
High-resolution MRI method generates virtual mouse brains
Gwynne D. Koch
The prevalence of diverse imaging methods and the use of mouse models to study human diseases have generated massive amounts of data and provided insight into brain structure and function. However, comparing results from studies is difficult because wide ranges of image acquisition strategies and tissue contrasts have been employed.
Recognizing the need for methods to effectively phenotype new models and infrastructure to collect and organize the growing body of imaging data, a consortium of six institutions — known as the Mouse Biomedical Informatics Research Network (MBIRN) — has been established.
With funding from the National Center for Research Resources, a component of the National Institutes of Health, the consortium has developed a series of integrated methods to generate 3-D, high-resolution digital images sharp enough to detect subtle structural changes in tissues and vessels caused by genetic differences. MBIRN also has developed the necessary computer infrastructure to collect and store up to 2 TB of data and to make it all available on the Web.
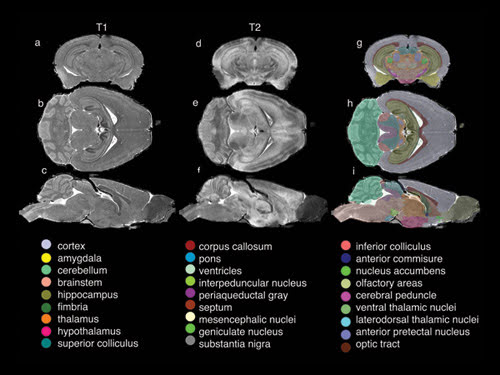
Using statistical algorithms and quantitative volume measurements, the researchers segmented the data into 33 of the most critical structures of the brain. As shown in the composite image above, these structures are computer-enhanced to produce color-coded and -labeled volume renderings of selected anatomical details, such as the ventricles and hippocampus, in three dimensions.
The scientists are performing conventional optical histology and electron and magnetic resonance (MR) microscopy to study the connections between genes and brain structure with the goal of providing insight into the basic mechanisms of cancer and neurodegenerative disorders such as Alzheimer’s and Huntington’s diseases.
Led by G. Allan Johnson, researchers at Duke University Medical Center in Durham, N.C., are focusing on MR histology, a term they coined for using MR microscopy to characterize tissue structure. The team has developed a standardized protocol that enables acquisition of 3-D MR images of the fixed, stained mouse brain at 43-μm resolution in less than 30 min, as well as a higher-resolution protocol with isotropic spatial resolution of 21.5 μm that can be executed in 2 h.
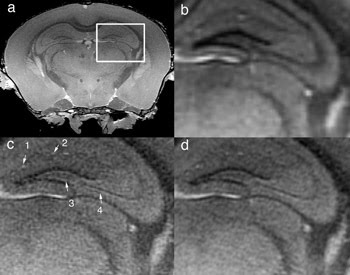
Researchers have developed a series of integrated methods that enable acquisition of 3-D MR images of mouse brains at up to 100,000 times higher spatial resolution than a routine clinical head scan. The image above shows a representative 21.5-μm slice of a region through the hippocampus (a), of the image reconstructed at 43 μm (b), and of it reconstructed at 21.5 μm with the team’s expanded dynamic range partial Fourier method with reduced acquisition time (c) and at 21.5 μm with full Fourier scan (d).
MR images are acquired at up to 100,000 times higher spatial resolution than a routine clinical head scan. To achieve this dramatic increase in resolution, the team devised a series of integrated steps, employing an active staining method, or perfusion, with a contrast agent mixed with a fixative to simultaneously preserve the tissue and enhance the MR signal by nearly 10 times by reducing the spin lattice relaxation time.
The researchers also developed a method for sampling Fourier space with expanded dynamic range to amplify high-frequency information and coupled that to an asymmetric sampling, or partial Fourier, acquisition and reconstruction strategy. As Johnson explained, there is redundancy in the data gathered in an MRI experiment that can be exploited.
Their method — active staining and extended dynamic range with partial sampling — reduces the scan time by nearly 50 percent. The entire process of specimen preparation, image acquisition, reconstruction, automated analyses, and data archiving and distribution has been streamlined and integrated to enable high throughput.
“With these methods and the MBIRN infrastructure, we can make the method available worldwide so the community of neuroscientists studying mouse models of genetic disease can have a common reference, a common database and a common standard for comparison of the impact of genotype on morphologic phenotype,” Johnson said.
The system employs an Oxford 8.9-cm vertical bore magnet operating at 9.4 T. The researchers have adapted a GE Excite clinical imaging console for use at 400 MHz — a much higher frequency than is routine in clinical use. The result is a system that enables the team to use imaging strategies developed in the clinic for imaging the mouse, accelerating the transition of their research from “bench to bedside.”
According to Johnson, MR histology does not replace, but rather complements, conventional optical methods. Because MR histology is nondestructive, the brain specimen does not have to be removed from the cranium, and there is no shrinkage from dehydration or distortion from sectioning. The same specimen may be scanned multiple times, and when MR studies are complete, conventional histology can be performed. Also, by adjusting the acquisition parameters, a wide range of soft-tissue contrasts can be derived from the same tissues. Tissues can be differentiated based on variances in proton density, on spin lattice, and on spin-spin relaxation times and diffusion.
Although the team has acquired images at 10 μm, to achieve the high throughput desired, it is limited to a spatial resolution of about 20 μm. According to Johnson, the fundamental resolution limit is imposed by diffusion — the Brownian motion of the water molecules that occurs during the time (~10 ms) required for the spatial encoding.
To improve understanding of the underlying contrast mechanisms, the consortium is creating an extensive atlas to correlate the MR images with those from more traditional optical methods. The group also is developing the imaging technology to translate the spatial resolution obtained in fixed specimens to live animal studies.
NeuroImage, Aug. 1, 2007, pp. 82-89.
Published: September 2007