
Low-light imaging and detection offer insight into plant development
Gary Boas
Plants as well as humans have evolved circadian clocks that generate self-sustained daily rhythms, enabling them to adapt to daily fluctuations in their environment — fluctuations in light and temperature, for example. Indeed, studies have shown that plants with an internal-clock cycle in synch with the daily changes in their environment maintain a competitive advantage over plants with no such timekeeping mechanism.
The plant hormone auxin has been linked to a number of aspects of plant growth and development — most notably, growth toward light and away from gravity. In the August issue of PLoS Biology, Michael F. Covington and Stacey L. Harmer of the University of California, Davis, reported a study in which they explored whether the circadian clock is involved in the auxin signaling pathway —that is, whether it controls plant transcriptional responses to auxin.
To achieve this, they needed an auxin-sensitive reporter gene with activity that they could follow on a circadian (daily) time scale. They made transgenic plants expressing firefly luciferase under the control of a synthetic auxin-responsive promoter to demonstrate that the transcriptional responses are regulated by the circadian clock — explaining decades-old observations that sensitivity to the hormone varies over the course of the day.
They encountered some obstacles during the study, initially observing that various plant tissues exhibited different regulation of auxin signaling. They addressed this challenge by focusing on a particular plant tissue. In a follow-up study, though, they investigated the responses of a range of plant tissues to auxin treatment — to this end using the XR/MEGA-10Z ICCD camera manufactured by Stanford Photonics Inc. of Palo Alto, Calif.
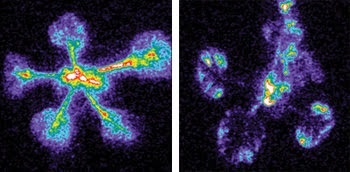
Researchers are investigating the responses of various plant tissues to treatment with the plant hormone auxin by using an ICCD camera that enables low-light detection and imaging with high sensitivity. These photos, acquired with the camera, show that the various promoters drive luciferase activity in differing parts of the plant. The researchers used transgenic plants expressing firefly luciferase to study the role of the circadian clock in the auxin signaling pathway.
This product was designed to provide single-photon imaging with low background levels. It has dark counts of fewer than 50/s for a 1400 x 1000, 10-μm-pixel array. It has a high-quantum-efficiency Peltier-cooled GaAsP photocathode that is used with a special microchannel plate design and a fiber-coupled interline transfer CCD to produce images at frame rates that are as high as 30 to 1000/s with virtually no dark counts. Proprietary software allows the camera to provide “photon accumulation imaging” — a summation of selectable image frames containing sparse single-photon data into gray-scale images that represent minutes’, hours’, days’ and even weeks’ worth of time-lapse, low-light images that have been the goal of the Davis group and others studying circadian rhythms in plant and animal studies.
Mike Buchin, president and owner of Stanford Photonics, recalls that the camera actually has its roots in two worlds that don’t often overlap — the military and the life sciences. In the early 1990s, the company built a number of GaAsP-based ICCD cameras for the US Navy as part of its ongoing night-vision business. Through this program, the company gained exclusive access to the technology for scientific imaging, and a small number of key research centers in the US were able to dramatically improve their live-cell and single-molecule imaging capabilities via GaAsP ICCDs. Unfortunately, the company that was making the tubes was purchased, and the product was mothballed. Throughout the 1990s, Stanford Photonics continued to port advanced military night-vision technologies into biological imaging applications. Ultimately, the company was able to find a supplier that was willing to resurrect the GaAsP material.
Harmer said that the camera’s high sensitivity and low noise made it extremely well-suited for detecting the very low light levels emitted by the transgenic plants they had produced. “The remarkable sensitivity of this camera has allowed us to define the kinetics of auxin responses in different tissues in a way that wasn’t possible using our other CCD cameras,” she said. “Our exposure times are ten times faster than what we have to use for our other cameras.”
Investigation of the tissue specificity of clock regulation of auxin signaling is ongoing. The researchers are exploring whether clock regulation of plant responses to gravity varies according to tissues, for example. Also, until now, the clock has seemed to run in the same manner in different organs — in leaves versus roots, and so on. However, the recently acquired data suggest that the connection between the clock and auxin pathways is in fact wired differently among the organs. They plan to examine this question further by looking at the tissue-specific temporal expression of central clock genes compared with various clock outputs.
Finally, Harmer said, “We have evidence that the clock regulation of auxin transcriptional responses changes during organ development, and we are going to use [the camera] to define when and in what cells this transition takes place. Then we can start to dissect where the auxin and clock signaling pathways intersect and how this changes over developmental time.”
Contact: Stacey L. Harmer, University of California, Davis; e-mail: [email protected]; Mike Buchin, Stanford Photonics; e-mail: [email protected].
Published: September 2007