A start-up company is developing fibers that could provide real-time information as cardiologists open clogged arteries. Another start-up is using fibers to enable gastroenterologists to search the esophagus and colon for tissue that could become cancerous. And a headset that uses optical fibers to channel light to the brain could enable physicians to examine patients who have experienced brain injury without the cost and bulk associated with an MRI or CT scanner. These are just some of the ways that optical fibers could save lives in the years to come.
Improving cardiology
According to the American Heart Association, coronary heart disease is the leading cause of death in the US. This disease is primarily caused by atherosclerosis, a condition in which the arteries that provide oxygen and other nutrients to the heart become blocked by hard plaques that form initially from deposits of substances such as fats. The blockage of these blood vessels leads to chest pain and heart attacks.
Cardiologists use wire-mesh tubes called stents to open coronary arteries that have become blocked, delivering these stents either via the femoral artery in the groin or the brachial artery in the arm, through the aorta and into the coronary arteries.
The location of the blockage in the coronary arteries is commonly determined using a technique called fluoroscopy. A dye is injected into the arteries to provide contrast so that the doctors can see where the vessels are narrowed and likely to be blocked.
The stent, which is crimped onto a balloon angioplasty catheter, is then placed at the site of the blockage and expanded, so that blood can flow again. The balloon catheter is pulled out of the patient, but the stent stays there to keep the artery open.

Shown is a CorNova FiberHalo balloon catheter fully inflated inside the company’s Valecor Platinum stent. Courtesy of CorNova.
Sometimes, despite the presence of the stent, the inside of the blood vessel can fill with scar tissue and become narrow again, a problem that cardiologists call restenosis. Additionally, a blood clot, or thrombosis, can occur inside the blood vessel. Cardiologists attempt to prevent these events using various methods, including using another balloon catheter to further expand the stent and intravascular ultrasound or optical coherence tomography to determine if stent expansion is adequate.
According to Dr. Eric Ryan, founder and CEO of CorNova of Burlington, Mass., current methods of assessing stent failure are inadequate. Fluoroscopy produces a blurry image that only roughly shows narrowing of blood vessels. Other procedures, such as intravascular ultrasound, can measure the area inside the stent but require expensive equipment and additional time.
Moreover, all of these techniques require a catheter for imaging that is separate from the one used for balloon inflation. But with CorNova’s FiberHalo stent delivery balloon catheter, there’s no need for two catheters because it does both jobs. The catheter both expands the stent at the lesion site and measures the area inside the blood vessel.
The FiberHalo catheter contains hair-thin optical fibers through which various wavelengths of light can be shined on the interior of the artery. The light reflected from the blood vessel can be used to determine the area inside the blood vessel, which has been shown to have predictive value for restenosis and thrombosis.
The company plans to test the FiberHalo in preclinical trials with animals before it moves to human clinical trials. Before marketing a medical device worldwide, companies must get the CE mark, and companies marketing medical devices in the US also must receive what is called 510(k) clearance from the FDA. Typically, a stent is categorized as a Class III device, meaning it goes inside the patient, which makes obtaining FDA clearance difficult. However, because the FiberHalo catheters are surrounded by a stent, they are in Class II, so attaining FDA approval is easier.
Besides the FDA, medical devices have to win over insurance companies and the doctors themselves. Ryan pointed out that insurance companies already reimburse cardiologists for balloon catheters, which are the standard of care in the field, and FiberHalo catheters are modifying this existing technology. Therefore, he believes that his FiberHalo catheters will have a relatively easy time garnering the approval of cardiologists and insurance companies compared with devices that are trying to create a totally new market.
The FiberHalo technology was developed solely within CorNova, a private company funded by angel and institutional investors and the second business that Ryan has run solo. “I’m an engineer that went to medical school,” he said, “I had always wanted to integrate technology and medicine.”
Detecting cancer early
Many times, start-ups will fail even when they have a technological advantage. Such was the case for SpectraScience, a San Diego company that went bankrupt despite selling a device that can detect precancerous tissue in the colon in two seconds with 96 percent sensitivity.

The WavStat from SpectraScience can diagnose cancer in two seconds with 96 percent sensitivity. Courtesy of SpectraScience.
SpectraScience recently was revived by an experienced entrepreneur, Jim Hitchin. The company raised $5.5 million last year and recently bought rival company Media Spectra.
Hitchin believes that he can succeed where others have failed because he is going to concentrate on the esophagus instead of the colon. “In the colon, doctors are very entrenched with their current standard of care,” he said, “[The] current standard of care in the esophagus is very time-consuming, hurts and is expensive.”

This is a picture of the end of the WavStat device that combines an optical fiber within customized forceps. Courtesy of SpectraScience.
The device sold by SpectraScience is called the WavStat, a spectrometer that can be combined with endoscopes through fiber optics. It uses blue laser light to illuminate a suspect area, and that area autofluoresces. The information from the spectrometer goes to a personal computer that can analyze the tissue. The display lights up green or red, depending on whether or not the tissue is suspect.
“Since 85 percent of all cancers occur in the epithelial layer of tissue, autofluorescence should work for places in the body where you can place a fiber,” Hitchin said. “If we can detect precancers earlier, we can save a lot of people.”
The device has both FDA clearance and the CE mark. Therefore, doctors can buy it and use it right now.
In March, actress Natasha Richardson, wife of actor Liam Neeson, was skiing without a helmet on a beginner’s course when she suffered a head injury. She walked away feeling fine. An hour later, she was taken to the hospital, where her injuries were found to be critical; she died two days later.
Richardson’s story is reflective of the 1.4 million Americans who suffer traumatic brain injury each year, according to the Centers for Disease Control in Atlanta. Although patients often feel fine initially, they frequently experience lasting cognitive and neurological deficits later on. That is, if they live.
Current methods are inadequate
Current methods of assessing brain injury are inadequate, argues Dr. Luca Pollonini, who is doing research under George Zouridakis at the University of Houston. Pollonini said, “If I rush into the ER because my kid fell off his bike, and I want to be sure he is fine, the only way now to assess brain injury is to run a CT scan, which delivers radiation to the body, or an MRI scan, for which you have to be immobile. With pain and emotional distress emerging after a traumatic episode, it is hard for everybody to stay still during these examinations.”
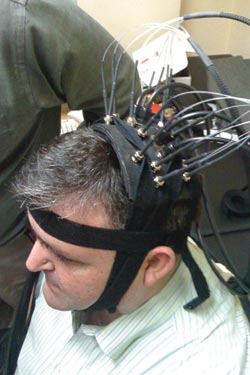
George Zouridakis wears headgear with optical fibers to give everyone an idea of what his device may look like in the future. He believes that optical fibers like these can be used with near-infrared spectroscopy to assess brain injury noninvasively. Zouridakis plans to combine a fiber optic headset with electrodes for EEG. Courtesy of G. Zouridakis.
Moreover, both CT and MRI scanners are huge machines that cost more than a million dollars and require specialized personnel to operate. Zouridakis and colleagues are developing a portable and cost-effective device to monitor brain injury. The researchers imagine that the device will fit on the head of the patient. The headset probably will combine fiber optics, laser diodes and electrodes to monitor the brain after injury.
The fiber-coupled laser diodes enable near-infrared spectroscopy – NIRS – which shows blood flow in the brain, and the electrodes enable electroencephalography – EEG – which measures electrical signals from the brain. Current NIRS and EEG equipment cost just $200,000 apiece, a bargain compared to CT and MRI.
The fiber-coupled laser diodes send light into the brain. The light becomes scattered as it enters brain tissue and is reflected out of the brain and measured by a set of sensors. The reflected light indicates what is going on in the brain. This can be done totally noninvasively or, in other words, without cutting a hole in the patient’s head.
In 2002, the Zouridakis group developed a portable EEG that was equipped with 256 data-recording electrodes. It was built by BioSemi BV of Amsterdam, the Netherlands. Pollonini is working with Nirox srl of Italy, and guest researcher Mikio Kubota of Seijo University is leveraging his influence with Shimadzu Corp. of Japan.