Researchers use targeted fluorophores for flow cytometry in clinical trials and for optical imaging in living mice.
David L. Shenkenberg, Associate News Editor
All it takes is one tumor cell circulating through the bloodstream to turn a manageable tumor into metastatic disease, and just one tumor cell can cause cancer to recur in patients who have received treatment. Although high-resolution clinical imaging methods such as CT and MRI can detect masses much smaller than 1 cm, they cannot identify an individual tumor cell.
Flow cytometry of blood samples can detect tumor cells that were circulating in patients, but blood samples represent only a small fraction of a patient’s blood. For example, blood makes up about 7 percent of the body, so a 5-ml sample from a 100-kg man would represent only1/1400 of his blood. Therefore, all of a patient’s blood must be measured to ensure detection of all circulating tumor cells.
Optical imaging of surface veins potentially can enable doctors to search all the blood within patients for circulating tumor cells. Furthermore, optical imaging is noninvasive, in contrast with taking blood samples from patients. Prior to optical imaging, cancer-targeting optically detectable labels must be administered. However, antibody-based labels are used to mark tumors in blood and tissue samples, and, in the body, white blood cells rapidly eat antibody-labeled cells.
Instead of using antibodies, researchers from Purdue University in West Lafayette, Ind., and from Mayo Clinic in Rochester, Minn., used fluorophores directly bound to the targeting molecule, folate. They used optical imaging with the folate-conjugated fluorophores to noninvasively detect single circulating tumor cells in living mice, a step toward translating the method to human patients.
While developing the imaging method, the investigators also used the folate receptor-targeting fluorophores and flow cytometry to detect circulating tumor cells in blood samples drawn from cancer patients in phase II clinical trials with an accuracy of about two cells per 2 ml.
The folate-conjugated fluorophores are superior to antibody labels for flow cytometry because antibody labeling necessitates an extra purification step that not only adds more time to the process but also can reduce cancer cell detection accuracy.
The researchers chose the folate receptor because it is overexpressed in 40 percent of tumors and more so in certain tumors. For example, more than 90 percent of ovarian and endometrial cancers overexpress the receptor, and it is overexpressed in more than 78 percent of non-small-cell lung carcinomas, the type of cancer that smokers tend to develop.
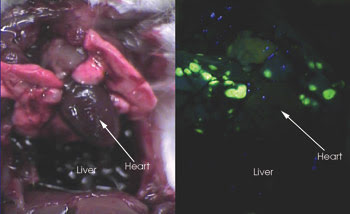
Researchers targeted fluorescein or rhodamine to the folate receptor, which is overexpressed by many tumors. They injected a mouse with folate-fluorescein and captured this image of metastatic lung cancer nodules. Only cancerous tissue is labeled (green), whereas healthy tissues — such as the heart and liver — are not labeled.
The scientists reported the imaging of mice and the clinical trial data in the July 10 issue of PNAS. Philip S. Low, principal investigator from Purdue University, stated more recent information regarding the clinical trial. The participants so far have included 21 ovarian cancer patients and nine healthy volunteers. Folate-Alexa-Fluor 488 was mixed into 2-ml blood samples and placed in a flow cytometer. Made by Invitrogen Corp., AlexaFluor 488 is a chemically modified version of fluorescein with high fluorescence intensity and photostability.
Flow cytometry reported an average number of 0.35 false-positive hits for cancer cells per 2-ml blood sample acquired from healthy volunteers. Therefore, in initial clinical trials, the method has a demonstrated accuracy of less than one cell per 2 ml of blood. In 2-ml blood samples from ovarian cancer patients, the technique reported an average of 109 cells, somewhat skewed by patients with high levels of cancer cells. The median number of cancer cells reported was 35, a number that more accurately represents the results. Low said that these data indicate that their method successfully detects cancer cells in patients.
To detect circulating tumor cells in living mice, the researchers injected the mice with folate-rhodamine, placed a microscope objective in the ear of each mouse and monitored blood flow in an arbitrary vessel. They used two-photon excitation because it enabled them to see more deeply through the mouse ear and thus to more clearly visualize the blood vessel.
They used a Ti:sapphire laser from Coherent Inc. of Santa Clara, Calif., for two-photon excitation, a microscope made by Olympus of Tokyo to see the blood vessel, and a photomultiplier tube from Hamamatsu Corp. of Bridgewater, N.J., to detect fluorescence.
The researchers modified the microscope so that it scanned back and forth across a blood vessel in the same spot, rather than scanning back and forth across a large field of view. This modification increased the scan rate 500 times and enabled them to view blood vessels as large as 1 mm. Low said that they probably could see even larger blood vessels, but 1 mm is about as large as blood vessels grow in mice.
Because blood flows faster in larger vessels, scanning larger blood vessels increases the imaging speed. If the procedure is translated to human patients, faster imaging means that patients can undergo the procedure in less time and that doctors will receive results more quickly.
Using two-photon microscopy, they could see single cancer cells traveling through the bloodstream. They could quantitate these single cancer cells with a signal-to-noise ratio of 15:1.
By direct illumination with a fluorescent lamp, they also could distinguish solid masses from healthy tissue. “The tumors are very bright,” Low said, “You can see them with the naked eye.”
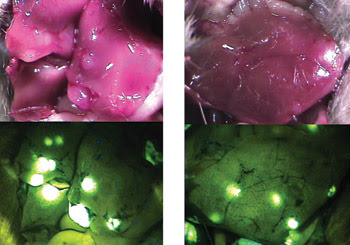
Folate-conjugated fluorophores label tumors so well that they can be seen with the naked eye and a light source. Above, fluorescence imaging with folate-fluorescein shows liver cancer nodules in a mouse. In translational research with living mice, scientists quantitated circulating tumor cells using the fluorophores and optical imaging.
Low said that the imaging method will help surgeons see the margin of tumors, so that they can excise entire tumors without leaving behind any cancer cells. He also said that it will help detect recurrence, particularly of ovarian cancer. Forty percent of ovarian cancer patients experience a recurrence of the disease, and the cancer usually is not caught until it has spread through the endometrium or throughout the body.
Low said that, to translate optical imaging of mice to cancer patients, they need to change a few aspects of their imaging setup. Instead of a microscope objective, they probably would use an optical fiber about 0.1 mm in diameter, about the thickness of a human hair. They would tape the fiber to the patient to prevent it from moving if the patient wiggles around, whereas they used anesthesia to immobilize the mice. They would use the fiber to monitor the surface veins of the skin for circulating tumor cells. They also could use the folate-conjugated fluorophores and a fiber to look for fluorescently labeled masses in the body, Low said. In any case, they need FDA approval to use the folate-fluorophore conjugates in patients. Fluorescein already has approval for other uses in humans.
More recently, the scientists tied folate to near-IR fluorophores ranging from 700 to 900 nm. “All of these show excellent tumor specificity,” Low said. They are better for imaging because tissue is transparent to near-IR wavelengths. The researchers also have conjugated fluorophores to a molecule that targets prostate cancer.
“Clinical applications for nontoxic optical imaging abound if the optical imaging agents can be selectively targeted to the pathological cells of interest,” Low said.