What you can’t see can hurt you. Cancer and other diseases can remain undetected until it’s too late. On the other hand, sometimes what you can’t see can help you: Increasingly, photons in nonvisible wavelengths are being used to spot disease, providing a picture that couldn’t be captured otherwise. For example, thanks to advances in detectors, smaller x-ray doses can provide more complete information. At the other end of the spectrum – literally – infrared imaging, which uses nonionizing radiation, is being put to use monitoring cancer and other ailments.
Taming x-rays
While not possessing superpowers, doctors for years have been able to do their own impression of Superman. Thanks to x-ray technology, they can look inside people and discover problems. But, as often is the case in fiction, this power comes at a price. The energetic x-ray photons ionize and damage living tissue as they pass through, increasing the risk for cancer. That can be a particular concern for diagnostic screening such as mammography. Because many healthy people will be exposed, the x-ray dosage must be kept to a minimum to ensure that the benefit far outweighs the added risk.
A key to reducing dosage lies within the detector, said Behnam Rashidian, a senior life sciences product manager at Dalsa Corp., a Canadian company based in Waterloo, Ontario, that makes, among other things, image sensors for x-ray medical applications.
In this implementation, a CCD or CMOS sensor sits behind a scintillator that turns the incoming x-rays into visible photons at a wavelength of about 550 nm. One of the ways the sensor can help cut x-ray dosage is by having high quantum efficiency, Rashidian said. “When you have higher quantum efficiency, it means you use more of the photons that are available.”
Dalsa has a relatively new line of CCD sensors with quantum efficiency above 70 percent, which Rashidian said is as much as double the efficiency of other front-illuminated CCDs. Combining that with automatic exposure control and sensor cooling can cut x-ray dosage by a factor of two or three as compared to what’s needed without these enhancements.
Such a reduction will be even more important in the future because of a trend toward 3-D imaging, which studies have shown makes for a better diagnostic tool. For x-rays, this involves capturing 2-D images from different angles and then reconstructing the full 3-D data. Keeping the overall patient dose the same while going to 3-D can be done only if the individual 2-D images require less dosage than normal.
Economics an issue
Economics dictates that diagnostic imaging, particularly in the case of breast cancer screening, must not take any longer in the 3-D case than it does for traditional 2-D imaging. “This trend imposes technological challenges as higher-speed detectors are required,” Rashidian said.
He added that CCD and CMOS sensors have the required speed. That, he asserted, is not necessarily the case for other x-ray imaging technologies.
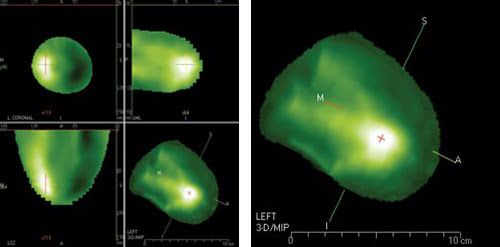
Increased absorption of the near-IR indicates an abnormal network of blood vessels and possible cancer. Here a breast cancer diagnostic tool based on the near-IR displays a high-grade ductal carcinoma as a bright spot in the volume-rendered image (right) and in the 2-D views (left). Courtesy of Imaging Diagnostic Systems Inc.
Of course, one way to eliminate the cancer concerns brought on by x-rays is to get rid of the offending photons altogether. For that reason and others, a number of researchers and companies are looking into infrared imaging.
One such company is Imaging Diagnostic Systems Inc. of Plantation, Fla., which is developing a breast cancer diagnostic tool intended to be an adjunct to traditional x-ray mammography. The difference is that the new technique is based on near-IR wavelengths.
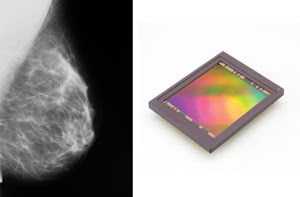
X-ray mammography is the gold standard in breast cancer detection, but it involves ionizing radiation (left). New image sensors with high quantum efficiency (right), along with other improvements, could cut dosages in half. Courtesy of Dalsa Corp.
The patient lies facedown in the scanner, with one breast hanging into a specially designed chamber. A laser diode operating at 808 nm scans the breast, and a ring of silicon photodiodes picks up the resulting optical absorption. The scanning plane moves until the entire breast is imaged. If a tumor is present, the abnormal network of vessels around it will appear as a localized region of increased optical absorption.
Steven L. Ponder, the company’s director of advanced development, pointed to two benefits of this scheme. “The advantages are patient comfort, since the breast is never touched by the scanner, and the use of nonionizing radiation.”
He added, “There are no cumulative hazards associated with multiple scans, so patients may have frequent follow-up exams without concern.”
The laser diodes and photodetectors are standard devices, although Ponder noted that better performance from either could be a plus. Other improvements, which are ongoing, come from the proprietary reconstruction algorithms used to make sense of the data. The company hopes this year to have premarket approval from the FDA to sell the device within the US.
ART Advanced Research Technologies of Montreal is another company working on a near-IR breast cancer imaging solution. The company uses time-domain optical imaging, firing a brief laser pulse into tissue and capturing the resulting photons as they emerge over time. One result from this imaging technology could be a reduction in the number of breast biopsies, many of which turn out to be unnecessary. The company’s device has received regulatory approval for commercialization in Canada and Europe but not in the US.
Going long
Finally, longer-wave IR also is being investigated for biomedical imaging. Sensors that capture such thermal IR can pinpoint heat and so can help detect local hot spots, thereby providing vital information. In addition to disease, thermal IR can monitor surgery, particularly anything that involves heating by a laser or other means.

Thermal infrared cameras image heat, which can be useful for bio-medical imaging or for monitoring heat-producing procedures. Courtesy of Flir Systems Inc.
Jason Styron, business development manager at infrared camera maker Flir Systems Inc. of Portland, Ore., noted that these types of applications present a challenge for his company. Flir, he noted, traditionally sells into the industrial market, which is less sensitive to price than medical imaging applications.
However, there is hope on the cost front, he said. A few years ago, the standard price for a thermal IR camera might have been $35,000. “We have reduced that to $15,000, while providing better performance, better connectivity and a smaller package size.”
With regard to performance, one improvement has been that the number of pixels has gone up. Today a state-of-the-art handheld thermal camera offers 640 × 480 pixels, four times the number provided by previous systems but still far less than the pixel count of a visible camera.
Part of the explanation for the smaller price tag has been improvements in technology, with contributing factors including design changes in the fundamental detector cell layout on the chip and more efficient manufacturing. Another reason for the price drop has to do with increased volume.
As for the future, Styron noted that there may come a time when material costs will not allow continued price decreases, but he doesn’t foresee that happening anytime soon. “We do expect the trend to continue.”