OCT identifies diagnostic markers in esophageal malignancies.
Florian Bazant-Hegemark and Dr. Nick Stone, Gloucestershire Royal Hospital, and Dr. Gordon McKenzie and Jon Holmes, Michelson Diagnostics Ltd.
Optical coherence tomography (OCT) has been applied successfully in ophthalmology since the early 1990s, a success story triggering research efforts in the imaging of early epithelial cancers. Typical nonophthalmic systems achieve a resolution of 10 to 20 μm in dense tissue, but imaging depth is limited to little more than 1 mm.
Furthermore, early systems did not provide sufficient image contrast or penetration depth, and they scanned too slowly to combat motion blur when used in vivo. However, advances in the underlying technology (particularly light sources) have led to developments and even products with a high scan rate and much improved image resolution and quality.
Because of these developments, there is a realistic short-term opportunity to introduce OCT as a clinical tool for the assessment of surgically removed cancer. The cancer already would have been located by conventional means, and OCT would aid in determining whether the tissue margins were disease-free. Currently, tissue at the margins is surgically removed and investigated by histopathology, but this process can take several days and can require a patient to return to the operating room.
Researchers at Gloucestershire Royal Hospital in the UK decided to investigate whether diagnostic markers in esophageal tissue could be detected with real-time OCT using a state-of-the-art scanner. They wanted to see whether a large-scale trial of this technology for improving the present assessment of surgical outcome would be warranted.
6-mm-wide images
For the study, an OCT microscope from Michelson Diagnostics Ltd. of Kent, UK, was used. This system uses an ~1300-nm swept-source laser and can capture real-time images 6 mm wide with better than 10-μm optical resolution in both axial and lateral directions over the 1-mm imaging depth (in tissue). The measurements were performed by the researcher on site — in the operating theater — and did not disturb staff or routine procedures (Figure 1). Although the acquisition time for a B-scan (cross section) is short, the procedure for scanning several B-scans of a sample typically took a few minutes because of sample handling and positioning.
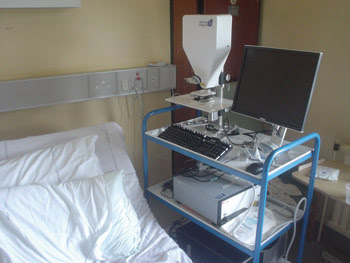
Figure 1. The mobile Michelson Diagnostics Ltd. OCT unit is shown in a typical endoscopy suite at Gloucestershire Royal Hospital in Cheltenham, UK.
Esophageal tissue samples from 11 patients undergoing routine treatment at Gloucestershire Royal Hospital NHS Foundation Trust in Cheltenham, UK, were scanned immediately after excision before undergoing routine histopathological assessment. OCT scans and histopathology results were assessed in cooperation with an expert histopathologist.
The samples fell into two groups:
(1) Endoscopic mucosal resections, which are obtained routinely from patients who need monitoring of precancerous lesions with a risk of progressing to cancer.
(2) Lymph nodes from esophagectomies, which are investigated routinely to assess spread of the tumor.
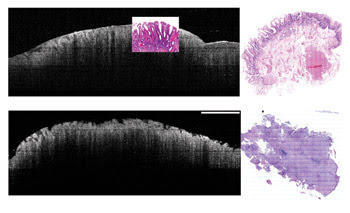
Figure 2. These images show endoscopic mucosal resections of nondysplastic esophageal specimens (noncancerous top, cancerous bottom). On the left is the OCT B-scan, and on the right is a cross section of the whole stained histopathology sample. The histopathology image of the cancerous lesion shows a complete lack of structure, with ulcerations on the surface, and the corresponding OCT image shows these ulcerations. The shape of these ulcerations differs from the smooth villus structure of the glands seen in the top images. Scale bar = 1 mm.
Images of endoscopic mucosal resections show features from within about 1 mm below the surface. Figure 2 shows endoscopic mucosal resections of a nondysplastic sample, including areas of squamous and glandular epithelia. These two types of epithelia are visually different on the OCT image.
Figure 3 shows representative images from lymph nodes that were under investigation for tumor spread. There is a clear structural difference between the healthy lymph node and the invaded, cancerous counterpart. Although OCT imaging depth does not allow full penetration of the specimen, germinal centers of cell clusters can be seen.
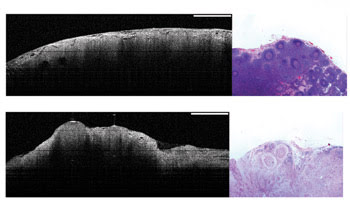
Figure 3. These OCT images (left) and histopathology slides (right) show a healthy lymph node with dilated sinuses, reactive follicles and lipid vacuoles (top images) as well as an invaded, cancerous lymph node, with dense follicle and lipid vacuoles (bottom images). Scale bar = 1 mm.
These findings are promising for the design of a large-scale trial: Diagnostically relevant features have been identified, allowing staff to be trained for double-blinded trials to establish sensitivity and specificity. Furthermore, the results suggest that there is considerable potential for in vivo application of OCT. Clinical testing of a prototype instrument equipped with an endoscopic OCT probe will start during 2008.
Another development that is expected to improve OCT images further is scanning at a shorter wavelength — close to 1 μm. A consortium consisting of Michelson Diagnostics, University of Cardiff, Gloucestershire Hospitals NHS Foundation Trust, National Physical Laboratory, semiconductor specialist Kamelian Ltd. and medical imaging systems specialist Tactiq Ltd. has started development and testing of such a system.
OCT researcher Wolfgang Drexler, director of research and a professor at the University of Cardiff department of optometry and vision science, said, “We believe that images acquired at 1-μm wavelength will offer improved contrast and resolution that will help clinicians to distinguish between healthy and cancerous tissue.” The project is supported by the UK government with a £325,000 grant.
OCT has a realistic chance of guiding biopsy and, hence, of reducing the need for biopsy from distinctly normal areas. This could speed up referral for cancer operations, reduce the pressure on overloaded pathology departments, improve planning and follow-up of surgery, and streamline overall cancer management.
Acknowledgments
This work is funded by the Pump Prime Fund of Cranfield University and by Gloucestershire Hospitals NHS Foundation Trust. The clinical prototype was kindly provided by Michelson Diagnostics Ltd.
Meet the authors
Florian Bazant-Hegemark is a researcher in the biophotonics research group of Gloucestershire Royal Hospital in Cheltenham, UK.
Nick Stone is a consultant clinical scientist and head of the biophotonics research group at Gloucestershire Royal Hospital.
Gordon McKenzie is applications director, and Jon Holmes is chief executive at Michelson Diagnostics Ltd. in Kent, UK; e-mail: [email protected].