
Visualizing nutrient passage through chain-forming bacteria
Michael J. Lander
Some bacteria perform a biological juggling act, producing oxygen through photosynthesis while conducting oxygen-sensitive nitrogen fixation. How nutrients flow between the cells without disrupting the processes has not been totally clear. However, scientists at Portland State University in Washington, at Lawrence Livermore National Laboratory in Berkeley, Calif., and at the University of Southern California, Los Angeles, have used images to better understand the nature and pace of the exchange.
For detailed imaging of element assimilation in filamentous cyanobacteria, Douglas G. Capone and colleagues turned to nanoscale secondary ion mass spectrometry. To rapidly growing cultures of the bacteria, the researchers added carbon dioxide and nitrogen gas enriched in the stable isotopes carbon-13 and nitrogen-15, respectively.
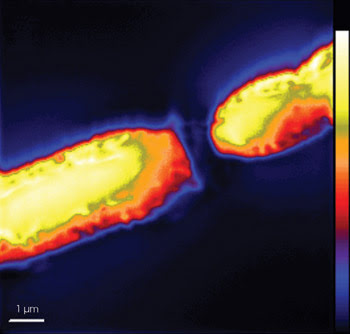
Secondary ion mass spectrometry images of a cyanobacterium filament show nitrogen distribution among interconnected cells. The black region dividing the cells represents a heterocyst’s polar plug, a polysaccharide-rich zone low in nitrogen. Reprinted with permission of The ISME Journal.
Capone noted that the technique was unique in allowing visualization of nitrogen in the cell using nitrogen-15 instead of the radioisotope nitrogen-13. Unlike carbon and other elements with long-lived radioisotopes that are readily available, nitrogen-13 is short-lived and very hard to obtain.
Software helped the scientists create clear images of the cells based on ratios between the elements and their isotopes. In contrast with other methods, the system generated images showing subcellular details, permitting more accurate determination of the isotopes’ destinations.
Where a cell was about to divide, the scientists saw the concentration of fixed nitrogen peak significantly. They attributed this action to the formation of structures involved in division, which comprise nitrogen-containing proteins.
They were more surprised to find that most of the strands’ specialized nitrogen-fixing cells, called heterocysts, contained comparatively low levels of freshly fixed nitrogen and carbon. Instead, these elements were concentrated in the photosynthetic cells connecting the heterocysts, which indicated that the nitrogen fixed within the heterocysts was exported rapidly to these carbon-fixing cells.
Heterocysts, they reasoned, under-went little growth and thus needed just enough of the substances to maintain their nitrogen-fixing machinery. As cells divided and the strand became longer, the researchers noted that newly fixed nitrogen and carbon became scarce in the centermost cell between existing heterocysts. They concluded that such cells represent newly transforming hetero-cysts, the observation of which may help clarify the process of cell differentiation in the cyanobacteria.
The researchers plan to use the spectrometry technique to examine a more complex bacterium in which photosynthesis and nitrogen fixation are not clearly compartmentalized. In this context, the method’s ability to finely resolve multiple stable isotopes at once may prove critical. It also could find use in analyzing biofilm formation and other processes involving intercellular exchanges.
The ISME Journal, August 2007, pp. 354-360.
Published: September 2007