown here.
The microgripper design – six three-jointed digits extended from a central “palm” – resembles a crab. In fact, the joint design actually was inspired by that of arthropod animals. To fabricate the microgrippers in their initial flat position with all digits fully extended, the researchers employ photolithography, the same process used to make computer chips. When the tiny devices are inserted in the body and moved magnetically, the gold-plated nickel in the palm and digits will allow doctors to see and guide the grippers with medical imaging units such as an MRI or CT.
The microgrippers’ grasping ability is rooted in the chemical composition of the joints embedded in the fingerlike digits. These joints contain thin layers of chromium and copper with stress characteristics that would normally cause the digits to curl themselves closed like fingers clasping a baseball. But the researchers added a polymer resin, giving the joints rigidity to keep the fingers from closing.
When the microgrippers arrive at their destination, however, the researchers raise the temperature to 40 °C (or 104 °F, equivalent to a moderate fever in humans). This heat softens the polymer in the joints, causing the fingers to flex shut. The researchers also found an alternative method: Some nontoxic biological solutions also can weaken the polymer and cause the grippers to clamp down on their target.

This optical video snapshot shows the thermally actuated capture and retrieval of a cell mass from a 1.5-mm tube. It demonstrates the remote manipulation of a microgripper into a capillary tube (accessible at only one end) and retrieval of a cell mass that is dyed with neutral red stain.
In their lab experiments, the Johns Hopkins researchers used a microgripper, guided by a magnet, to grab and transport a dyed bead from among a group of colorless beads in a water solution. Team members also captured dozens of live animal cells from a cell mass at the end of a capillary tube. The cells were still alive 72 hours later, indicating that the capture process did not injure them. Also, the microgrippers captured samples from relatively tough bovine bladder tissue.
The experiments showed that the tetherless microgripper concept is viable and has great potential for medical applications, the researchers said. Gracias’ team is now working to overcome some remaining hurdles. As currently designed, each biologically compatible gripper can close on a target only once and cannot be reactivated to reopen and release its contents. (A similar device from the Gracias team, aimed at industrial microassembly applications, can be directed to both capture and release its load, but this requires chemicals that are not safe for patients.)
This pick-and-place microgripper was described in a recent article in the Journal of the American Chemical Society. Experiments using the devices were reported in the online early edition of Proceedings of the National Academy of Sciences for the week of Jan. 12 to 16.

This fluorescent micrograph features a single microgripper with live cells within its grasp. The tetherless microgripper was triggered to close and capture these live fibroblast cells with thermobiochemical cues.
Gracias, who also is affiliated with the Institute for NanoBioTechnology at Johns Hopkins, hopes to collaborate with medical researchers who can help to move the microgrippers closer to use as practical biopsy and drug delivery tools in humans.
In September, he received a $1.5 million New Innovators Award from the National Institutes of Health. He plans to use the five-year grant to develop an entire mobile, biochemically responsive micro- and nanoscale surgical tool kit.
The lead author of the PNAS microgripper article was Timothy G. Leong, who was a doctoral student supervised by Gracias. The paper’s other co-authors, all supervised by Gracias at Johns Hopkins, were Christina L. Randall, a doctoral student in the department of biomedical engineering; Brian R. Benson, a junior supported by a Provost’s Undergraduate Research Award; Noy Bassik, who is enrolled in an MD/PhD program involving the school of medicine and the department of chemical and biomolecular engineering; and George M. Stern, a master’s degree student in chemical and biomolecular engineering.
The Johns Hopkins Technology Transfer staff has obtained a provisional US patent covering the team’s inventions and is seeking international patent protection.
Funding for the research was provided by the National Science Foundation, the National Institutes of Health, and the Dreyfus and Beckman foundations.
For more information, visit: www.jhu.edu