Temperature-controlled fiber optic laser system is used for laser bonding of tissues.
Abraham Katzir, Tel Aviv University
One of the vital steps of most surgical procedures is bonding the edges of human tissue. Surgeons use sutures, staples or adhesives to close incisions in tissue, which encourages natural wound healing processes. An alternative method, which has been known for 30 years, is to use laser heating.
Two approaches have been used for laser-assisted bonding: laser welding, in which a laser beam heats the approximated edges of incisions, and laser soldering, in which a biological solder is applied onto the approximated edges of an incision, and then a laser beam heats the solder and the underlying tissue. Both methods show a lot of promise.
Laser heating methods are less traumatic to tissue because they cause fewer foreign body reactions, and potentially they lead to faster wound healing and result in much less scarring. Laser bonding is easier to master and faster to perform than suturing, and it generates an immediate watertight seal. In addition, laser bonding techniques lend themselves to endoscopic applications. However, laser welding and laser soldering have not been widely used in the clinical setting. This is probably because of the thermal damage that may be caused to the bonded tissue and because the initial strength of the repair has not been sufficient for most practical applications.
Controlling temperature
Only a narrow temperature range exists in which tissue bonding is expected to be efficient (roughly 65 °C). A lower temperature might yield no bonding, and a higher temperature might cause thermal damage to tissue (which could interfere with the natural wound healing process). In both cases, the immediate tensile strength is expected to be low. It has been known that thermal damage depends exponentially on temperature and linearly on time; therefore, the exact temperature at which laser bonding is performed is of critical importance.
Researchers in the applied physics group at Tel Aviv University in Israel who have studied laser bonding assumed that the accurate monitoring and control of the temperature of the site of the bonded tissue are essential to reduce thermal damage, to increase the immediate bond strength and to yield reproducible results. Thus, in recent work, our group delivered laser radiation through a suitable fiber to heat a spot on the surface of some tissue (Figure 1).
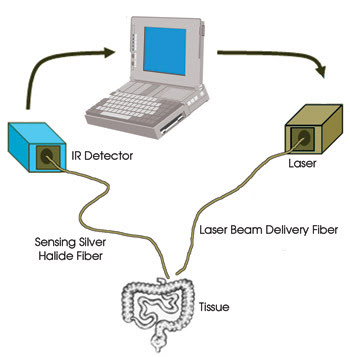
Figure 1. With the temperature-controlled fiber optic laser soldering system, laser energy is delivered through one optical fiber (right), which heats a spot on the tissue. An IR fiber optic thermometer (left) determines the temperature of the spot, based on the IR emitted. A PC adjusts the laser power so that the temperature stays constant.
The heated spot emitted infrared radiation whose intensity depended on the temperature of the spot. We used an IR-transmitting fiber to deliver the radiation emitted from the spot to an IR detector that monitored the spot’s temperature. The electrical signal generated by the detector was read by the electronics circuitry linked to a PC that controlled the power emitted by the laser. This system allowed the temperature of the heated spot (in vivo) to be controlled to within ±3 °C of a desired set temperature.
At first, our group used this fiber optic laser system for laser welding of tissue, but then we (and others) found that laser soldering yielded much better results. Thus we moved forward in performing laser soldering either with pure albumin heated by a CO2 laser or with albumin that contained indocyanine green (ICG) heated by a GaAs laser. (Both lasers were manufactured by Lumenis Ltd. of Yokneam, Israel.) In both cases, the solder is applied onto a cut, and the laser system heats the solder spot by spot. Each spot is heated (under temperature control) to 65 °C for 10 s.
In animal experiments, we found that, under such conditions, the immediate tensile strength of the soldered tissue was similar to, or higher than, that obtained with tissue adhesives. We found also that the strength of the soldered incisions increased with time, matching the strength of sutured incisions a few days after the laser bonding procedure.
Using the temperature-controlled system, we carried out laser soldering experiments in vitro and in vivo. We used a GaAs laser heating and albumin + ICG solder for in vitro experiments, including end-to-end colon anastomoses in the porcine model, end-to-end bowel anastomoses in the rabbit model and end-to-end soldering of the conjunctiva in the porcine model. These experiments produced excellent results. For example, in the bowel soldering experiments, the burst pressure was comparable to that of the native tissue (230 mm Hg).
CO2 laser heating with albumin solder was used for in vivo experiments involving laser bonding of cuts in the urinary bladders of rabbits, reconstruction of dural defects by laser soldering of fascia patches onto holes in the dura in the porcine model, closure of arteriotomy incisions in the femoral veins of rats and corneal transplant in the porcine model. In addition, laparoscopic laser soldering was used to repair ureteropelvic junction incisions in the porcine model. Laser bonding of cuts in all these animal experiments produced a tensile strength and no complications. The histopathological results did not show signs of thermal damage, and reduced scarring was achieved.
To illustrate the experiments using CO2 laser and albumin solder, we show the results of an experiment where eight cuts were made on the back of a farm pig. After four cuts were sutured and four were laser-soldered, we observed that the immediate strength of the soldered cuts was sufficient (Figure 2). The soldered cuts healed faster than the sutured cuts; moreover, soldering led to reduced scarring. The experiments were carried out by David Simhon and Tamar Vasilyev.
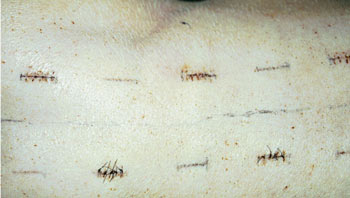
Figure 2. Seven days after the operation, sutured and soldered incisions on the back of a pig show that the soldered incisions result in much less scarring.
The team recently carried out a first series of clinical experiments in which inflamed gallbladders were surgically removed in an endoscopic procedure called laparoscopic cholecystectomy. In this case, four cuts were made in the abdomen; at the end of the procedure, two cuts were sutured, and two were soldered using the CO2 laser system with the albumin solder. The results were similar to those obtained in the porcine model. This research will help move laser bonding of tissue from the laboratory table to the operating table.
Meet the Author
Abraham Katzir is a professor and the incumbent of the Carol and Mel Taub chair in applied medical physics in the school of physics and astronomy at Tel Aviv University in Israel; e-mail: [email protected].